Note: Descriptions are shown in the official language in which they were submitted.
CA 02588614 2007-05-23
Method For Analyzing The Amount Of
Intraabdominal Adipose Tissue
This is a divisional application of Canadian Patent Application Serial No.
2,366,523 filed on Apri16, 2000.
BACKGROUND OF THE INVENTION
1. Field of the Invention
The present invention relates to a method for analyzing the amount of
intraabdominal adipose tissue and its uses. It should be understood that the
expression
"the invention" and the like encompasses the subject matter of both the parent
and the
divisional applications.
2. Description of the Related Art
Recently, it was revealed that the increase of adipose tissue in the
peritoneal
cavity, particularly the increase of adipose tissue by accumulating fat into
adipose tissue
which is present around vessels flowing in the portal veins, such as
mesenteric adipose
tissue or omental adipose tissue, is related closely to the onset of diseases
including
metabolic disorders such as diabetes, hyperlipemia and arteriosclerosis as
well as cardiac
vessel disorders such as coronary artery diseases, angina and myocardial
infarction
("Naizo Shibogata Himan (Visceral Fatty-Type Obesity"), 1995, published by
lyaku
Journal Ltd.). In order to predict the risk of onset of such diseases, there
is a need for the
development of an analytical method which can easily and rapidly manage the
amount of
intraabdominal adipose tissue.
As a method for analyzing the amount of intraabdominal adipose
tissue, a method for estimating a waist/hip peripheral diameter ratio (W/H
ratio) as an indicator (J. Clin. Endocrinol. Metab., vol. 54, p. 254. 1982)
has
1
CA 02588614 2007-05-23
been reported, but the W/H ratio provides a rough estimate of the amount of
fat in the whole abdomen area and thus cannot clearly distinguish the
amount of subcutaneous adipose tissue from the amount of intraabdominal
adipose tissue. Accordingly, the method for estimating the amount of
intraabdominal adipose tissue by using the W/H ratio as an indicator fails
to be satisfactorily accurate and is inadequate as a method for analyzing the
amount of intraabdominal adipose tissue.
SUMMARY OF THE INVENTION
Accordingly, there is demand to develop an analytical method
which can easily and rapidly manage with satisfactory accuracy, the
amount of intraabdominal adipose tissue.
As a result of studying under these circumstances and thereby
arriving at the present invention, the present Inventors have found that
there is a direct relationship or positive correlation between the
concentration of a certain protein in blood and the area of intraabdominal
adipose tissue in a section of the abdomen. As such, the amount of
intraabdominal adipose tissue can be determined, based on the
concentration of said protein.
That is, the present invention provides:
1. a method for analyzing the amount of intraabdominal adipose tissue
comprising:
determining the amount of intraabdominal tissue in a test-animal from the
concentration of a protein in a body fluid, tissue or cell of said test-animal
or
in a prepared test solution from the body fluid, tissue or cell of said test-
2
CA 02588614 2007-05-23
animal, based on the positive relationship between the amount of the
intraabdominal adipose tissue in an animal and the concentration of the
protein in a body fluid, tissue or cell of said animal or in a prepared
control
solution from the body fluid, tissue or cell of said animal,
wherein the protein is (1) encompassed by the amino acid sequence shown
in SEQ ID NO:1 or is (2) recognized by an antibody against a protein
encompassed by the amino acid sequence shown in SEQ ID NO: 1.
2. a method according to the above 1, wherein the amount of
intraabdominal adipose tissue is the area of intraabdominal adipose tissue
in a section of the abdomen.
3. a method according to the above 1, wherein the relationship is
expressed as a linear function of a correlative function.
4. a method according to the above 1, wherein an immunochemical
analysis determines the concentration of the protein in the body fluid tissue
or cell of the animal or in a prepared solution from the body fluid, tissue or
cell of said animal.
