Note: Descriptions are shown in the official language in which they were submitted.
CA 02575865 2007-02-15
REMOVABLE EMBOLUS BLOOD CLOT FILTER
This application is a divisional of Canadian application Serial No. 2,261,120
filed internationally on July 16, 1997 and entered nationally on January 15,
1999.
Backuound of the Invention
In recent years, a number of medical devices have been designed
which are adapted for compression into a small size to facilitate
introduction into a vascular passageway and which are subsequently
expandable into contact with the walls of the passageway. These devices,
among others, include blood clot filters which expand and are held in
position by engagement, with the inner wall of a vein. It has been found to
be advantageous to form such devices of a shape memory material having
a first, relatively pliable low temperature condition and a second, relatively
rigid high-temperature condition. By forming such devices of temperature
responsive material, the device in a flexible and reduced stress state may be
compressed and fit within the bore of a delivery catheter when exposed to
a temperature below a predetermined transition temperature, but at
temperatures at or above the transition temperature, the device expands and
becomes relatively rigid.
Known self expanding medical devices have been formed of Nitinol,
an alloy of titanium and nickel which provides the device with a thermal
memory. The unique characteristic of this alloy is its thermally triggered
shape memory, which allows a device constructed of the alloy to be cooled
below a temperature level and thereby softened for loading into a catheter
in a relatively compressed and elongated state, and to regain the memorized
shape in an austenitic state when warmed to a selected temperature, above
the temperature transformation level, such as human body temperature. The
two interchangeable shapes are possible because of the two distinct
CA 02575865 2007-02-15
-2-
microcrystalline structures that are interchangeable with a small variation
in temperature. The temperature at which the device assumes its first
configuration may be varied within wide limits by changing the
composition of the alloy. Thus, while for human use the alloy may be
focused on a transition temperature range close to 98.6 F, the alloy readily
may be modified for use in animals with different body temperatures.
U.S. Patent No. 4,425,908 to Simon discloses a very effective blood
clot filter formed of thermal shape memory material. This filter, like most
previously developed vena cava filters, is a permanent filter which, when
once implanted, is designed to remain in place. Such filters include
structure to anchor the filter in place within the vena cava, such as elongate
diverging legs with hooked ends that penetrate the vessel wall and
positively prevent migration in either direction longitudinally of the vessel.
Within two to six weeks after a filter of this type has been implanted, the
endothelium layer grows over the diverging legs and positively locks the
hooks in place. Now any attempt to remove the filter results in a risk of
injury to or rupture of the vena cava.
A number of medical procedures subject the patient to a short term
risk of pulmonary embolism which can be alleviated by a filter implant.
In such cases, patients are often adverse to receiving a permanent implant,
for the risk of pulmonary embolism may disappear after a period of several
weeks or months. However, most existing filters are not easily or safely
removable after they have remained in place for more than two weeks, and
consequently longer term temporary filters which do not result in the
likelihood of injury to the vessel wall upon removal are not available.
In an attempt to provide a removable filter, two filter baskets have
been formed along a central hub which are conical in configuration, with
CA 02575865 2007-02-15
-3-
each basket being formed by spaced struts radiating outwardly from a
central hub. The central hubs are held apart by a compression unit, and the
arms of the two baskets overlap so that the baskets face one another.
Devices of this type require the use of two removal devices inserted at each
end of the filter to draw the baskets apart and fracture the compression unit.
The end sections of the arms are formed to lie in substantially parallel
relationship to the vessel wall and the tips are inclined inwardly to preclude
vessel wall penetration. If a device of this type is withdrawn before the
endothelium layer grows over the arms, vessel wall damage is minimized.
However, after growth of the endothelium layer the combined inward and
longitudinal movement of the filter sections as they are drawn apart can tear
this layer. U.S. Patent No. 5,370,657 to Irie is illustrative of a prior art
removable filter requiring two removal devices.
Summarv of the Invention
It is a primary object of the present invention to provide a vessel
implantable filter of shape memory material having temperature induced
austenitic and martensite states which may be easily removed by a single
removable device after an extended period of time without injuring the
vessel wall.
Another object of the present invention is to provide a blood clot
filter of Nitinol which operates in a temperature induced austenitic state to
exert a force on the walls of a vessel by means of oppositely disposed legs
to maintain the filter in place, but which may easily be removed after the
endothelium layer has covered the ends of the filter legs.
