Note: Descriptions are shown in the official language in which they were submitted.
CA 02493733 2005-01-26
WO 2004/015158 PCT/US2003/024541
1
PLASMA SPHEROIDIZED CERAMIC POWDER
FIELD OF.THE INVENTION
The present invention relates to ceramic powders, pardcularly zirconia
powders, and a
process for the production of ceramic powders in which the powders have a
highly uniform
composition.
BACKGROUND OF THE INVENTION
Stabilized zirconia powders are widely used to provide thermally stable and
abrasion
resistant coatings to parts that are exposed to very high temperatures during
use but which are
also exposed to ambient temperatures.. It does, however, have a well-known
drawback in that, as
it cycles between high and low temperatures, it undergoes a crystal phase
change from the
tetragonal crystal phase structure, which is stable at elevated temperatures,
to the monoclinic
crystal phase structure, which is stable at room temperature. Volume changes
occur as this
' crystal phase change takes place compromising the physical integrity of the
zirconia coating.
There is another phase of zirconia which is also stable at temperatures above
the
monoclinic/tetragonal transition temperature, (the "cubic" phase), but since
litfle or no volume
change occurs on the transition frorn cubic to tetragonal, this is treated for
the purposes of this
Description as a form of the tetragonal phase and is not dis6nguished
therefrom.
In order to resolve the integrity problems with zirconia coatings resulting
from the crystal
phase changes, it is common to use stabilized zirconia in powder coatings.
Stabilization can be
achieved by the addition of a number of additives that have the effect of
inhibiting the conversion
from the tetragonal crystal phase to the monoclinic crystal phase upon
cooling. Such additives
include stabilizing oxides such as calcia, magnesia, yttria, ceria, hafnia,
and rare earth metal
oxides.
Stabilized zirconia coatings are wideiy used to produce an abradable
protective coating
on surfaces or thermal barrier coatings. They are typically applied as sprays
by a flame spray or a
plasma spray approach.
In the production of stabilized zirconia powders, the most common technique is
described
in U.S. Patent No. 4,450,184 to Longo et al., in which an aqueous slurry-
comprising a blend of
zirconia and stabilizer materials is fed into a spray dryer to form dried
porous particles. The
porous particles are fused into hornogenous hollow structures with a plasma or
flame spray gun
CA 02493733 2005-01-26
WO 2004/015158 PCT/US2003/024541
2
which tneits and fuses the components such that the par6cles ejected therefrom
are stabilized
zirconia, Thermal spraying of the hollow spheres creates a porous
and.abradable coating.
However the Longo process does not achieve a high degree of uniformity
of'composition.
U.S. Patent No. 5,418,015 to Jackson et al. discloses a feed composition for
thermal
spray applications composed of stabilized zirconia mixed with zircon and a
selected oxide to form
an amorphous refractory oxide coating. Such products do not however have the
required level of
size and compositional uniformity that would be desirable to secure good
thermal barrier coating
compositions for high temperature applica6ons. This is at least in part
because there are many
opportunities for variability in the resultant coating as a result of
differing particle sizes in the feed,
the flame or plasma gun designlshape, feed rate pressures, and the like.
Another method of forming stabilized zirconia involves sintering wherein the
components
are blended together as powders, sintered, and upon cooling, the sintered mass
is broken up into
particles, These particles may then serve as feed for a flame spray device.
Unfortunately, this
process does not provide for a high level of chemical homogeneity in the
stabilization and results
in widely varying shapes and par6cle sizes in the feed.
Ceramic mixtures such as stabilized zirconia may also be mad'e by
electrofusion. The
fused mixtures are much more uniform than those made by the processes
discussed above
because they are the result of complete melting of the components. However,
the components
are difficult to melt and have poor flow characteris6cs as a result of their
high density and irregular
shape generated when the fused masses are crushed to provide particles. Thus,
the currently
available stabilized zirconia powders made by electrofusion have a high degree
of un-melted
material in the spray process resulting in poor efficiencies and coatings with
a high content of such
un-melted material particies. The un-melted particies introduce stresses into
the,coating due to
the varying density of the coating in and around the un-melted particles. As a
result, the
longevity of the resultant coating is diminished, particularly under stressful
conditions.
