Note: Descriptions are shown in the official language in which they were submitted.
CA 02463491 2004-04-13
WO 03/030722 PCT/US02/32444
MAGNETIC FIELD ENHANCEMENT OF TUMOR TREATMENT
TECHNICAL FIELD
The present invention has to do with the treatment
of cancerous tumors.
BACKGROUND ART
A central problem in cancer treatment is that of
preserving healthy tissue while destroying cancerous tissue.
Although radiation therapy generally involves the focusing
of radiation on a tumor, at least some healthy tissue
generally is located in the irradiated field. This healthy
tissue is exposed to and to some degree damaged by the
radiation. In chemotherapy healthy tissue is exposed to the
chemotherapy agent and may be damaged.
Moreover, research has shown that much of the
effect of radiation therapy and a substantial component of
some approaches to chemotherapy are mediated by free radical
effects in tumor tissue. The mechanisms whereby free
radicals produce tumor cell death include direct enzymatic
effects, DNA damage and induction of apoptotic pathways.
Our understanding of the physics and chemistry of
free radicals and paired radicals has gradually increased
over the past ten years. A free radical is any chemical
species capable of an independent existence that has an
unpaired electron in its valence shell. The presence of an
unpaired electron in the valence shell causes free radicals
to be paramagnetic and exhibit magnetic properties when
exposed to a magnetic field.
Free radicals may be formed by any of several
mechanisms including but not limited to:
Ultraviolet induced homolytic fission as may be
encountered in laser ablation therapy of tumors;
CA 02463491 2004-04-13
WO 03/030722 2 PCT/US02/32444
Specific chemical reactions as encountered with
pharmacological chemotherapy e.g. bleomycin;
Ionizing radiation as the result of external beam
irradiation, antibody directed or site selective
radio-nucleotide administration or through implantation
radiotherapy e.g. prostatic brachyotherapy;
Thermal induction as in hyperthermic therapy; or
Ultrasound induced acoustic cavitation.
Free radicals once generated may recombine. The
biologic effects of free radicals in tissue are determined
by the net reactive fraction namely the "escape" population
that does not recombine rapidly. Factors, which influence
pair recombination, include the viscosity of the reaction
environment, temperature, bystander effects and the quantum
state of the free radical. The quantum state of the free
radical is defined by the applicable Schrodinger equation
(H~I'=ELI') where H is a Hamiltonian operator and LY are sets of
wave functions (Eigenfunctions). The Eigenfunctions are
defined by a set of four quantum numbers: n-the principal
quantum number, 1-the orbital quantum number, M1_the
magnetic quantum number and MS_the spin quantum number. Of
particular significance to this discussion is the spin
quantum number.
The spin quantum number for an unpaired orbital
electron can assume one of two values either +1/2 or -1/2.
The wave distribution function determined by spin
quantitization is a vector quantity and subject to influence
by a superimposed magnetic field. When two electrons share
an orbital space they must have opposite spin polarity.
This phenomenon is dictated by the Pauli Exclusion Principal
that postulates that no two electrons can occupy the same
quantum state.
CA 02463491 2004-04-13
WO 03/030722 3 PCT/US02/32444
Spin polarity is conventionally referred to as up
spin (T) +1/2 or down spin (.~)-1/2. Shared valence
electrons in the formation of chemical bonds also must have
opposite spin polarity. When covalent bonds are severed as
in the formation of free radicals spin polarity is
preserved.
The unpaired electron in the valence orbital of a
free radical in a magnetic field will precess in a manner
comparable to Larmor precession described for charged
particles in classic electrodynamics. Quantum precession
leads to spin phase transitions between the singlet state
where antiparallel spin vectors apply and triplet states
where parallel spin vectors apply. The singlet state is
favorable for recombination because antiparallel spin
orientation is preserved and a covalent bond can be
established. Triplet state configurations are unfavorable
for recombination because parallel spin orientation is
induced. In a magnetic field there are three triplet state
configurations, which are vector quantities that due to
precession in the magnetic field are no longer energy
equivalent and are said to be nondegenerate.