CA 02575865 2007-02-15
-4-
A further object of the present invention is to provide a novel and
improved filter of shape memory material having a first group of arms and
second group of legs which initially incline in the same direction from a
central axis. The ends of the arms in the first group of arms are oriented
to engage a vessel wall to prevent longitudinal movement of the filter along
the vessel in a first direction, and the ends of the legs of the second group
of legs are oppositely oriented to engage the vessel wall to prevent
longitudinal movement of the filter along the vessel in a second opposite
direction. The ends of both groups of arms and legs are configured to
permit withdrawal from the endothelium layer without risk of injury to the
vessel wall.
A still further object of the present invention is to provide a novel
and improved filter of shape memory material designed to facilitate removal
of the filter from a vessel by a two stage removal process accomplished at
one end of the filter.
According to the invention, a resilient, longitudinally extended blood
clot filter is inwardly radially collapsible toward its longitudinal axis into
a collapsed configuration for insertion into a vein, but is adapted for
automatic radial expansion into contact with the inner wall of the vein at
two longitudinally spaced peripheral locations therein. The filter has
leading and trailing ends and comprises a plurality of wires. The wires, in
the normal expanded configuration of the filter, are in the form of a
plurality of elongated arms and legs with openings between the wires
providing filter baskets at the leading and trailing ends of the filter. The
wires have peripheral portions for contact with the inner wall of the vein
at two longitudinally spaced peripheral locations.
CA 02575865 2007-02-15
-5-
To provide a filter that is inwardly radially collapsible from its
normally expanded configuration toward its longitudinal axis into a
collapsed configuration for insertion into a vein, the blood clot filter is
preferably formed from a plurality of wire portions composed of a shape
memory material having a first, low-temperature condition and a second,
high-temperature condition. The material in its low-temperature condition
is relatively pliable (so that the wire portions may be straightened) and in
its high-temperature condition is resiliently deformable and relatively rigid,
and takes a pre-determined functional =form.
In the high-temperature condition of the material, the filter comprises
coaxial first and second filter baskets, each filter basket being generally
symmetrical about the longitudinal axis of the filter with one filter basket
being concave and one being at least partially convex relative to the filter
leading end.
The invention provides according to an aspect, for a blood clot filter
having a central longitudinal axis which is collapsible into a collapsed
configuration toward the longitudinal axis for insertion into a blood vessel
and which is radially expansible outwardly from the longitudinal axis to an
expanded configuration for contact with an inner wall of the blood vessel
at two longitudinally spaced locations, the blood clot filter having leading
and trailing ends and comprising: a first plurality of spaced wires which, in
the expanded configuration of the filter curve outwardly away from the
longitudinal axis toward the trailing end of the blood clot filter to form a
first filter basket and a second plurality of spaced wires which, in the
expanded configuration of the filter angle outwardly away from the
longitudinal axis toward the leading end of the blood clot filter to form a
second filter basket opening toward the leading end.
According to another aspect, the invention provides for a method
for removing a blood clot filter having a leading end and a trailing end
CA 02575865 2007-02-15
- 5a -
from engagement with the walls of a vessel, the blood clot filter having a
longitudinal axis, a first plurality of elongate arms curving outwardly away
from the longitudinal axis toward the trailing end to form a first filter
basket, and a second plurality of elongate legs angling outwardly away
from the longitudinal axis toward the leading end to form a second filter
basket, the method including the steps of: holding the filter against
longitudinal movement while moving the arms thereof toward the leading
end of the filter to disengage the arms from engagement with the vessel
walls, and subsequently moving the filter longitudinally in the direction of
the trailing end thereof to withdraw the elongate legs from engagement
with the vessel wall.
According to yet another aspect, the invention provides for a
commercial package which comprises a blood clot filter according to the
invention. The package also comprises written instructions supplied either
together with the filter or separately, the instructions describing removal
of the blood clot filter by holding the filter against longitudinal movement
while moving the arms thereof toward the leading end of the filter to
disengage the arms from engagement with the vessel walls, and
subsequently moving the filter longitudinally in the direction of the trailing
end thereof to withdraw the elongate legs from engagement with the
vessel wall.