Notwithstanding the atate of the technology, it would be desirable t9 provide
a ceramic
powder having a high level of chemical and morphological uniformity, which in
turn, provides a
durable thermal spray coating.
CA 02493733 2005-01-26
WO 2004/015158 PCT/US2003/024541
3
SUMMARY OF THE INVENTION
In a first aspect, the present invention is directed to a zirconia powder
particularly adapted
for use as a thermal barrier coating which comprises morphologically and
chemically uniform
stabilized zirconia in the form of substantially spheroidal hollow spheres.
The zirconia is chemically uniform and by this meant that the zirconia is at
least 90% pure
and is at least about 96% by weight stabilized in a tetragonal crystal phase.
The zirconia is also
morphologically uniform and by this is mean that at least 95 volume % of the
zirconia is in the
form of spheres with a particle size of less than about 200 micrometers. The
spheres may be
somewhat deformed but are identifiably based on spheres rather than having
random shapes.
The spheres are preferably at least 7.5% hollow spheres. In a preferred
embodiment a chemically
uniform stabilized zirconia is heat treated by plasma fusion to obtain the
substantially spheroidal
shape. Preferably, the stabilized zirconia contains less th .an i.0% by weight
monoclinic zirconia.
In a preferred aspect, the present invention is directed to a thermally
sprayable
composition comprising hollow spheres of yttria stabilized zirconia, the
hollow spheres having a
parkicle size of less than about 200 micrometers, wherein the yttria is
uniformly incorporated into
the zirconia by electrofusion prior to formation of the hollow spheres.
Preferably, the zirconia
contains less than 2.0% by weight monoclinic zirconia. The hollow spheres are
preferably formed
by plasma fusion.
In yet another aspect, the present invention is directed to a process for
producing
spheroidized ceramic powder comprising the steps of: providing a chemically
uniform, stabilized
zirconia; and heat treating the zirconia to form substantially hollow
spheresthereof of
morphological uniformity. Preferably, the stabilized ceramic material
comprises zirconia
stabilized in a tetragonal crystai phase and contains less than about 2.0% by
weight monoclinic
zirconia. The stabilized zirconia is preferably formed by electrofusion of
zirconia and a stabilizing
oxide. Preferably, beat treating occurs in a.plasma spray gun or a flame spray
gun. The process
may further include the step of comminuting the stabilized ceramic materials
prior to'heat treating.
In still yet another aspect, the present invention is directed to a process of
forming a
thermal sprayable powder coafing comprising the steps of: providing a zirconia
feedstock whetein
the zirconia is at least 96% by weight stabilized in a tetragonal crystal
phase; and plasma fusing
the zirconia feedstock to form substantially hollow spheres thereof.
Preferably, the stabilized
zirconia is formed by electrofusion. -
CA 02493733 2005-01-26
WO 2004/015158 PCT/US2003/024541
4
The invention also comprises a process for the application of a thermal
barrier coating to
a substrate which comprises thermal spray coating the substrate using a
sprayable composition
comprising zirconia, of which at least 96% is stabiiized.in the tetragonal
form, having a
substantially uniform spherical morphology with particle sizes smaller than
200, and more
preferably smaller than 100, micrometers. In referring to particle sizes it is
understood that the
reference is to volume average particle sizes unless otherwise apparent from
the context.
BRIEF DESCRIPTION OF THE DRAWINGS
Figures 1 through 4 are elemental line scans of well sintered particies from
commercially
available stabilized zirconia powders.
Figure 5 is an elemental line scan of a hollow spheroidized zirconia particle
made in
accordance with the present invention.
DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENT
The present invention is directed to a thermal sprayable zirconia powder
having a
very uniform chemical composition and morphology. The thermal sprayable
ceramic
powder preferably has a spheroidized shape, and even more preferably, the
spheroidized
particles are substantially hollow so that the particle's melt more rapidly
forming either
dense coatings or coatings with uniform porosity depending on the spray
conditions. In a
most preferred embodiment, the thermal sprayable zirconia powder of the
present
invention comprises at least 90 volume % zirconia,. and the zirconia is at
least about 96%
by weight stabilized in the tetragonal form by a-stabilizing oxide. More
preferably, the
zircon,ia is at least 98% by weight stabilized in the tetragonal form, and
most preferably, at
least about 99% by weight stabilized in the tetragonal form.
