Note: Descriptions are shown in the official language in which they were submitted.
CA 02578803 2007-03-05
FLUID DISPENSER HAVING A HOUSING AND FLEXIBLE INNER BLADDER
15
This application is a division of Canadian Patent Application No.
2,426,182 filed October 23, 2001.
Field of the Invention
The present invention relates generally to dispensers for dispensing
fluids or other substances and, more particularly, to a dispenser having a
rigid vial, a
flexible bladder disposed within the rigid vial and defining 'a chamber
between the
flexible bladder and rigid vial for receiving therein a fluid or other
substances, and a
nozzle and pump assembly coupled in fluid communication with chamber for
dispensing fluids or other substances therefrom.
-1-
CA 02578803 2007-03-05
Background Information
Typical fluid dispensers include a container defining therein a chamber for
receiving a fluid to be dispensed, a nozzle and pump assembly mounted on the
container, and
a dip tube extending downwardly from the nozzle into the chamber for pumping
the fluid
from the bottom of the chamber, through the dip tube, and out of the
dispenser. Other known
dispensers include a vial and a flexible bladder received within the vial. For
example, U.S.
Patent No. 6,062,430 to Fuchs shows in FIG. 1 a dispensing container with
variable volume
compensation including a bottle-shaped vessel 2 in the form of a thin-walled,
hollow body
made from soft elastic plastic, and a reception container 15 formed of a
wrinkle film
l0 encapsulated within the vessel body 2.
One of the drawbacks associated with typical prior art fluid dispensers is
that
the fluid chamber(s) are not maintained in a substantially airless condition
throughout the
storage, shelf life and/or usage of the dispenser. For example, the nozzles
and/or valves used
in typical prior art dispensers frequently are incapable of maintaining the
dispenser in a
hermetically sealed condition. Such nozzles and/or valves allow the passage of
air or other
gases therethrough and into contact with the medicament or other substance
contained within
the fluid chamber(s). In addition, such nozzles and/or valves frequently allow
vapor loss
therethrough either during the storage, shelf life or usage of the dispensers.
Another drawback associated with prior art dispensers is that the materials of
'.0 construction may undergo creep that, in turn, causes seals foamed within
the dispensers to
leak. Many medicaments are maintained in storage and/or on store shelves for
at least
several, and in some instances, many months. During transportation and
storage, the
dispensers can be subjected to varying atmospheric conditions involving large
variations in
atmospheric temperature, pressure and/or humidity. As a result, the dispensers
are frequently
-2-
CA 02578803 2007-03-05
subjected to substantial differential thermal expansion and/or contraction
that, in turn, cause
the materials of construction to undergo creep. The seals and other components
of such prior
art dispensers typically are not designed to address such creep, and as a
result, the dispensers
develop leaks or otherwise allow air ingress and/or vapor loss when subjected
to such long
periods of storage or varying atmospheric conditions. For example, some
polyethylene
dispensers have been known to lose between about 10% to 25% of the weight of
their
contents during storage. Such weight loss is believed to be due to vapor loss
from the
medicament or other fluid-containing chambers through the polyethylene walls
of the
dispensers and/or through leaks otherwise created in the seals or other
structural interfaces of
the containers. The vapor loss is typically offset by air ingress into the
chambers. Vapor loss
and/or air ingress is particularly problematic for dispensers containing
medicaments, such as
pharmaceutical preparations or vaccines, because they tend to dilute each
predetermined
dosage of the medicament dispensed from the container, and/or cause the
dispenser to
dispense inconsistent concentrations of medicament from one dose to the next.
Yet another disadvantage associated with prior art dispensers is that because
they cannot reliably maintain the medicament or other substance contained
therein in an
airtight condition, they cannot be used for either multiple dose applications
or preservative-
free formulations. The use of single dose dispensers can be substantially more
expensive
than multiple dose dispensers. In addition, the preservatives used in many
medicaments, such
as pharmaceutical preparations and vaccines, can cause adverse reactions in
patients and/or
dilute the effect of the medicament on the patient.
Another drawback of prior art dispensers is that the ullage or "dead space"
inherent in such dispensers allows sediment build-up. Many medicaments and
other
formulations contained within such dispensers are suspensions. The ullage or
dead space in
1
CA 02578803 2007-03-05
the prior art dispensers allows the solutes or other solid components of such
suspensions to
form sediment therein. Such settling of the suspensions dilutes the
medicaments or other
substances contained within the dispensers and, in turn, alters the medicament
and/or the
concentration of medicament in each patient dose.
Another drawback associated with many prior art dispensers is that they can
only dispense the medicament or other substance contained therein in an
upright or other
single orientation. This drawback prevents such dispensers from being used
effectively in
other orientations, such as upside down. In addition, because such dispensers
do not maintain
the medicament or other substance contained therein in an airless condition,
they cannot be
used in low gravity envirorunents, such as outer space.
Accordingly, it is an object of the present invention to overcome one or more
of the above-described drawbacks and disadvantages of the prior art.
Summary of the Invention
The present invention is directed to a dispenser for dispensing a fluid. The
dispenser comprises a rigid housing, and a flexible bladder mounted within the
housing and
defining an interior chamber within the flexible bladder, and a fluid-
receiving chamber
between the flexible bladder and the rigid housing. The dispenser further
comprises means
for creating a first pressure within the fluid-receiving chamber greater than
a second pressure
within the interior chamber of the bladder to thereby prevent the ingress of
gases or vapors
into the fluid-receiving chamber. In a currently preferred embodiment of the
present
invention, the means for creating the pressure differential is formed by a
resilient material of
the bladder that flexes the bladder outwardly toward an expanded condition,
and thereby
creates the first pressure within the fluid-receiving chamber greater than the
second pressure
in the interior chamber of the bladder. Preferably, the resilient bladder is
molded in the
-4-
CA 02578803 2007-03-05
expanded condition, and therefore the resilient bladder will inherently tend
to force itself into
the expanded condition and thereby create the desired pressure differential
between the fluid-
receiving chamber and the interior chamber of the bladder.
A currently preferred embodiment of the dispenser further comprises a pump
coupled in fluid communication with the fluid-receiving chamber for pumping a
fluid
received therein from the dispenser; and a one-way valve coupled in fluid
communication
with the pump for allowing the passage of the pumped fluid therethrough and
preventing the
passage of fluids in the opposite direction. The one-way valve is preferably
formed by a
nozzle, and a flexible cover overlying the nozzle and creating the one-way
valve at the
interface of the nozzle and cover.
The preferred dispenser of the present invention further comprises a seal
formed between the flexible bladder and the rigid vial for sealing the fluid-
receiving
chamber. The seal includes a first protuberance extending radially outwardly
on an outer
surface of the flexible bladder, and a second protuberance axially spaced
relative to the first
protuberance and extending radially inwardly on an inner surface of the
bladder. The first
and second protuberances are subject to radial compression to seal the
interface between the
flexible bladder and rigid vial. Preferably, the first protuberance extends
about an outer
peripheral surface of the bladder and defines an outer annular sealing
surface, and the second
protuberance extends about an inner peripheral surface of the bladder and
defines an inner
annular sealing surface. In addition, the first protuberance defines a tapered
surface for
directing bladder material in approximately the direction of the second
protuberance to
thereby facilitate maintaining a fluid-tight seal in the event of bladder
material creep. The
seal preferably further includes a peripheral flange extending about an open
end of the
-5-
CA 02578803 2007-03-05
flexible bladder and subject to axial compression for further sealing the
interface between the
bladder and rigid vial.
The dispenser of the present invention further comprises a plug receivable
within an open end of the rigid vial and engageable with at least one of the
first and second
protuberances for radially compressing the protuberances to seal the interface
between the
flexible bladder and rigid vial. The plug defines at least one aperture
therethrough in fluid
communication with the interior chamber of the flexible bladder. Preferably, a
two-way
valve of the dispenser is coupled in fluid communication between the interior
chamber of the
flexible bladder and the aperture of the plug for preventing fluid
communication between the
interior chamber of the bladder and the ambient atmosphere when a pressure
differential
across the two-way valve is less than a threshold level. The two-way valve
preferably is
formed by a flexible, annular protuberance extending radially inwardly from an
inner
peripheral surface of the flexible bladder, and engageable with an annular
surface of the plug
to thereby seal the interface between the flexible bladder and plug. In a
currently preferred
embodiment of the present invention, the annular protuberance defines axially-
opposed
surfaces that taper inwardly in the radial direction to facilitate flexing of
the protuberance in
response to the pressure differential across the protuberance exceeding the
predetermined
threshold level.
The flexible bladder of the dispenser further defines an open end and a closed
end, and is movable between a collapsed condition and an expanded condition.
Upon
expansion of the flexible bladder from the collapsed condition into the
expanded condition,
the flexible bladder and rigid vial define an annular gap therebetween. In a
currently
preferred embodiment, the annular gap defines an increasing width in the axial
direction from
the open end toward the closed end of the flexible bladder, to facilitate
removal of fluid from
-6-
CA 02578803 2007-03-05
the fluid-receiving chamber upon expansion of the bladder. Preferably, the
flexible bladder
initially contacts the rigid vial adjacent to or near the open end of the
bladder, and then
progressively engages the rigid vial in the axial direction from the open end
toward the closed
end of the flexible bladder with further expansion thereof Also in accordance
with a
preferred embodiment of the dispenser, the flexible bladder defines an
external morphology
in an expanded condition, the rigid vial defines an internal morphology, and
the external and
internal morphologies are substantially the same to thereby allow the flexible
bladder to
conformably contact the rigid vial and substantially eliminate any dead space
in the fluid-
receiving chamber therebetween.