Accordingly to a further aspect, the invention provides for a kit
which comprises the blood clot filter accordingly to the invention. The kit
also comprises a and a filter removal device comprising a housing for
gripping by a user, the housing having a front end and an open-ended
chamber opening towards the front end, a retractable snare having
proximate and distal ends, the proximate end retractable into the
chamber, actuation means for retraction of the snare, a snare device at
the distal end for engagement with the axis of the filter, removal tube
encircling the snare and having a proximal end at the proximal end of the
CA 02575865 2007-02-15
- 5b -
snare, the removal tube displaceable along the snare, and separate and
independent actuation means for extending the snare to engage the filter
and for displacing the removal tube for moving the removal tube over the
filter into contact with the arms for disengaging the arms from a vessel
wall while avoiding contact with the legs, the removal tube comprising a
hollow tube having an inside diameter sufficient to receive the filter in a
collapsed position.
Brief Description of the Drawinjzs
Figure 1 is a side view of an expanded blood clot filter of the
present invention;
Figure 2 is a diagrammatic view of the filter of Figure 1 in place
within a vessel;
Figure 3 is a diagrammatic plan view of the filter of Figure I in
place within a vessel;
CA 02575865 2007-02-15
-6-
Figure 4 is a side view of a second embodiment of the blood clot
filter of the present invention;
Figure 5 is a side view of a third embodiment of the blood clot filter
of the present invention;
Figure 6 is a diagrammatic view of the filter of Figure 5 in place
within a vessel; and
Figure 7 is a partially sectional cutaway view of a removal tool for
the blood clot filter of the present invention.
Detailed Description
By forming the body of a blood clot filter of a Nitinol alloy material,
such as Nitinol wire, transition between the martensitic and austenitic states
of the material can be achieved by temperature transitions above and below
a transition temperature or transition temperature range which is at or below
body temperature. Such controlled temperature transitions have
conventionally been employed to soften and contract the Nitinol filter body
to facilitate insertion into a catheter and to subsequently expand and
rigidify
the body within a vascular or other passageway. Although the filters of the
present invention are preferably formed from a temperature responsive
shape memory material, such as Nitinol, they can also be formed of a
compressible spring metal such as stainless steel or a suitable plastic.
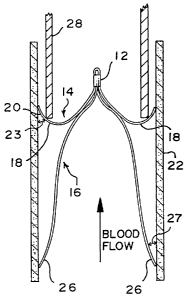
Referring now to Figures 1 and 2, an expanded blood clot filter 10
is illustrated which is made from sets of elongate metal wires. The wires
CA 02575865 2007-02-15
-7-
are held together at one end at a hub 12 where they are spot welded
together or otherwise joined. In the low temperature martensite phase of
wires made of shape memory material, the sets of wires can be straightened
and held in a straight form that can pass through a length of fine plastic
tubing with an internal diameter of approximately 2 mm (#8 French
catheter). In its high temperature austenitic form, the filter 10 recovers a
preformed filtering shape as illustrated by Figure 1. Similarly, wires of
spring metal can be straightened and compressed within a catheter or tube
and will diverge into the filter shape of Figure 1 when the tube is removed.
In its normal expanded configuration or preformed filtering shape,
filter 10 is a double filter, having a first filter basket section 14 and a
second oppositely disposed filter basket section 16. The two filter sections
provide peripheral portions which engage the inner wall of the vein at two
longitudinally spaced locations and, the two basket filter sections are
generally symmetrical about a longitudinal axis passing through the hub 12.
The first filter basket section 14 is formed from short, arcuate
lengths of wire 18 which form arms that curve downwardly, outwardly and
upwardly from the hub 12 toward the trailing end of the filter 10. The tip
sections 20- of the wires 18 are substantially straight lengths with ends
which lie on a circle at their maximum divergence and the tip sections
engage the wall 22 of a vessel at a slight angle 23 (preferably within a
range of from ten to forty-five degrees) to anchor the filter 10 against
upward movement in Figure 2. Normally, there are six wires 18 of equal
length extending radially outward form the hub 12 and circumferentially
spaced, such as for example by sixty degrees of arc as shown in Figure 3.