The zirconia feedstock used in the present invention is stabilized with a
stabilizing
oxide such as, but not limited to, yttria, calcia, ceria, -hafnia, magnesia, a
rare earth metal
oxide, and combinations thereof. To achieve a high chemical uniformity in the
stabilized
zirconia feedstock, the stabilizing oxide is preferably electrofused. with the
zirconia. The
amount of stabilizing oxide used may va.ry depending on the result desired. A
sufficient
amount of the stabilizing oxide is an amount which substantially stabilizes
the zirconia in
the tetragonal crystal phase. The stabilizing oxide is desirably fully reacted
with and
incorporated into the zirconia crystal structure such that X-ray analysis
cannot detect a
CA 02493733 2007-08-02
significant amount, (no more than 4%), of the monoclinic zirconia. The amount
of the
stabilizing oxide present can be up to about 10% by weight but some
stabilizers are
effective at lower levels. For example, in the case of zirconia stabilized
using yttria, an
effective amount may be about 1% but can be as high as 20% by weight ; for
magnesia, about 2% to about 20% by weight is effective ; for calcia, about 3%
to about
5% by weight may be used; and for a rare earth metal oxide, about 1% to about
60%
by weight. A mixture of stabilizing oxides may be used.
The stabilizing oxide, preferably yttria, is arc fused with the zirconia at a
temperature range of about 2750 C to about 2950 C such that components are
completely molten and, since this is above the transition temperature, the
zirconia is
substantially completely in the tetragonal crystal phase. Upon cooling to room
temperature, the stabilizing oxide maintains this tetragonal state even below
the
normal transition temperature. To enhance this effect, the molten material is
preferably
rapidly cooled with water or air such that the melt flow is broken up into a
flow of
droplets and cooled to provide fine particles of stabilized. zirconia with a
very
homogenous chemical composition. A method of quenching the molten zirconia and
stabilizing oxide, where the rapid solidification tends to stabilize the
tetragonal form of
zirconia, is disclosed in U. S. Patent No. 5,651, 925. Preferably, the
resulting fine
particles of stabilized zirconia are further comminuted. Typically, the fine
particles are
milled to a size of less than about 5 microns, preferably less than about 2
microns,
more preferably about 0. 5 microns. The fine particles of stabilized zirconia
are
collected as agglomerated particles. Although the agglomeration step is not
essential
to the practice of the invention, it does provide a more useable size for
further heat
treatment of the stabilized zirconia as discussed below.
The agglomerated particles are further heat treated to form substantially
hollow spheres thereof having uniform morphology. A particularly preferred
form of
heat treatment is a plasma fusion process where the particles are melted
together in a
plasma flame and collected as a fine powder having a high level of chemical
and
morphological uniformity. Substantially hollow spheres of the stabilized
zirconia are
formed which preferably contain less than about 4% by weight, more preferably
less
than about 2% by weight, and more preferably less than about 1% by weight,
monoclinic zirconia. Preferably, the substantially hollow spheres have a
particle size of
less than about 200
CA 02493733 2005-01-26
WO 2004/015158 PCT/US2003/024541
6
microns, more preferably less than. about 100 microns, and most preferably,
less than
about 75 microns.
Unexpectedly, the substantially hollow spfieres of the stabilized zirconia
feedstock have a high level of chemical and morphological uniformity wherein
the zirconia
is at least about 96% by weight stabilized in the tetragonal crystal phase,
preferably at
least about 98% by weight stabilized in the tetragonal crystal phase, and more
preferably
at least about 99% by weight in the tetragonal crystal phase. Thus, thermal
sprayable
spheroidized powders of the present invention form more stable and durable
coatings due
to the high level of chemical uniformity due to the electrofusion of the
zirconia and
stabilizing oxide which substantially stabilizes the zirconia. The
spheroidized particles of
the stabilized zirconia melt more readily because of the hollow sphere
morphology and
complete reaction of the stabilizer with the zirconia. The coatings sprayed
have very
predictable density from high density to controlled porosity depending on the
spray
conditions.