In accordance with a preferred embodiment of the present invention, the pump
of the dispenser comprises a piston, and a slide for slidably receiving the
piston therein. At
least one of the piston and the slide is reciprocable relative to the other.
In addition, the
piston is made of a relatively hard material, the slide is made of a
relatively soft material, and
the piston causes a compression zone of the slide to flex outwardly upon
moving at least one
of the piston and the slide relative to the other to thereby effect a fluid-
tight seal between the
piston and slide. In addition, forming the slide from a relatively flexible
material allows the
slide to be formed integral with a nozzle, such as by molding the two
components in a single
part, wherein the integral nozzle and slide may be released from a core pin by
injecting
pressured air therebetween.
The dispenser of the present invention preferably further comprises means for
controlling the flexible bladder to collapse into a predetermined collapsed
condition. In one
embodiment of the present invention, the means for controlling includes a
plurality of legs
extending axially inwardly into the interior chamber of the flexible bladder
for conformably
contacting the flexible bladder upon collapse thereof. In another embodiment
of the present
-7-
CA 02578803 2007-03-05
invention, the means for controlling is defined by at least one axially
elongated surface
discontinuity formed in the flexible bladder.
In accordance with another embodiment of the present invention, the flexible
bladder is capable of being penetrated by a needle or like injection member
for introducing a
predetermined substance into the fluid-receiving chamber. In this embodiment,
the flexible
bladder includes a first portion substantially infusible in response to the
application of
thermal energy thereto and compatible with the substance to be received within
the fluid-
receiving chamber, and a second portion overlying the first portion and
fusible in response to
the application of thermal energy thereto. Thus, the second portion enables
the formation of
a substantially fluid-tight seal between the flexible bladder and fluid-
receiving chamber in a
region thereof penetrated by the needle or like injection member. In one
embodiment of the
present invention, the second portion is formed of either a thermoplastic or
an elastomeric
material, and the bladder, including the first portion thereof, is formed of
vulcanized rubber.
One advantage of the dispenser of the present invention is that the pressure
differential between the fluid-receiving chamber and the internal chamber of
the bladder and
ambient atmosphere substantially prevents the ingress of air or other gases or
vapors through
the flexible bladder, or otherwise into the fluid-receiving chamber. As a
result, the dispensers
of the present invention may maintain the medicaments or other substances
contained therein
in an airless condition throughout substantial periods of storage, shelf life
and/or use.
Accordingly, the dispensers of the present invention are particularly well
suited for
dispensing multiple doses of non-preserved medicaments or other substances
requiring
storage in an airless condition.
Another advantage of the dispensers of the present invention is that the seal
formed between the flexible bladder and the rigid vial radially and axially
directs the material
-s-
CA 02578803 2007-03-05
of the flexible bladder to persistently maintain a fluid-tight seal regardless
of any creep of the
material during the storage or shelf-life of the dispenser. In addition, the
one-way valve
employed in the preferred embodiments of the present invention further
maintains the fluid-
receiving chamber in a hennetically-sealed condition throughout the storage,
shelf-life and/or
use of the dispenser.
Yet another advantage of the dispensers of the present invention is that
because the medicament or other substance is maintained in an airless
condition in the fluid-
receiving chamber, the dispensers may be used in virtually any orientation,
and furthermore,
may be used in low gravity environments.
Another advantage of the dispensers of the present invention is that the
flexible bladder preferably defines an external morphology substantially
matching the
internal morphology of the rigid vial. As a result, the flexible bladder may
expand and
conformably contact the rigid vial throughout the interface between these two
parts and, in
turn, eliminate any dead space within the fluid-receiving chamber.
Yet another advantage of the dispensers of the present invention is that the
two-way valve coupled in fluid communication between the interior chamber of
the flexible
bladder and the ambient atmosphere prevents any exchange of gases or vapors
between the
interior chamber of the bladder and ambient atmosphere, provided the pressure
differential
across the valve is less than a predetermined level. As a result, the two-way
valve creates a
relatively stable micro-atmosphere within the interior chamber of the flexible
bladder, thus
insulating the interior chamber from fluctuations in pressure and/or humidity
in the ambient
atmosphere and thereby further preventing the ingress of gas or vapors into
the fluid-
receiving chamber.
-9-
CA 02578803 2007-03-05
Other objects and advantages of the present invention will become apparent in
view of the following detailed description and accompanying drawings.
Brief Description of the Drawings
FIG. 1 is a partially broken-away, perspective view of an ocular treatment
apparatus having a dispenser mounted therein in accordance with a preferred
embodiment of
the present invention.
FIG. IA is a view similar to FIG. 1 absent the dispenser.
FIG. 1 B is a perspective view of the ocular treatment apparatus of FIG. 1.
FIG. 1 C is an exploded view, in perspective, of the ocular treatment
apparatus
of FIG. 113.
FIG. I D is an exploded view, in side elevation, of the ocular treatment
apparatus of FIG. 1 B.
FIG. 2 is a side elevational view, partly in section, illustrating in further
detail
the dispenser of FIG. 1 including the pump assembly, vial and a bladder and
wherein the
pump assembly is disposed in a closed position.
FIG. 3 is a view similar to that of FIG. 2, although the dispenser is rotated
90
with respect to its orientation in FIG. 2, and the pump assembly is disposed
in an extended
position.
FIG. 4 is a central cross-sectional view taken along a longitudinal axis of a
piston of the pump assembly of FIG. 1.
FIG. 5 is a front elevational view of a tip of a nozzle of the pump assembly
of
FIG. 1.
FIG. 6 is a longitudinal cross-sectional view taken along line 6-6 of FIG. S.
-10-
CA 02578803 2007-03-05
FIG. 7 is a central cross-sectional view taken along a longitudinal axis of a
slide or body of the pump assembly of FIGS. 2 and 3 and forming essentially
the
compression zone.
FIG. 8 is a central cross-sectional view taken along a longitudinal axis of a
flexible pump cover of the pump assembly of FIGS. 2 and 3, and illustrating
the manner in
which the pump cover extends from the tip of the nozzle to the rigid vial and
is configured to
allow reciprocal movement of the piston connected to the vial.
FIG. 9 is a central, cross-sectional view taken along a longitudinal axis of
the
rigid vial of the dispenser of FIGS. 2 and 3.
FIG. 9A is an enlarged view of a portion of the vial of FIG. 9 showing a rear
mounting portion for receiving the bladder of FIG. 10.
FIG. 10 is a central, cross-sectional view taken along a longitudinal axis of
the
bladder of the dispenser of FIGS. 2 and 3 showing in this configuration a
three-ribbed
structure provided to allow the bladder to collapse into a predetermined
collapsed condition.
FIG. 1 OA is an enlarged view of a portion of the bladder of FIG. 10.
FIG. l OB is a highly enlarged view of a portion of the bladder of FIG. 10.
FIG. 11 is a cross-sectional view taken along a transverse axis of the bladder
of FIG. 10.
FIG. 12 is a schematical view showing, in cross section, another embodiment
of the bladder of the dispenser of FIGS. 2 and 3 disposed within the rigid
vial and including
elongated discontinuities or elongation buffers disposed in an outer wall of
the bladder to
facilitate the collapse of the arcs that pass through the chords of the
respective arcs.
-11-
CA 02578803 2007-03-05
FIG. 13 is a top plan view of the rear plug employed to close the rear end of
the inner bladder by forming a sandwich-type structure between the rigid vial
and rear plug to
hermetically seal the dispenser of FIGS. 2 and 3.
FIG. 14 is a sectional view of the rear plug taken along line 14-14 of FIG.
13.
FIG. 14A is an enlarged portion of the rear plug of FIG. 14 showing further
detail of an a nular side wall of the plug.
FIGS. 15A-C are sequential side elevational views, partly in section, showing
the reduction in volume of fluid and corresponding expansion of the bladder in
the full, half-
full and empty conditions of the dispenser of FIG. 1, respectively.
FIGS. 16A-C are sequential side elevational views, partly in section, showing
the steps of assembling the bladder to the vial during sterilization and
filling of the dispenser
of FIGS. 2 and 3.
FIG. 17 is a perspective view of another embodiment of a dispenser of the
present invention.
FIG. 18 is an end elevational view of the dispenser of FIG. 17.
FIG. 19 is a cross-sectional view of the dispenser of FIGS. 17 and 18 taken
along line 19-19 of FIG. 18.
FIG. 20 is a cross-sectional view of the dispenser of FIGS. 17 and 18 taken
along line 20-20 of FIG. 18.
FIG. 21 is a perspective view of the rear plug of the dispenser of FIG. 17.
FIG. 22 is cross-sectional view of the rear plug of FIG. 21.
FIG. 23 is a partial, enlarged cross-sectional view of the rear plug of FIG.
21.
-12-
CA 02578803 2007-03-05
Fig. 24 is a partial, cross-sectional view of the axially-extending and
radially-
projecting legs of the rear plug of FIG. 21 illustrating the flexible bladder
conformably
engaging the legs in the predetermined collapsed condition.
FIG. 25 is a partial, cross-sectional view of the legs of the rear plug and
bladder illustrating the bladder in the expanded condition.