The second filter basket section 16 is oppositely oriented relative to
the first filter basket section 14, and normally includes six
circumferentially
CA 02575865 2007-02-15
-8-
spaced wires 24 forming downwardly extending legs which tilt and bow
outwardly of the longitudinal axis of the filter 10 from the hub 12. The
wires 24 are of equal length and are preferably much longer than the wires
18, and have tip sections 26 which are substantially straight lengths with
ends which lie on a circle at their maximum divergence. As shown in
Figure 2, the tip sections 26 engage the vessel wall 22 at a slight angle 27,
preferably within a range of from ten to forty-five degrees, to anchor the
filter against downward movement in Figure 2. As will be noted in Figure
3, the wires 24 are radially offset relative to the wires 18 and may be
positioned halfway between the wires 18 and also may be circumferentially
spaced by sixty degrees of arc. Thus the combined filter basket sections 14
and 16 can provide a wire positioned at every thirty degrees of arc at the
maximum divergence of the filter sections. With reference to the direction
of blood flow in Figure 2, the filter section 14 forms a convex filter basket
opening toward the trailing end of the filter 10 while the filter section 16
forms a concave filter basket opening toward the leading end of the filter
10.
After the filter 10 has remained in place within a vessel for a period
of time in excess of two weeks, the endothelium layer will grow over the
tip sections 20 and 26. However, since these tip sections are substantially
straight sections of wire oriented at a small angle to the vessel wall 22, the
filter can be removed leaving only twelve pin point lesions in the surface
of the endothelium. To accomplish this, a catheter or similar tubular unit
28 is inserted over the hub 12 and into engagement with the arms 18.
While the hub 12 is held stationary, the catheter is moved downwardly in
Figure 2 forcing the arms 18 downwardly and thereby withdrawing the tip
sections 20 from the endothelium layer. Then the hub 12 is drawn into the
CA 02575865 2007-02-15
-9-
catheter to withdraw the tip sections 26 from the endothelium layer and
collapse the entire filter 10 within the catheter. When the filter is formed
from shape memory material, cooling fluid can be passed through the
catheter to aid in collapsing the filter. It is important to first disengage
the
wires 18 by downward movement of the catheter but not to engage the
wires 24, for the catheter would force the legs 24 inwardly causing the tips
26 to move inwardly tearing the vessel wall. Once the arm tips 20 are
disengaged, the catheter is held stationary and the filter is pulled upwardly
into the catheter.
Figure 4 illustrates a second embodiment of the blood clot filter of
the present invention wherein elements having the same structural
configuration and operation as those shown in Figures 1-3 are designated
with the same reference numerals.
In the filter of Figure 4, a flexible mesh 30 is engaged with the wires
24 of the second filter basket section 16 to expand and contract therewith.
The mesh strands may be secured to the wires 24 or the mesh, which forms
a pocket 32 with an open end 34, may surround the wires 24 and be
attached to the hub 12. The mesh 30 will permit blood to pass through the
section 16 but will capture bone chips and small particles which might
otherwise pass between the wires 24 and 18. As the filter is withdrawn
collapsing the section 16, the mesh is collapsed around particles contained
therein which are then withdrawn with the filter.
With reference to Figures 5 and 6, a third embodiment of the blood
clot filter is indicated generally at 36. Here, the wire arms 38 of a first
filter basket 40 extend outwardly from a first juncture 42 on a central shaft
44 and the straight wire legs 46 of a second filter basket 47 extend
outwardly from a second juncture 48 on the central shaft spaced below the
CA 02575865 2007-02-15
- 10 -
first juncture. It should be noted that initially the shorter arms 38 extend
from the first juncture 42 in an angular direction relative to the central
shaft
44 which is the same angular direction that the longer legs 46 extend from
the second juncture 48. In fact, the arms and legs may extend from the
first and second junctures at the same angle; i.e. an angle of up to 70
degrees. These shorter arms have extended straight sections 50 which
extend angularly out from the juncture 42 to form the first filter basket
which throughout substantially its extent is coextensive but spaced from the
second filter basket 47 formed by the legs 46. At the ends of the straight
sections are elbows 52 which join a vessel wall engaging section 54. The
wall engaging sections 54 are substantially straight lengths of wire which
engage the vessel wall 22 at an angle 56 within a range of from ten to
ninety degrees.