To obtain durable zirconia thermal sprayable coatings, a uniform stabilization
of
the tetragonal crystal phase of the zirconia is crucial. It has now been shown
that in
comparison to commercially available zirconia powders stabilized with yttria,
the
spheroidized zirconia powder of the present invention shows substantial
incorporation of
the yttria into the zirconia. Table I illustrates an example of a zirconia
powder of the
present invention in comparison with commercially available stabilized
zirconia powders
with regard to volume percent of each crystal phase through X-ray Diffraction
data (XRD).
TABLEI
Ex. Tet * ZrCh Nbno.Znh YA (vnl.
WN W%) %)
PF 100 .0,0
PX 88.3 11.7 - -
ST 98.9 1.1 - -
M1 95.6 4.4 --
M2 89.4 10.6 - -
CA 02493733 2005-01-26
WO 2004/015158 PCT/US2003/024541
7
" Includes cubic zirconia as well as tetragonal
PF = zirconia powder of the present invention.
PX = PRAXAIR ZROn~ available from Praxair, Inc., DanbUry, Connecticut.
ST = STARK YZ available from H.C.Stark GmbH.
Ml = METCO 204NS-G available from Sulzer Metco, The Coatings Co., Westbury, W.
M2 = METCO 204 available from Sulzer Metco.
Although the concentration of yttria was not detected by X-ray diffraction
(XRD) in
all samples, it is the concentration of the monoclinic zirconia which
determines whether
the zirconia has been substantially stabilized in the tetragonal crystal
phase. Elemental
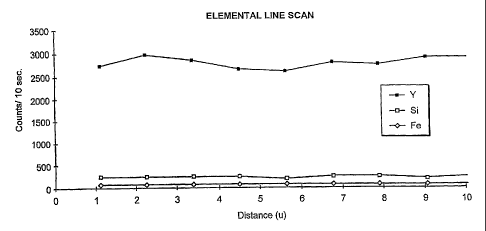
line scans of particles of Examples PX, ST, M1, and M2 are illustrated in
Figures 1
through 4 to determine the composition of the particles. In Figure 1, the
elemental line
scan, edge to edge, of a well-sintered particle of Example PX shows that the
particie
analyzed did not have a uniform composition given the non-linear line
representing
yttrium. Therefore, although XRD did not detect yttrium, the elemental line
scan shows
that the yttria did not completely co-fuse with the zirconia, and as such, the
composition is
not sufficienfly chemically uniform. The spike in the silicon line further
attests that the
particle is also not chemically or morphologically uniform. In Figure 2, the
elemental line
scan of a well-sintered par6cle of Example ST, edge to edge, also shows
variations in the
yttrium concentration, and thus, the particle is not chemically uniform. In
Figure 3, the
elemental line scan of a well-sintered par8cle of Example Ml, again shows
variation in the
yttrium concentration, and thus, the particle is not chemically uniform. In
Figure 4, the
elemental line scan of a well-sintered par6cle of Example M2 again show
variations in the
yttrium concentration, and thus, the particle is not chemically uniform.
By electrofusing the stabilizing oxide, yttria, with the zirconia, the
stabilized
zirconia is relatively uniform in composi6on. Further heat treatment such as
plasma
fusion provides the morphological uniformity of the substantially hollow
spheres. The
unexpected chemical and morphological uniformity is clearly illustrated in the
elemental
line scan shown in Figure 5 of a hollow sphere of Example PF. The
substantially linear
yttrium line illustrates that a complete melt and re-solidification had
occurred to provide a
chemically uniform sphere. Also, the substantially flat silicon and iron
element lines
illustrate the morphological uniformity of the sphere.
CA 02493733 2005-01-26
WO 2004/015158 PCT/US2003/024541
8
Therefore, although the commercially available stabilized zirconia powders
appear to be similar on their face, the spheroidized zirconia powder of the
present
inven6on provide a more chemically and morphologically uniform particle for
thermal
spray applications. The chemical and morphological uniformity in turn produces
thermal
spray coatings of exceptional durability.
Other variations and modifications of the basic invention can be conceived
without depariang from the concepts described above. It is intended that all
such
variations and modifications be included within the broad understanding of
this invention.