FIG. 26 is a cross-sectional view of the flexible bladder of the dispenser of
FIG. 17.
FIG. 27 is a partial, enlarged cross-sectional view of a portion of the
flexible
bladder of FIG. 26.
FIG. 28 is a front elevational view of the integral nozzle and slide of the
dispenser of FIG. 17.
FIG. 29 is a cross-sectional view of the integral nozzle and slide taken along
line 29-29 of FIG. 28.
FIG. 30 is a side elevational view of the integral piston and rigid vial of
the
dispenser of FIG. 17.
FIG. 31 is a partial, cross-sectional view of the integral piston and rigid
vial of
FIG. 30.
FIG. 32 is a cross-sectional view of the flexible nozzle cover and bellows of
the dispenser of FIG. 17.
FIG. 33 is a cross-sectional view of another embodiment of the dispenser of
the present invention including a resealable portion on the flexible bladder
for inserting a
needle or like injection member therethrough to fill the dispenser with a
medicament or other
substance, and allowing the needle holes to be sealed by application of
thermal energy
thereto.
-13-
CA 02578803 2007-03-05
Detailed Description of the Preferred Embodiments
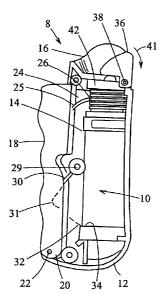
In FIG. 1, there is shown an ocular treatment apparatus 8 that may be used in
conjunction with a dispenser, shown generally at 10, in accordance with the
present
invention. As seen in FIGS. 1 and IA, the treatment apparatus 8 comprises a
housing 12 that
may be generally U-shaped in cross section, and defines an interior cavity 14
and an eye
cover 16. A trigger 18 is pivotally connected at one end 20 to the housing 12
via a hinge 22,
and includes at the other end an arm portion 24 defining a slot 25. As shown
best in FIG. 1A,
a pin 26 of a wheel 27 is fixedly secured within the slot 25, and the wheel 27
is rotatably
mounted on the interior wall of the housing 12. As best seen in FIG. 1, the
trigger 18 is
elongated and comprises finger grooves 28 for a comfortable fit with, e.g., a
patient's hand.
An approximately L-shaped spring arm 30 is fixedly secured at one end to a
post 29
projecting inwardly from the interior wall of the housing 12, and the spring
arm defines a
knee or bent portion 31 (shown in phantom) engaging an interior surface of the
trigger 18,
and a free end 32 engageable with a rim 34 formed at one end of the dispenser
10. An eyelid
depressor 36 is pivotally mounted by a hinge 38 to the end of the housing 12
adjacent to the
eye cover 16, and includes a hook 40 fixedly secured to the wheel 27 for
pivotally moving the
eyelid depressor upon actuating the trigger 18.
In use, the eye cover 16 is placed adjacent to the tissue surrounding the eye
with the eyelid depressor 36 engaging the tissue adjacent to the ocular cul-de-
sac. Upon
squeezing the trigger 18, the eyelid depressor 36 rotates in the direction of
the arrow 41, and
in turn moves the tissue adjacent to the eye to expose the ocular cul-de-sac.
Rotation of the
eyelid depressor 36 is caused by the wheel 27 which also uncovers a nozzle 42
formed at the
adjacent end of the dispenser 10. Simultaneously, the spring arm 30 forces the
rim 34 of the
dispenser 10 away from the fixed nozzle 42 to thereby prime the pump of the
dispenser, as
-14-
CA 02578803 2008-08-27
described in further detail below. Upon squeezing the trigger 18 and
correspondingly
extending the dispenser 10 within the housing 12, the free end 32 of the
spring arm 30
eventually disengages itself from the rim 34 of the dispenser to thereby
release the
extended dispenser from the spring arm. As a result, due to the resiliency or
spring-like
nature of the nozzle 42, as described further below, the extended dispenser
contracts or
moves back toward the nozzle and, in turn, releases a predetermined dosage of
medicament (or other substance) from the nozzle and into the ocular cul-de-sac
of the
user's eye. Then, when the user removes the ocular treatment apparatus 8 from
his or her
eye and releases the trigger 18, the spring arm 30 automatically returns to
its original or
resting position as shown in FIG. I with the free end 32 engaging the rim 34.
The force
exerted by the spring arm 30 upon returning to its original position also
rotatably drives
the wheel 27 in the direction opposite that of the arrow 41 and, in turn,
causes the eyelid
depressor 36 to return to its original position, as shown. The ocular
treatment apparatus is
then ready to dispense another predetermined dosage of medicament or other
liquid
contained therein.
Other examples of ocular treatment apparatus that may employ the dispenser
10 are described in U.S. Patent Nos. 4,981,479 and 6,033,384, which are
assigned to the
assignee of the present invention. Accordingly, as may be recognized by those
of ordinary
skill in the pertinent art based on the teachings herein, the dispensers of
the present
invention may be utilized in any of numerous different apparatus or systems to
facilitate
holding and dispensing medicaments or other fluids, liquids or other
substances contained
therein, such as nasal inhalers.
Referring now to FIGS. 2 and 3, the dispenser 10 is shown partly in cross
section
to illustrate the internal components thereof The dispenser 10 is generally
cylindrical
- 15 -
CA 02578803 2007-03-05
in outer configuration and comprises a pump assembly 50, a generally rigid
vial 52, and a
flexible bladder 54 disposed within a main fluid chamber 55 of the vial. The
pump assembly
50 comprises a piston 56 for discharging predetermined doses of medicaments or
other
substances contained within the fluid chamber 55, a slide or body 60 for
slidably receiving
therein the piston and defining a predetermined dosage chamber therebetween,
and a pump
cover 62 forming with a nozzle 58 a one-way valve at the dispensing tip, and a
spring-like
bellows for allowing either the piston or nozzle to be moved toward the other
to eject a dose
of medicament or other substance through the nozzle, and to force either the
piston or the
nozzle away from the other upon releasing the predetermined dose. The nozzle
58
hermetically seals the dispensing tip of the dispenser and ejects the pumped
medicament or
other substance therethrough.
Referring now also to FIG. 4, the piston 56 may be composed of any durable
and moldable material, such as a plastic substance and, preferably, the
material is suitable for
use in connection with medicaments. A suitable material is a low density
polyethylene. The
piston 56 comprises a base portion 64, an elongated portion 66 extending from
the base
portion 64, and a central bore 68 which is in fluid communication with the
main fluid
chamber 55. The base portion 64 is generally disc-like in outer configuration,
and comprises
a connecting flange 70, an annular mounting portion 72, a first annular groove
74, and a
second annular groove 76 spaced inwardly relative to the first annular groove
and
surrounding the inlet end of the central bore 68. The connecting flange 70 is
configured to
engage, e.g., in a snap-lock manner, the vial 52 defining a correspondingly
dimensioned
mounting flange 78 (FIG. 2). As shown in FIG. 2, the mounting portion 72 and
first annular
groove 74 receive an annular flange 80 and rib 82, respectively, of the pump
cover 62 which
is composed of a flexible material, as discussed in more detail below, and
which thereby seals
-16-
CA 02578803 2007-03-05
the main fluid chamber 55 of vial 52. As further shown in FIG. 2, when the
piston 56 is
assembled to the vial 52, the second annular groove 76 is located adjacent to
the main fluid
chamber 55. The second annular groove 76 thereby functions to provide a
capture area to
receive any gas bubbles improperly disposed within the main fluid chamber and
to prevent
the bubbles from passing into the central bore 68.
As shown best in FIG. 4, the elongated portion 66 comprises an annular
groove 84, a laterally-extending bore 86, and a terminal end defining a
receptacle portion 88.
The annular groove 84 is configured to receive a seal 90 (FIG. 2), such as an
o-ring, for
sealing the piston in contact with the slide 60. The laterally extending bore
86 is in fluid
communication with the central bore 68 and terminates adjacent to an annular
interior surface
92 of the slide 60 (FIG. 3). As shown best in FIG. 4, the receptacle portion
88 comprises an
annular wall 94, a tapered portion 95 extending between the annular wall 94
and bore 86, and
a piston surface 96 for stopping movement of the nozzle 58 as described in
more detail below
in connection with FIGS. 5 and 6. The annular wall 94 defines a generally
increasing outer
diameter toward the distal end, and slidably engages the annular interior
surface 92 of the
slide 60 as described in more detail below in connection with FIG. 7.
As shown in FIG. 3, the piston 56 further includes two generally
symmetrically-located hook portions 97, and each hook portion 97 defines in
combination
with an outer surface 101 of the piston 56 a respective slot 99. As described
in more detail
?0 below in conjunction with FIG. 7, the slide 60 is reciprocally disposed
within the slots 99 for
allowing relative movement of the piston within the slide upon actuation of
the pump.
Referring now to FIGS. 5 and 6, the nozzle 58 may be composed of any
suitably durable, moldable, somewhat flexible material (in the configuration
wherein the
nozzle and body are made of one piece), such as a plastic material, and
currently is composed
-17-
CA 02578803 2007-03-05
of a material which has been found to be compatible with medicaments, such as
those
materials sold under the trademarks VELEX and LEXAN, both owned by the General
Electric Company of Pittsfield, MA. The nozzle 58 is preferably molded of one
piece and
comprises a truncated, conical-shaped body portion 98, and a disc portion 100
disposed
coaxially with the conical-shaped portion and extending radially therefrom. It
will be
recognized that the conical-shaped portion 98 and disc portion 100 may be
molded together
or separately. The conical-shaped portion 98 comprises a tapered outer surface
102, a partial
central bore 104, and an engagement portion 106. The partial central bore 104
terminates at a
lever wall 108 which is dimensioned and configured to allow flexing of the
tapered outer
surface 102 in the direction of the arrow 110. The engagement portion 106 is
configured to
mate with the receptacle portion 88 of the piston 56, described above in
connection with FIG.