The strength of the applied magnetic field, which
maximizes the spin phase mixing effect, is dependent on the
quantum state of the free radical or the system of free
radicals. In general optimum phase mixing is achieved at
relatively low magnetic field strengths (0.1-10.0 mTesla)
within the hyperfine coupling energy levels of the radical
pair.
The singlet state (S1) characterized by
antiparallel spin vectors will prevail in the absence of a
magnetic field when homolytic fission of a covalent bond
occurs to form a free radical pair. In the presence of a
magnetic field of appropriate strength, the triplet states,
CA 02463491 2004-04-13
WO 03/030722 4 PCT/US02/32444
T-1, To and T+1 are equally probable energy states and are
distinct and nondegenerate. The theoretic distribution
between singlet and triplet states will be 25o singlet and
75$ triplet. Such a distribution will theoretically
increase the effective concentration of escape radicals by
75$. In experimental situations the yield is limited by
non-quantum factors including viscosity effects,
temperature, diffusion and bystander effects. However,
increases in escape radical reactivity of 20-40o are
documented in experimental systems where free radical escape
reactions are measured.
DISCLOSURE OF THE INVENTION
The present invention is a method of treating a
tumor in which an elevated concentration of free radicals is
created in the tumor by one of several methods described. In
addition, a magnetic field is created that traverses the
tumor and that inhibits the recombination of the free
radicals in the tumor enhancing escape radical reactivity,
which results in enhanced tumoricidal effect.
The foregoing and other objectives, features and
advantages of the invention will be more readily understood
upon consideration of the following detailed description of
the invention, taken in conjunction with the accompanying
drawings.
BRIEF DESCRIPTION OF THE DRAWINGS
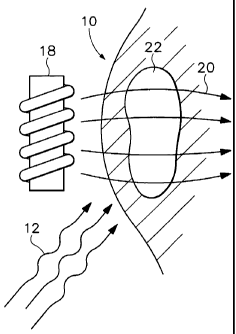
FIG. 1 is an illustration of a tumor being treated
by radiation therapy augmented by a magnetic field.
FIG. 2 is an illustration of a tumor being treated
by strips of implanted radioactive material interspersed
with strips of implanted magnetic material.
FIG. 3 is an illustration of a tumor into which a
magnet has been introduced by a laparoscope.
CA 02463491 2004-04-13
WO 03/030722 5 PCT/US02/32444
FIG. 4 is an illustration of a tumor that is being
treated by a combination. of radioactive beads and magnetic
beads.
BEST MODES OF CARRYING OUT THE INVENTION
Referring to FIG. l, in a first preferred
embodiment a patient's body 10 is subjected to radiation 12
that travels through the body 10 in a first direction. A
magnet 18 is oriented so as to create a magnetic field 20 in
the vicinity of a tumor 22 of one mini Tesla (mT). The
magnet 18 is aligned so that the area of intersection of the
radiation and the magnetic field conforms to the outline of
the tumor. Typically in this operation magnets 18 are
electromagnets, as they may be controlled to vary the
intensity of the magnetic field over time. In one preferred
embodiment, however, static magnets are used as they can be
more easily shaped to reflect the cross section of the
tumor. The magnetic field may also be induced by magnetite,
sprayed or painted magnetic films or implants or any other
method of creating a magnetic field. In addition, magnetic
shielding may be introduced to block ambient magnetic field
effects.
The radiation 12 used may be gamma ray, x-ray or
photon radiation. In addition, as used in this application,
the term radiation also encompasses sound waves as in
ultrasound-induced acoustic cavitation, and radiation 12 may
take this form.
Referring to FIG. 2, a tumor 22 may be treated by
placing radioactive strips 30 about it. Additionally,
magnetic strips 32 are placed to create a magnetic field in
the tumor 22.