To remove the blood clot filter 36, a removal wire or rod is engaged
with a hook 58 at the end of the central shaft 44 to hold the filter in place
while a catheter or removal tube is moved into engagement with the arms
38 over the hook. The catheter is moved downwardly until the wall
engaging sections 54 are withdrawn from the endothelium layer and the
vessel wall- 22. The angle of the wall engaging sections combined with the
angle of the straight sections 50 causes the wall engaging sections to move
downwardly and outwardly from the endothelium layer without tearing the
vessel wall.
It is important that the catheter or removal tube not contact the legs
46, as inward movement of these legs, when encased by the endothelium
layer would be likely to damage the vessel wall. Once the arms 38 are
disengaged from the vessel wall, the hook 58 is drawn into the catheter or
CA 02575865 2007-02-15
- 11 -
removal tube to draw the legs 46 upwardly out of the endothelium layer
and into the catheter or removal tube.
The blood clot filter 36, like the blood clot filter 10, may be formed
of spring metal or plastic, but is preferably formed of shape memory
material as previously described. In the case of both filters, it is possible
to have the first filter basket expand at a different transition temperature
than that of the second filter basket by annealing the wires of the respective
filter baskets differently to set different transition temperatures. This may
also be achieved by making the wires of one filter basket thicker or greater
in cross section than those of the remaining filter basket, at least in some
section along the length of the wires.
Figure 7 illustrates a removal device 60 for removing the blood clot
filters 10 and 36 of the present invention. This removal device includes a
housing 62 from which a handle 64 projects. An open ended chamber 66
is formed in the front portion of the housing 62 and a trigger 68 pivoted to
the housing at 70 projects into the chamber 66. Extending through the
chamber is a tubular cover 72 for a snare wire 74 and this cover is secured
at 76 to the housing 62 so that the cover is restrained against longitudinal
movement. - One end of the snare wire is connected to a snare lock 78
which may be variably positioned in a slot 80 in the housing, and the
opposite end of the snare wire projects from the tubular cover 72 and
includes a hook or other snare device 82 for engagement with the hook 58
or the hub 12 of a filter. A filter removal tube or sheath 84 is inserted over
the cover 72 and into the chamber 66 until it contacts the end of the trigger
68.
To remove a filter, the removal tube is inserted into close proximity
with the hub 12 or hook 58, and the snare wire is moved longitudinally
CA 02575865 2007-02-15
- 12 -
until the snare device 82 engages the hub or hook. With the snare device
engaged, the snare lock 78 is tightened to lock the snare wire against
longitudinal movement. Now the trigger 68 is pivoted to the dotted line
position in Figure 7 to move the removal tube longitudinally out of the
chamber 66 while the snare wire holds the filter against movement. The
trigger moves to the extended dotted line position through a slot 86 in the
housing, and the trigger is designed so that the distance travelled between
the retracted solid line position of the trigger and the extended position
moves the extraction tube over the filter for a distance sufficient to
disengage the filter arms but not sufficient to bring the extraction tube into
contact with the filter legs. The trigger mechanism can be designed to vary
the distance that the extraction tube travels to accommodate different filter
sizes, such as example by altering the position of the pivot point 70.
Once the arms of the filter have been disengaged, the extraction tube
is manually grasped to hold it stationary while the handle 64 is moved to
the left in Figure 7 to disengage the filter legs and draw the filter into the
extraction tube. Thus the basic method for removal of the filter is to first
grasp the trailing end and hold the filter against longitudinal movement.
Then, while the filter is held stationary, an extraction tube is moved over
the trailing end and into contact with the arms of the filter. The extraction
tube is moved longitudinally toward the leading end of the filter for a
distance sufficient to disengage the filter arms from the vessel wall while
preventing engagement of the extraction tube with the filter legs. Finally,
the filter is moved longitudinally toward the trailing end to disengage the
filter legs from the vessel wall. The housing 62 may be provided with an
infusion opening so that cooling fluid can be delivered through either the
CA 02575865 2007-02-15
- 13 -
extraction tube 84 or the tubular cover 72 to aid in the extraction of the
filter.
The filter may be positioned initially within a vessel by known
delivery devices such as the one shown in U.S. Patent No. 4,425,908 to
Morris Simon.