4, and comprises a truncated conical configuration terminating in an
engagement surface 112.
As illustrated in FIGS. 2 and 3, when the piston 56 reaches the end of its
stroke upon
dispensing a predetermined dose, the engagement surface 112 of the nozzle is
received within
5 the guide wall 94 and engages the piston surface 96 to terminate further
movement. It will be
recognized that a variable stroke volume 113 is defined between the engagement
surface 112
of the nozzle 58 and the piston surface 96 of the piston 56. As illustrated in
FIG. 3, the
maximum stroke volume is defined by the maximum extension of the engagement
surface
112 from the piston surface 96.
'-0 As shown in FIG. 6, the disc portion 100 comprises an annular mounting
portion 114 for affixably mounting the nozzle 58 to the pump cover 62 and
slide 60 (FIGS. 2
and 3), and also comprises a slot 116 for the passage of fluid or other
substances
therethrough. The mounting portion 114 comprises an annular thickened portion
118 and a
neck portion 120 disposed between a pair of annular grooves 122 and 124. As
shown in
-18-
CA 02578803 2007-03-05
FIGS. 2 and 3, the annular groove 122 is configured to engage a rib 126 of the
pump cover 62
(FIG. 8), and the annular slot 124 is configured to engage a correspondingly
configured
terminal end portion 128 of the slide 60 (FIG. 7). As shown in FIG. 6, the
annular groove
124 defines an annular crevice 130, employed, e.g., for easing assembly of the
slide 60 to the
nozzle 58. The slot 116 is disposed adjacent to a flattened portion 132 of the
tapered outer
surface 102, and provides fluid communication from the variable stroke volume
113 through
the disc portion 100 to the tapered outer surface.
As illustrated in FIG. 7, the slide 60 defines a tubular body and may be
composed of a similar substance to that described above with respect to the
nozzle 58 (FIGS.
5 and 6). As described above and referring also to FIG. 2, the slide 60
comprises an annular
inside surface 92 within which the piston 56 and engagement portion 106 of the
nozzle 58 are
disposed after assembly of the dispenser 10. The inside surface 92 defines a
bore 134 with a
neck portion 136 of reduced diameter disposed between a first transition zone
138 of
relatively rapid increase in diameter, and a second transition zone 140 of
relatively gradual
increase in diameter. Referring now to FIG. 2, it will be understood that
during relative
movement of the nozzle 58 and the slide 60 away from the piston 56, the
annular wall 94 of
the piston (FIG. 4) will engage the second transition zone 140 (FIG. 7) in
sealing engagement
to thereby force fluid contained within the variable stroke volume 113 into
the slot 116 of the
nozzle tip.
?0 With reference to FIG. 7 and as described above in connection with FIGS. 5
and 6, the slide 60 defines a terminal end 128 that includes an annular ridge
142 configured to
engage the crevice 130 of the nozzle 58. As shown in Fig. 3, the opposite end
of the slide 60
defines a flange 144 that is configured to engage the hook portion 97 of the
piston 56.
-19-
CA 02578803 2007-03-05
FIG. 8 depicts a cross-sectional view of the flexible pump cover 62. The
flexible pump cover 62 may be composed of any durable, resilient and flexible
material, such
as an elastomeric material. Preferably, the pump cover 62 is composed of a
thereto-elastic
material, such as a styrene-butadiene elastomer sold under the trademark
KRATON by GLS
of Illinois. Other suitable materials include polyvinylchloride, SantopreneTM
and butyl
rubber. The pump cover 62 comprises a mounting portion 146, a bellows portion
148, and a
nozzle cover 150 which cooperates with the slot 116 (FIG. 6) to provide an
elastic valve, as
described further below in connection with FIG. 5. As described above in
connection with
FIG. 4, the mounting portion 146 comprises an annular flange 80 that fits
within the
mounting groove 72 adjacent to the mounting flange 78 of the vial 52 (FIG. 2).
As shown in
FIG. 8, the rib 82 defines in cross section a truncated conical shape
corresponding to the
configuration of the annular groove 74 of the piston (FIG. 4). Because of the
resilient nature
of the material of the pump cover 62, the annular flange 80 may be slightly
oversized in order
to provide a resilient fit with the vial 52 and piston 56 and thereby, in
combination with the
rib 82, hermetically seal the main fluid chamber 55 (FIG. 2).
The bellows portion 148 extends between the mounting portion 140 and
nozzle cover 150, and comprises a plurality serpentine or inversely curled
portions 152 which
function to provide resiliency in a direction generally parallel to a central
axis 154 and
sufficient spring-like force to either drive the piston or the nozzle away
from the other and
return the piston to the top of its stroke upon dispensing a predetermined
dose of a
medicament or other substance contained within the chamber 55. Referring also
to FIGS. 2
and 6, the nozzle cover 150, when mounted, is dimensioned and configured to
resiliently
engage the nozzle 58 and slide 60, and includes the annular rib 126 extending
axially from a
disc engagement portion 156. The disc engagement portion 156 is disposed
between a slide
-20-
CA 02578803 2007-03-05
engagement portion 158 and a nozzle body engagement portion 160. Referring
also to FIG.
6, the nozzle body engagement portion 160 is configured to engage the tapered
outer surface
102 of the nozzle 58 to thereby form a normally-closed, one-way valve
therebetween. As can
be seen in FIG. 8, the cross-sectional thickness of the nozzle engagement
portion 160
gradually decreases in the axial direction from the disc engagement portion
156 toward the
dispensing tip 161. The gradually-decreasing cross-sectional thickness of the
nozzle
engagement portion 160 facilitates the release of the medicament or other
substance through
the one-way valve formed by the elongated, annular interface between the
relatively flexible
nozzle engagement portion 160 of the cover and the tapered surface 102 of the
nozzle body,
while simultaneously preventing air or other gases from passing through the
valve in the
opposite direction, in accordance with the teachings of the below-mentioned
patents.
In operation, as described above in connection with FIGS. 1 and 1 A,
movement of the vial 52 in the axial direction causes the piston 56 to move
from the position
shown in FIG. 2 into the position shown in FIG. 3 (or vice versa), e.g., by
actuating the
trigger 28 of FIG. 1 or other actuator, which draws fluid into the variable
volume fluid
chamber 113 from the main fluid chamber 55 via the central bore 68 and
laterally-extending
bore 86 of the piston. Referring now also to FIGS. 6 and 8, as the piston 56
moves toward
the nozzle 58 (or vice versa), the fluid is injected through the slot 116
(FIG. 6), along the
flattened surface 132, between the tapered surface 102 and nozzle body
engagement portion
160, and then outwardly of the nozzle tip. Further details of pump assemblies
that may be
used in the practice of the present invention are described in U.S. Patent
Nos. 5,944,702,
5,875,931 and 5,746,728, which are assigned to the same assignee as the
present invention,
-21-
CA 02578803 2007-03-05
One advantage of the pump configuration of the illustrated embodiment, and
as indicated by the arrow indicating the path of fluid flow in FIG. 6, the
pumped fluid follows
a fairly straight path extending in a direction parallel to the axis 154 from
the variable stroke
volume 113, over the tapered surfaces of the engagement portion 106, through
the slot 116,
and between the one-way valve formed by the interface of the nozzle engagement
portion 160
of the cover and the tapered outer surfaces-132 and 102 of the nozzle body.
This relatively
straight and smooth fluid flow path allows the pumped fluid to flow through
the nozzle with
relatively little head loss, thus allowing lesser force to dispense the fluid
and otherwise
facilitating precise control over the type of fluid flow to be emitted at the
dispensing tip, such
as control over drop size, flow velocity, or spray droplet size, spray
pattern, etc.
Yet another advantage of the illustrated pump configuration is that the
bellows
148 is sealed relative to the variable-stroke volume 113 to thereby prevent
any of the
medicament or other substance contained within the chamber 55 from collecting
in the space
between the bellows and the piston or slide. As can be seen, the o-ring or
like seal 90.forms a
fluid-tight seal between the piston and the slide, thus preventing any fluid
from flowing
therethrough and into the bellows. Similarly, fluid-tight seals are formed at
the interfaces of
the cover 62, nozzle 58 and slide 60, including fluid-tight seals at the
interfaces of the slide
engagement portion 158 of the cover and the slide 60, and at the interface of
the annular rib
126 of the cover and the annular groove 122 of the nozzle 58.