Referring to FIG. 3, a laparoscope 40 is used to
introduce a magnet 42 directly into the tumor. The tumor is
then additionally treated with an injection of chemotherapy
CA 02463491 2004-04-13
WO 03/030722 6 PCT/US02/32444
agents, which could also be from the laparoscope 40 or with
radiation as in FIG.. 1. Laparoscope 40 may also include a
light source, for performing photon source radiation. In one
preferred embodiment, laparoscope 40 injects a chemical
agent that is activated by light waves to yield free
radicals and also includes a light source for activating the
chemical agent.
FIG. 4 shows a tumor that is being treated by a
set of radioactive beads 50 and a set of magnetic beads 52,
adapted to create a magnetic field.
For any of the above-described techniques, the
magnetic field created is preferably between about 0.1
mTesla and 10 mTesla (10 - 1,000 gauss).
In an additional preferred embodiment, substances
that form free radical pairs in the presence of radiation
(including among other forms, light radiation) are injected
into the tumor or into the vicinity of the tumor. In a
variant of this embodiment, the substance that is injected
forms free radical pairs that are particularly likely to
cause apoptosis (cell death). In another variant, the
substance that is injected forms free radical pairs that are
particularly sensitive to a magnetic field. The free radical
pairs produced may be easily induced into the triplet state
by way of the application of a low intensity magnetic field.
In tumor treatment systems where the effectors of
tumor cell killing are the escape free radicals, analysis
indicates that a magnetic field on the order of 0.1 to 1.0
milliTesla will cause a 30-40$ increase in tumor lethality.
Accordingly, in one preferred variant of a treatment system
using radiation mediated free radical production, the
equivalent tumoricidal effect is achieved at significantly
lower overall radiation levels. In another variant, higher
tumoricidal effect is achieved at equivalent radiation
levels.
CA 02463491 2004-04-13
WO 03/030722 ~ PCT/US02/32444
Furthermore, since the magnetic effect is a vector
quantity, the magnetic field, in one preferred embodiment,
is contoured to fit the topography of an irradiated tumor
. allowing more focused radiation effect and sparing normal
S tissues. This topologic modeling complements and improves
prior art radiation treatment field design by introducing an
independent vector specific variable.
As noted, at least one preferred embodiment makes
use of oscillating or alternating magnetic fields to
influence radical reactivity. However, the frequency of the
oscillating or alternating field will be dependent on the
short radical recombination time window. Subtle
combinatorial magnetic effects, which combine static and
modulated magnetic field effects, may offer advantages in
specific situations.
At least one preferred embodiment makes use of
modifications of the ambient magnetic field environment to
optimize the magnetic effect described. In cases where
electronic equipment near to the intended tumor target
modifies the electromagnetic environment, shielding is used
to prevent ambient electromagnetic interference. In one
preferred embodiment, low carbon steel shields are used for
this purpose. In another preferred embodiment mumetal
shielding is employed.
Radical pair recombination as mentioned will be
influenced by the biologic reaction environment and
modifications to viscosity, temperature and structural
properties including cell membranes and organelles may be
exploitable parameters to further enhance the tumoricidal
benefit described here.
In a further preferred embodiment, the magnetic
field and the substances injected are specifically adapted
to destroy specific cell constituents that are targeted by
known targeting mechanisms, e.g. antigen-antibody targeting.
CA 02463491 2004-04-13
WO 03/030722 8 PCT/US02/32444
INDUSTRIAL APPLICABILITY
The present invention finds application in the
fields of health care and medical equipment.
The terms and expressions which have been employed
in the foregoing specification are used as terms of
description and not of limitation, and there is no
intention, in the use of such terms and expressions, of
excluding equivalents of the features shown and described or
portions thereof, it being recognized that the scope of the
invention is defined and limited only by the claims which
follow.