?0 Referring now to FIGS. 9 and 9A, the vial 52 is preferably composed of a
suitably rigid and moldable material, such as a rigid polymeric material,
e.g., polycarbonate
or polyvinylchloride. Preferably, this material is selected to be compatible
with a wide
variety of medicaments, such as that sold under the trademark Lexan of the
General Electric
Corporation of Pittsfield, MA. The vial 52 is tubular in configuration and
comprises an outer
-22-
CA 02578803 2007-03-05
wall 77 that defines the main fluid chamber 55, the annular mounting flange 78
discussed
above in connection with FIGS. 2 and 4, and an annular connecting portion 162
formed on an
opposite end of the vial relative to the mounting flange 78. The main fluid
chamber 55 is
dimensioned such that it is large enough to contain a predetermined quantity
of a fluid to be
dispensed, such as a medicament, along with the flexible bladder 54 (FIG. 2)
discussed in
more detail below. The mounting flange 78 includes an annular ridge 164 for
mounting the
vial 52 into sealing engagement with the nozzle cover 62 (FIG. 2) and
preventing movement
of the cover during use of the dispenser 10. As seen in FIGS. 2 and 3, the
mounting portion
146 of the cover 62 is sandwiched between the base 64 of the piston 56 and the
rigid vial 52
to form a fluid-tight seal.
As shown in FIG. 9A, the annular connecting portion 162 comprises a tapered
end 166 and a peripheral'groove 168 spaced inwardly therefrom on an increased
diameter
portion 170. An annular ridge 172 is provided for engaging the flexible
bladder 54 (FIG. 2). .
As described further below, the increased diameter portion 170 and annular
ridge 172
function to allow hermetic sealing of the main fluid chamber 55 after assembly
of the vial 52.
As also described fiirther below, an annular groove 174 is provided for
retention of the vial
52 during filling of the main fluid chamber 55.
Referring now to FIG. 10, the flexible bladder 54 may be composed of any
suitably flexible material, and preferably defines barrier properties to
prevent the passage
therethrough of vapor, moisture and gas. For ease of manufacture, the material
preferably
may be molded and is compatible with a wide variety of medicaments or other
substances to
be contained within the chamber 55, and therefore in a preferred embodiment
may be formed
of a rubber or synthetic rubber. Alternatively, the flexible bladder 54 may be
composed of a
thermo-elastic material, such as the styrene-butadiene elastomer sold under
the trademark
-23_
CA 02578803 2007-03-05
KRATON as discussed above in connection with the pump cover 62. Similarly,
materials
sold under the trademarks VISKAFLEX owned by the AES Company, ALCRYN or
HYTREL owned by the Dupont Company of Wilmington, D.E., and SARLINK owned by
the DSM Company may be used instead. These materials are only exemplary,
however. As
may be recognized by those skilled in the pertinent art based on the teachings
herein, the
flexible bladder may be made of any of numerous other materials that are
currently or later
become known for performing the fraction of the flexible bladder as disclosed
herein.
In the preferred embodiments of the present invention, the flexible bladder 54
is made of a resilient material as described above and is molded in the
expanded condition.
Accordingly, when collapsed in the manner described further below, the
resilient bladder
tends to force itself outwardly and, in tun, increase the pressure of the
medicament or other
fluid in the main fluid chamber 55 in comparison to the pressure in the
interior of the bladder.
A significant advantage of this pressure differential is that it facilitates
in preventing the
ingress of air, other gases or vapors located within the interior chamber of
the bladder
through the bladder or otherwise into the main fluid chamber. As a result, the
dispensers of
the present invention are particularly well suited for containing multiple
dose, non-preserved
medicaments or other substances, and in maintaining such substances in a
sterile, airless
condition, throughout substantial periods of storage, shelf life and/or use of
the dispensers.
This advantageous feature also facilitates in preventing any changes in the
ambient conditions
of the dispenser from affecting the airless condition of the main fluid
chamber 55, and
otherwise prevents the ingress of air, other gases or vapors into the main
fluid chamber.
The flexible bladder 54 preferably also provides a barrier to the passage of
gas, such as air, through the flexible bladder, and thus may be composed of a
single layer of
material that has a substantially reduced permeability to air. In one
embodiment of the
-24-
CA 02578803 2007-03-05
present invention, the bladder 54 is composed of a multi-layered material. For
example, as
illustrated in FIG. l OB, a bladder wall 175 may comprise a first flexible
layer 177 of an
elastomer that is relatively porous to air, and a barrier layer 179, such as a
metallized
MYLAR, e.g., an aluminum and polyester composition, sold by the Dupont
Corporation of
Wilmington, Delaware, that is relatively impervious to air. The barrier layer
179 may be
disposed between a polyethylene upper layer 181 and lower layer'183 to
facilitate adhesion of
the barrier layer to the bladder wall 175 while maintaining flexibility.
Alternatively, the
barrier layer 179 may be composed of polyvinylidene chloride sold under the
mark SARAN
owned by the Dow Chemical Company of Midland, Michigan. It will be appreciated
that the
barrier layer 179 is preferably dimensioned to cover as much of the bladder
wall 175 as
permitted in order to reduce the passage of air therethrough without
interfering with the
various functions of the flexible bladder as more fully described below. The
barrier layer 179
is also preferably disposed on the interior of the bladder wall 175.
Optionally, the barrier
material may be a butyl rubber-based material, such as that used for the
manufacture of
syringe stoppers, or used in the tire industry. As may be recognized by those
skilled in the
pertinent art based on the teachings herein, the flexible bladder and barrier
materials
disclosed herein are only exemplary, and any of numerous other materials that
are currently
known, or later become known for performing the functions disclosed herein,
may be equally
employed.
Referring now again to FIG. 10, the flexible bladder 54 is tubular in
configuration and comprises a closed end 176 and an open end 178 that fluidly
communicates
with a cavity 180. The bladder 54 defines an external diameter dimensioned to
fit within the
vial 52 (FIG. 2) when in the expanded condition as shown in FIG. 10. As shown
in FIGS. 2
and 3, the outer surface of the bladder 54 preferably defines a shape or
morphology
-25-
CA 02578803 2007-03-05
substantially the same as that of the interior surface of the rigid vial 52 so
that upon
expanding the flexible bladder, the flexible bladder conforms to and contacts
the rigid vial
throughout the interface of these two components to thereby eliminate any
ullage or dead
space between the components, and force all of the medicament or other
substance within the
chamber 55 into the variable stroke volume 113 of the pump 50 for dispensing
therefrom. In
addition, the outer diameter (or width) of the flexible bladder when fully
expanded is
preferably slightly greater than the inner diameter (or corresponding width)
of the rigid vial,
so that the expanded bladder may exert a resilient force against the vial to
maintain at least a
slight pressure differential between the chamber 55 on one side of the bladder
and the interior
l 0 of the bladder and thereby prevent the ingress of air, other gases or
vapors through the
bladder and into the main fluid chamber, as described above.
As shown in FIGS. 10 and 11, longitudinally extending stiffeners or rib
portions 182 are disposed along the inside surface 184 and function to provide
a supporting
structure about which the bladder 54 may collapse as will be described in more
detail below
in conjunction with FIG. 12. To achieve this, the rib portions 182 extend
axially along the
interior surface 184 and are approximately equally spaced about the
circumference of the
interior surface. It will be recognized that other configurations of the rib
portions 182 and/or
locations at which the rib portions may be employed are contemplated by the
present
invention. For example, the rib portions 182 also may extend along the inside
surface 183 of
?0 the closed end 176 of the flexible bladder 54.
As illustrated in FIGS. 10 and 1OA, the flexible bladder 54 includes a
mounting portion 186 that comprises an annular flange 188 formed at the rear
end of the
bladder, an outer annular lobe 190 spaced axially inwardly relative to the
flange 188, and an
inner annular lobe 192 spaced between the outer annular lobe 190 and the
flange 188. As
-26-
CA 02578803 2007-03-05
shown in FIG. 1 OA, the annular flange 188 defines on its underside an
annular, V-shaped
indent 194 for sealing engagement with the annular ridge 172 of the vial 52
(FIG. 9A). In
addition, the annular flange 188 is over-sized so that during initial assembly
with the vial 52,
as will be discussed in more detail below, the peripheral surface of the
annular flange may
engage the corresponding annular groove 168 of the vial 52 (FIG. 9A).
During storage and/or shelf life of the dispenser 10, the material of the
flexible
bladder 54 may flow or move in order to equalize the tensile and compressive
forces that it is
subject to. Creep, as used herein, refers to a change in property of the
material wherein there
is a loss in resilience and memory of the material. In particular, after
undergoing creep the
elastic material may permanently deform and lose at least some of its original
elasticity.
Accordingly, after assembly and during filling of the dispenser 10, the cavity
180 of the
flexible bladder 54 may be subject to low pressure which causes collapse and
elastic
deformation thereof which is maintained by the pressure of fluid filled in the
main fluid
chamber 55 (FIG. 2). Thereafter, the filled dispenser may be maintained in
storage and/or on
a store or other shelf for at least two or more months prior to use, during
which the material
of the bladder may undergo creep causing at least some deformation thereof. To
properly
manage the movement of the material during creep of the flexible bladder 54,
and as shown
best in FIG. I OA, the bladder is provided with the outer annular sealing lobe
190 and the
inner annular sealing lobe 192 spaced axially between the outer sealing lobe
and the flange
188 so that, when creep resulting from compression of the elastomeric or
rubber-like material
occurs, the intra material pressure is balanced in between the two lobes
190,192 and a
persistent, fluid-tight seal is provided. This mechanical seal can then be
maintained due at
least in part to the material reservoir formed by the inner lobe 192 in which
creeping material
in the outer lobe 190 offsets that of the inner lobe.
-27-
CA 02578803 2007-03-05
As shown in FIG. 1 OA, the outer annular lobe 190 comprises a first angular
portion 198 located on one side of the lobe 192, and a second angular portion
200 disposed
on the opposite side of the lobe. The first angular portion 198 defines a
first acute angle "A"
with respect to a center axis 202 that may be within the range of from
approximately 0 to
approximately 30 , and more preferably within the range of approximately 0 to
approximately 10 . The second angular portion 200 defines a second acute angle
`B" relative
to the axis 202 that may be within the range of approximately 0 to
approximately 15 , and
more preferably within the range of approximately 0 to approximately 5 . In
order to ensure
that during creeping of the material of the flexible bladder 54 the material
moves
approximately in the directions of the arrow 204 and arrow 205, the first
angle A is larger
than the second angle B and the flexible bladder is axially fixed by the inner
annular sealing
lobe 192 received within the corresponding annular groove 22 of the rear plug
(FIG. 14). As
shown in FIGS. 2 and when the flexible bladder 54 is fully received within the
rigid vial
52, the outer annular lobe 190 is pressed against the smooth interior wall of
the vial, the inner
annular lobe 192 is received within the corresponding annular groove 22 of the
rear plug
(FIG. 14), and the annular flange 188 is sandwiched between the rear plug and
the annular
ridge 172 of the rigid vial. Thus, the inner annular sealing lobe 192
functions as a material
reservoir for the outer annular sealing lobe 190, and as indicated by the
arrows 204 and 205,
the axially-offset lobes cause the material to flow generally from the outer
lobe 190 toward
the inner lobe 192, and from both lobes generally toward the annular flange
188. As a result,
the material flow is persistently directed toward the inner sealing lobe 192
and/or annular
flange 188 to thereby maintain a fluid-tight seal between the flexible
bladder, rigid vial and
rear plug, regardless of the degree of creep of the bladder material. As can
be seen, the shape
and relative position of the outer annular lobe 190 as described above
facilitates in directing
-28-
CA 02578803 2007-03-05
the forces within the bladder and thus the material in the directions of the
arrows 204 and 205
to thereby maintain the fluid-tight seal throughout the storage, shelf-life
and usage of the
dispenser 10.
As shown in FIG. 11, the flexible bladder 54 preferably also comprises at
least
one surface discontinuity 206 that facilitates and controls the collapse of
the bladder from a
tubular configuration to a predetermined collapsed configuration to thereby
substantially
eliminate the volume of the cavity 180 defined by the interior of the bladder.
In the
illustrated embodiment, the flexible bladder comprises three surface
discontinuities 206
located on the interior surface 184 of the bladder and approximately equally
spaced relative
to each other. As can be seen, the discontinuities 206 are each approximately
equally spaced
between adjacent elongated ribs 182. The discontinuities 206 are illustrated
in the
configuration of a crevice or crack terminating in a generally flat center
portion (not
numbered) in cross section as shown. As can be seen, the surface
discontinuities 206 cause
the bladder to collapse or fold onto itself about each elongated rib 182 to
thereby form in the
collapsed condition three folded sections or legs spaced about 120 relative
to each other. As
may be recognized by those skilled in the pertinent art based on the teachings
herein, and
illustrated by the additional embodiments below, any of numerous other
structures or
configurations may be equally employed to collapse the bladder into a
predetermined shape,
such as the predetermined collapsed shape formed by discontinuities and
elongated ribs
described above.
Referring now to FIG. 12, a cross section of an outer wall 208' of a flexible
bladder 54' is illustrated in schematic. The flexible bladder 54' is capable
of collapsing in the
direction of arrows 210' from an expanded position, shown in solid lines
adjacent to the outer
wall 77 of the vial 52 (FIG. 9) to a collapsed position shown in broken lines.
The flexible
-29-
CA 02578803 2007-03-05
bladder 54' is functionally similar to the flexible bladder 54 and thus like
elements are labeled
with like reference numerals followed by the prime (') symbol. However, it
will be
recognized that some differences in structure exist between the flexible
bladder 54 and the
flexible bladder 54'. For example, discontinuities 206' are illustrated as
being inverted with
respect to the discontinuities 206 and generally cover the entire cross-
sectional thickness (t')
of the wall 208'. While the discontinuities 206' are illustrated as generally
arcuate in
configuration, it will be appreciated that other configurations, such as that
of discontinuities
206, which also perform the function described below may be employed instead.
Also, it will
be appreciated that the rib portions 182' define mounting slots 209' for
receiving therein
correspondingly-shaped portions (not shown) of the vial 52.
It will be understood that both the discontinuities 206 and 206' function to
allow for a reduction in length of the portions of the wall 208' necessary to
collapse the
flexible bladders 54 and 54'. While this function is being described in
connection with the
embodiment of FIG. 12, this description is equally applicable to the
embodiment of FIG. 11.
As shown in FIG. 12, the wall 208' comprises a plurality of wall portions 212'
extending
between each discontinuity 206' and adjacent rib portion 182', and as shown in
solid lines
each wall portion 212' forms an arc when the bladder 54' is expanded. It will
be recognized
that during collapsing of the bladder 54' in the directions of the arrows
210', the wall
portions 212' become approximately linear and form a chord as shown in the
dashed and
dotted lines, and then inversely arcuate as illustrated in the dashed lines.
Accordingly, as
illustrated, a length Li of the discontinuities 206' shown in solid lines
shortens to a length L2
shown in dotted and dashed lines to thereby allow free movement of the arcs
212' in the
direction of the arrows 210'. Once the flexible bladder 54' has collapsed, the
bladder may
-30-
CA 02578803 2007-03-05
expand and the wall portions 212' may freely move in a direction opposite that
of the arrows
210.
As illustrated in FIGS. 13 and 14, the flexible bladder comprises a rear plug
214 configured to mate with the open end 178 of the flexible bladder 54 (FIG.
2) and to seal
the flexible bladder 54 disposed between the rear plug and the rigid vial 52.
The rear plug
214 may be composed of any suitably strong, moldable and durable material,
such as a
polymeric material, e.g., polyethylene, and is preferably composed of LexanTM
or a like
polycarbonate for its stress-resistant properties. The rear plug 214 comprises
an end wall
216 and a side wall 218 that, as seen best in FIG. 14A, preferably includes a
tapered portion
220 defining a gradually increasing diameter in the direction of the rear end
of the plug, an
annular groove 222 spaced rearwardly of the tapered portion 220, a stepped
portion 224, a
plurality of outwardly-protruding protective tabs 226 (or bumps for ease of
manufacturing)
angularly spaced relative to each other about the axis of the plug, and an
aperture 228
extending through the side wall for allowing fluid communication between the
interior
chamber 180 of the bladder and the ambient atmosphere. The tapered portion
220, because
of the increasing diameter thereof, provides for ease of assembly of the plug
214 to the
flexible bladder 54 (FIG. 2) and defines an annular space 230 (FIG. 2) located
between the
plug and the adjacent surface of the flexible bladder. As described above and
shown in FIGS.
2 and 3, the annular groove 222 is configured to receive the inner annular
lobe 192 (FIG. 10)
and the stepped portion 224 sandwiches the annular flange 188 of the flexible
bladder 54
against the annular ridge 172 of the rigid vial. As best seen in FIGS. 13 and
14, the safety
sealing tabs 226 project upwardly and outwardly from the stepped portion 224
and are
angularly spaced relative to each other about the axis of the plug. The
sealing tabs 226 are
provided for locking the plug 214 to the vial 52 (FIG. 2) and are configured
to snap-fit within
-31-
CA 02578803 2007-03-05
the annular groove 168 of the vial (FIG. 9) upon being pressed against the
annular flange 188
of the bladder to thereby maintain an airtight seal. In addition, because the
sealing tabs 226
are tapered outwardly as shown typically in FIG. 14A, the tabs easily snap
into the annular
groove 168 of the vial; however, the tabs cannot be moved out of the groove in
the opposite
direction and thereby form a tamper-proof seal. The aperture 228 provides for
fluid
communication between the annular space 230, chamber 180 (FIG. 2) and ambient
atmosphere, and is illustrated as being generally rectangular in
configuration. However, it
will be understood that other configurations, such as circular or other
shapes, may be
employed, providing that a sufficient volume of air may pass therethrough to
fill the interior
chamber 180 of the flexible bladder 54.
Returning to FIGS. 10 and 1 OA, the flexible bladder 54 also preferably
comprises a two-way valve 234 axially spaced below the sealing lobes 190 and
192 for
controlling the flow of air between the interior chamber 180 of the bladder
and ambient
atmosphere. The valve 234 comprises an annular operator 235 projecting
inwardly from the
i 5 interior wall of the bladder and having a generally ridge-like
configuration in cross section.
As shown in FIGS. 2 and 3, the end portion of the annular operator 235 engages
the annular
surface 232 formed at the base of the rear plug 214, and is disposed between
the annular
space 230 and the interior chamber 180 of the bladder. The flexible bladder 54
further
defines a plurality of support protuberances 236 that are axially spaced
adjacent to the
'.0 annular operator 235 and angularly spaced relative to each other about the
axis 202. The end
surface of each protuberance 236 is spaced inwardly relative to the end of the
annular
operator 235 to thereby allow the operator 235 to engage and seal the
interface between the
operator and rear plug, while simultaneously ensuring sufficient radial
spacing between the
rear plug and flexible bladder for allowing movement of the operator 235 in
either direction.
-32-
CA 02578803 2007-03-05
Thus, as can be seen, the operator 235 and annular wall 232 of the rear plug
form a two-way
valve allowing fluid to flow therethrough when the differential pressure
across the valve is
sufficient to axially flex the operator. It will be understood that the
rigidity of the operator is
set to allow fluid to pass therethrough when the pressure differential exceeds
a predetermined
threshold pressure. Thus, a significant advantage of the valve 234 is that it
maintains a
relatively stable micro-atmosphere within the inner chamber 180 of the
flexible bladder 54
and prevents a regular exchange of air, other gases or vapors between the
micro-atmosphere
within the bladder and the ambient atmosphere. For example, the valve 234
allows air to be
drawn into the chamber 180 upon dispensing the medicament or other substance
from the
main fluid chamber 55 to thereby allow the bladder to expand and fill the
space of the
dispensed medicament. However, the valve 234 otherwise prevents air or vapors
from
flowing freely between the micro-atmosphere and the ambient atmosphere. Thus,
the micro-
atmosphere within the chamber 180 may define different pressure and/or
humidity levels in
comparison to the ambient atmosphere. A significant advantage of this feature
is that it
insulates the micro-atmosphere from fluctuations in the pressure and/or
humidity levels of the
ambient atmosphere, thereby maintaining relatively stable pressure and
humidity levels
within the micro-atmosphere and thus preventing the ingress of air or vapors
through the
bladder wall and into the main fluid chamber.
In FIGS. 15A-15C, the dispenser 10 is illustrated in the full, half-full and
empty conditions, respectively. In FIG. 15A, the main fluid chamber 55 is
filled with, e.g., a
medicament (not shown) that the pump assembly 50 may pump outwardly of the
nozzle 58.
Accordingly, the bladder 54 is illustrated in a collapsed state. In FIG.. 15B,
the flexible
bladder 54 is shown in an expanded condition whereby the bladder has expanded
to displace
the volume of medicament dispensed from the main fluid chamber 55. To achieve
this result,
-33-
CA 02578803 2007-03-05
air has passed in the direction of arrow 240, through the valve 234 and into
the interior
chamber 180 of the flexible bladder. In FIG. 15C, the dispenser 10 is
illustrated in an empty
condition. As can be seen, the bladder 54 is fully expanded against wall 77 of
the rigid vial
and substantially conforms to the morphology of the rigid vial to thereby
eliminate any ullage
or dead space and force all medicament or other substances therein into the
pump 50.
Referring now to FIGS. 16A-I6C, initial assembly of the dispenser 10 for
purposes of sterilization, e.g., by irradiation of energy rays, is illustrated
in FIG. 16A. In
particular, the rear plug 214 is fitted to the flexible bladder 54, and the
plug and flexible
bladder are partially inserted into the vial 52. Turning now also to FIGS. 9A
and 10, the
flange 188 of the flexible bladder 54, when in the partially inserted
position, is disposed
within the annular groove 168 of the vial 52 to thereby form an air-tight, but
not a tamper-
proof seal between the bladder and vial. In this state, the dispenser 10 may
be sterilized
and/or transported in a sealed condition prior to filling the dispenser with a
medicament or
other substance to be contained therein.
The filling of the dispenser 10 is illustrated schematically in FIG. 16B,
wherein the flexible bladder 54 and plug 214 are separated from the vial 52 so
that the main
fluid chamber 55 may be accessed for filling. As can be seen, the annular
flange 188 of the
bladder may be pulled rearwardly and removed from the annular groove 168 of
the vial to
thereby open the vial and access the main fluid chamber 55. Preferably, this
operation is
carried out by transporting the sterilized dispensers through a sterile
transfer port, and filling
the dispensers within a sterile filling machine of the types disclosed in
commonly-assigned
U.S. Patent Nos. 5,641,004 and 5,816,772.. During filling, a vacuum may be
drawn on the
-34-
CA 02578803 2007-03-05
inner chamber 180 of the bladder to collapse the bladder, and the medicament
or other
substance to be contained therein may be introduced into the main fluid
chamber 55.
As shown in FIG. 16C, upon filling the main fluid chamber 55 with the
medicament or other substance to be contained therein, the flexible bladder
and rear plug
assembly are moved into the rigid vial such that the flexible flange 188 of
the bladder is
moved into engagement with the annular ridge 172, best seen in FIG. 9A, and
the rear plug is
pressed inwardly until the sealing tabs 226 are snapped into place within the
annular groove
168 of the vial to thereby form the airtight and tamper-proof seal. The
dispenser 10 may then
be installed within the ocular treatment apparatus 8 described above or other
suitable
apparatus for dispensing medicaments or other fluids, such as nasal inhalers.
In FIGS. 17 through 20, another embodiment of the dispenser of the present
invention is indicated generally by the reference numeral 310. The dispenser
310 is
substantially similar to the dispenser 10 described above, and therefore like
reference
numerals preceded by the numeral "3", or preceded by the numeral "4" instead
of the numeral
" 1 ", or preceded by the numeral "5" instead of the numeral "2",
respectively, are used to
indicate like elements. The primary differences of the dispenser 310 in
comparison to the
dispenser 10 are that (i) the rigid vial 352 and piston 356 are formed as
integral components;
(ii) the nozzle 358 and slide 360 are formed as integral components; (iii) the
flexible bladder
354 defines a smooth cylindrical configuration without any discontinuities or
ribs formed
thereon; and (iv) the rear plug 514 includes a plurality of inwardly
projecting legs 538 for
controlling the collapse of the flexible bladder into a predetermined
collapsed shape.
As shown in FIGS. 21-23, the rear plug 514 defines a plurality of inwardly
projecting, axially-elongated legs 538 defining a framework within the
interior chamber 480
of the flexible bladder 3 54 for controlling the collapse of the bladder into
a predetermined
-35-
CA 02578803 2007-03-05
collapsed shape. As shown in FIG. 21, the currently preferred embodiment of
the present
invention includes three legs 538 angularly spaced approximately 120 relative
to each other
about the axis of the rear plug. Each leg lies in a respective plane
intersecting the axis of the
rear plug and defines approximately planar side surfaces 540 extending
radially between the
axis of the plug and the inner wall of the rigid vial. As shown in FIGS. 19
and 20, the radial
edge 542 of each leg is radially spaced inwardly relative to the inner wall of
the rigid vial to
thereby allow movement of the flexible bladder between the radial edges of the
legs and the
vial. As also shown in FIGS. 19 and 20, the legs 538 extend axially into the
interior chamber
480 of the flexible bladder a distance sufficient to allow the legs to control
the collapse of the
bladder into the predetermined collapsed condition. In the illustrated
embodiment, each leg
53 8 extends along at least about one-half the axial extent of the bladder. As
shown in FIG.
24, in the predetermined collapsed condition, the flexible bladder 354
conformably engages
the outer surfaces of the legs 538 to thereby allow the main fluid chamber 355
to be filled
with a medicament or other substance. Then, as shown in FIG. 25, upon
dispensing all of the
medicament or other substance from the main fluid chamber 355, the resilient
nature of the
flexible bladder 354 causes the bladder to expand outwardly away from the legs
538. As
shown typically in FIGS. 19 and 20, when fully expanded, the flexible bladder
354
conformably engages the inner wall of the rigid vial to thereby eliminate any
ullage or dead
space and allow all of the medicament or other substance contained with the
main fluid
chamber 355 to be dispensed therefrom.
As described above, the flexible bladder 538 is preferably made of a
relatively
low permeability elastomer, such as a vulcanized butyl rubber, or other
rubbers. Such
rubbers have demonstrated proven stability and/or compatibility with a wide
variety of
medicaments, such as pharmaceutical preparations and vaccines, and other
substances, and
-36-
CA 02578803 2007-03-05
therefore are currently preferred for such applications. In the currently
preferred
embodiment, the flexible bladder 354 is molded in its expanded condition, and
when
collapsed, the resilient nature of the bladder tends to force the bladder
outwardly toward its
expanded condition. The resilient forces within the bladder apply a pressure
against the fluid
within the main fluid chamber 355, and therefore create a higher pressure in
the main fluid
chamber 355 in comparison to that of the interior chamber 480 of the bladder.
As a result,
the pressure differential prevents the ingress of air or other gases or vapors
through either the
flexible bladder or rigid vial, or otherwise into the main fluid chamber.
Thus, the material
and/or configuration of the bladder are preferably selected to maintain a
pressure differential
sufficient to prevent the ingress of air or other gases or vapors into the
main fluid chamber
under a variety of atmospheric conditions. As described above, the preferred
rubber
materials disclosed herein for constructing the flexible bladder are
exemplary, and numerous
other materials that are currently, or later become known for performing the
function of the
flexible bladder may be equally employed.
As shown in FIGS. 26 and 27, the spaced protuberances 236 described above
in connection with the flexible bladder 54 of FIG. I OA may be eliminated
depending upon
the material of construction and/or other structural features of the flexible
bladder 354. In
addition, the outer annular lobe may take a shape different than that
illustrated above in
connection with the bladder of FIG. 10A. As shown in FIG. 27, the outer
annular lobe 490 is
defined by an annular raised or thickened portion, and a tapered surface 498
extending
radially inwardly between the lobe or annular raised portion 490 and the outer
peripheral
surface of the flexible bladder 354. As shown in FIGS. 19 and 20, the annular
raised portion
490 is squeezed against the inner surface of the rigid vial 352 which, in
combination with the
axially offset, inner annular lobe 492 being fixedly received within the
annular groove 522 of
-37-
CA 02578803 2007-03-05
the rear end cap (FIG. 23), cause the material of the flexible bladder to
creep and/or otherwise
flow in the directions of the arrows 504 and 505 in FIG. 27 to thereby
persistently maintain
an airtight seal between the flexible bladder, rear plug and rigid vial. Thus,
the end seal of
the flexible bladder is both radially compressed at the axially offset lobes
between the rear
plug and rigid vial, and is axially compressed at the flange between the rear
plug and rigid
vial.
As shown typically in FIG. 22, the rear plug 514 defines three apertures 528
approximately equally spaced relative to each other about the axis of the
plug. In addition,
rather than defining the sealing tabs 226 described above in connection with
FIG. 14, the rear
plug 514 defines an annular lobe 526 projecting outwardly from the peripheral
surface of the
rear plug and dimensioned to be snapped into the annular groove 168 of the
rigid vial (FIG.
30). The dispenser 310 may be sterilized, temporarily closed, re-opened, and
filled in the
same manner as described above in connection with FIGS. 16A through 16C.
As shown in FIGS. 28 and 29, the nozzle 358 and slide 360 are formed
integral with each other. One advantage of this construction over the separate
nozzle and
slide described above in connection with the previous embodiment, is that the
integral
construction is typically less costly to manufacture and assemble, and
furthermore, reduces
the number of seals between components and thereby enhances the overall
reliability of the
dispenser.
As shown in FIGS. 30 and 31, the piston 356 and rigid vial 352 are also
formed integral with each other. As with the integral nozzle and slide
described above, one
advantage of this construction over the separate piston and slide described
above in
connection with the previous embodiment, is that the integral construction is
typically less
costly to manufacture and assemble, and furthermore, reduces the number of
seals between
-38-
CA 02578803 2007-03-05
components and thereby enhances the overall reliability of the dispenser. In
the currently
preferred embodiment of the present invention, the integral nozzle 358 and
slide 360 is made
of a relatively soft material, and the integral piston 356 and vial 352 is
made of a relatively
hard material. In the operation of the dispenser 310, on the downward stroke
of the piston
356, and upon reaching the compression zone 436 of the slide 360, the relative
hardness and
geometry of the illustrated piston causes the piston to force the compression
zone 436 of the
slide outwardly and thereby form a fluid-tight seal between the piston and
slide. As
illustrated in FIG. 30, the tip of the guide wall 394 defines a chamfer for
facilitating sliding
movement of the piston within the slide.
Forming the integral nozzle and slide of a relatively soft and/or flexible
material allows the compression zone 436 of the slide to flex outwardly in
order to remove
the part from a core pin upon molding the part, and thus enables the nozzle
and slide to be
integrally molded as a single part. Preferably, compressed air is injected
between the core
pin and interior surface 392 of the slide to facilitate removal of the part
from the core pin (not
shown).
As shown in FIGS. 19 and 20, when the flexible bladder 354 is at or near its
fully-expanded condition, an annular gap "C" is formed between the bladder and
vial. As can
be seen, the width of the gap C gradually increases in the axial direction
moving from the rear
end cap 514 toward the closed end 476 of the bladder. As can be seen, the gap
C starts about
half-way down the axial extent of the bladder and reaches its maximum width at
the curved
portion of the bladder between the side wall and end wall 476. The gap C may
be created by
forming the approximately cylindrical side wall of the flexible bladder 354
with a sufficient
draft to form the gap upon insertion of the bladder into the rigid vial. The
purpose of the
gradually-increasing gap C is to force all fluid within the main fluid chamber
355 in the
-39-
CA 02578803 2007-03-05
direction toward the pump 350 and prevent the formation of any pockets of
fluid within the
main fluid chamber that cannot be dispensed therefrom.
As shown typically in FIGS. 19 and 20, other than the slight differences
necessary to create the gap C, the flexible bladder 354 defines approximately
the same
morphology as the interior surfaces of the rigid vial 352, thus enabling
intimate and
conforming engagement of the bladder with the rigid vial upon full expansion
of the bladder.
In, addition, the flexible bladder 354 preferably defines in its fully
expanded condition an
outer diameter (or width) at least equal to or greater than the inner diameter
(or width) of the
chamber 355 of the rigid vial. These features, in combination with the
resilient nature of the
flexible bladder, prevent the ingress of gases or vapors into the main fluid
chamber 355, and
ensure usage of substantially all fluid contained within the chamber.
As shown in FIG. 32, the flexible cover 362 defines an annular mounting
flange 380 on one end thereof which is received within a corresponding annular
groove 374
formed on the integral piston and rigid vial (FIGS. 30 and 31) to fixedly
secure the flexible
cover thereto. In addition, the integral piston and rigid vial defines an
annular flange 381
adjacent to the annular groove 374 which is received within a corresponding
annular groove
382 of the flexible cover (FIG. 32) to further secure the cover thereto.
Turning to FIG. 33, another embodiment of the dispenser of the present
invention is indicated generally by the reference numeral 610. The dispenser
610 is
substantially the same as the dispenser 310 described above, and therefore
like reference
numerals preceded by the numeral "6" instead of the numeral "3", the numeral
"7" instead of
the numeral "4", or the numeral "8" instead of the numeral "5", respectively,
are used to
indicate like elements. The primary difference of the dispenser 610 in
comparison to the
dispenser 310 is that the dispenser 610 includes a resealable bladder to allow
the bladder to
j -40-
CA 02578803 2007-03-05
be filled in a sterile filling machine of the type disclosed in U.S. Patent
6,604,561
which is assigned to the same Assignee as the present invention.
As shown in FIG. 33, the flexible bladder 654 includes on its closed end
776 a resealable portion 844 overlying the closed end 776. In the illustrated
embodiment, the flexible bladder 354 is formed of a first material compatible
with the
predetermined medicament or other substance to be contained within the main
fluid
chamber 655, and defines on its external side a medicament-exposed surface
intended
to be exposed or otherwise placed in contact with the predetermined medicament
or
other substance contained within the main fluid chamber. The resealable
portion 844 is
penetrable by a needle or like filling member for introducing the
predetermined
medicament or other substance through the flexible bladder and into the main
fluid
chamber. The penetrable region of the flexible bladder is formed of a
vulcanized
rubber, and therefore is substantially infusible in response to the
application of thermal
energy thereto. The penetrable region of the resealable portion 844, on the
other hand,
is fusible in response to the application of thermal energy thereto, thus
allowing the
penetrable region of the resealable portion to be hermetically sealed upon
removing the
needle or like filling member therefrom. In the illustrated embodiment, the
resealable
portion 844 is insert molded onto the rubber bladder during which the
thermoplastic
resealable layer bonds itself to the underlying rubber layer. If necessary, a
mechanical
fastener of a type known to those skilled in the pertinent art may be used to
facilitate
attachment of the resealable portion to the end wall of the flexible bladder.
The resealable member 844 is preferably made of a resilient polymeric
material,
such as a blend of the polymeric material sold by GLS under the registered
-41-
CA 02578803 2007-03-05
trademark KRATON and a low-density polyethylene, such as the polyethylene
sold by
Dow Chemical Co. under the trademarks ENGAGETM or EXACTTM. An important
feature of
the resealable member 844 is that it be resealable to form a gas-tight seal
after inserting a
needle, syringe or like injection member through the resealable member.
Preferably, the
resealable member can be sealed by heating the area punctured by the needle in
a manner
known to those skilled in the pertinent art and described in the above-
mentioned co-pending
patent application. One advantage of the blended polymer described above is
that it is known
to minimize the degree to which the medicament or other substance can be
absorbed into the
polymer in comparison to KRATON itself.
As shown in FIG. 33, the rear plug 514 defines a filling aperture 846 formed
therethrough and overlying the resealable member 844. As shown in broken lines
in FIG. 33,
a double lumen needle or like injection member 848 may be reciprocally moved
through the
filling aperture 846 to, in turn, pierce both the resealable member 844 and
underlying closed
end 776 of the flexible bladder. The injection member 848 is coupled in fluid
communication
with a source (not shown) of medicament or other substance to be contained
within the main
fluid chamber 655 and is actuated to fill the chamber with the medicament or
other substance.
Upon filling the chamber, the flexible bladder 654 is collapsed into its
predetermined
collapsed condition, as shown above, and the needle is withdrawn. If
necessary, a vacuum
may be drawn on the interior chamber 780 of the flexible bladder during
filling to facilitate
collapse of the bladder. Upon withdrawing the needle, a laser or other energy
source (not
shown) transmits a beam of laser radiation onto the penetrated region of the
resealable
member to seal the needle hole in the manner described in the above-mentioned
co-pending
patent application and thereby maintain the medicament or other substance
contained therein
in a sterile, hermetically sealed condition. The filling aperture 846 may be
sealed with a cap
-42-
CA 02578803 2007-03-05
850 (shown in broken lines) to maintain the interior chamber 780 of the
flexible bladder in a
sealed condition.
As may be recognized by those skilled in the pertinent art based on the
teachings herein, numerous changes and modifications may be made to the above-
described
and other embodiments of the present invention, without departing from its
scope as defined
in the appended claims. Accordingly, this detailed description of preferred
embodiments is to
be taken in an illustrative, as opposed to a limiting sense.
-43-