5. a method for analyzing intraabdominal adipose tissue comprising:
determining the amount of intraabdominal adipose tissue in a test-animal
from the concentration of a protein in a body fluid, tissue or cell of said
test-animal or in a prepared solution from the body fluid, tissue or cell of
said test-animal,
3
CA 02588614 2007-05-23
wherein the protein is (1) encompassed by the amino acid sequence shown
in SEQ ID NO:1 or is (2) recognized by an antibody against a protein
encompassed by the amino acid sequence shown in SEQ ID N0:1.
6. a method according to the above 1 or 5, wherein an immunochemical
analysis determines the concentration of the protein in the body fluid, tissue
or cell of said test-animal or in a prepared solution from the body fluid,
tissue or cell of said test-animal.
7. a method according to the above 1 or 5, wherein the test-animal is a
mammal.
8. a method according to the above 1, 2, 3, 4, 5 or 6, wherein the body fluid,
tissue or cell of said test-animal or the prepared test solution from the body
fluid, tissue or cell of said test-animal is blood.
9. an examination method comprising:
predicting the risk of onset of a disease closely related to the amount of
intraabdominal adipose tissue or judging the recovery of said disease in an
individual, based on examining the increasing or decreasing amount of
intraabdominal adipose tissue of said individual for a predetermined period
by the method of any one of the above 1 to 8.
10. an examination method comprising:
predicting the risk of onset of a disease closely related to the amount of
4
CA 02588614 2007-05-23
intraabdominal adipose tissue or judging the recovery of a disease in an
individual of a test-animal, based on examining the increasing or
decreasing concentration of a protein in a body fluid, tissue or cell of said
test-animal or in a prepared test solution from the body fluid, tissue or cell
of said test-animal, for a predetermined period,
wherein the protein is (1) encompassed by the amino acid sequence shown
in SEQ ID N0:1 or is (2) recognized by an antibody against a protein
encompassed by the amino acid sequence shown in SEQ ID N0:1.
11. an examination method comprising:
determining the amount of intraabdominal tissue in a test-animal with a
method of any one of the above 1 to 8;
comparing the amount of intraabdominal adipose tissue in said test-animal
to the amount of intraabdominal adipose tissue in a healthy animal of the
same species of said test-animal; and
predicting the risk of onset of a disease closely related to the amount of
intraabdominal adipose tissue or judging the recovery of a disease in said
test-animal.
12. an examination method comprising:
determining the concentration of a protein in a body fluid, tissue or cell of
a
test-animal or in a prepared test solution from the body fluid, tissue or cell
of said test-animal;
comparing the concentration of the protein in said test-animal to the
concentration of the protein in a healthy animal of the same species of said
CA 02588614 2007-05-23
test-animal; and
predicting the risk of onset of a disease closely related to the amount of
intraabdominal adipose tissue or judging the recovery of a disease in said
test-animal,
wherein the protein is (1) encompassed by the amino acid sequence shown
in SEQ ID NO:1 or is (2) recognized by an antibody against a protein
encompassed by the amino acid sequence shown in SEQ ID NO:1.
13. use of a protein for analyzing the amount of intraabdominal adipose
tissue, wherein the protein is (1) encompassed by the amino acid sequence
shown in SEQ ID NO:1 or is (2) recognized by an antibody against a protein
encompassed by the amino acid sequence shown in SEQ ID NO:1.
14. a function expressing the positive relationship between the amount of
intraabdominal adipose tissue in an animal and the concentration of a
protein in a body fluid, tissue or cell of said test-animal or in a prepared
test
solution from the body fluid, tissue or cell of said test-animal,
wherein the protein is (1) encompassed by the amino acid sequence shown
in SEQ ID NO:1 or is (2) recognized by an antibody against a protein
encompassed by the amino acid sequence shown in SEQ ID NO:1.
15. a kit comprising as a standard for analyzing the amount of
intraabdominal adipose tissue in an animal, a protein encompassed by the
amino acid sequence shown in SEQ ID NO:1 or recognized by an antibody
against a protein encompassed by the amino acid sequence of SEQ ID NO:1.
6
CA 02588614 2007-05-23
16. a kit according to the above 15, further comprising an antibody
recognizing a protein encompassed by the amino acid sequence shown in
SEQ ID NO:1.
Further scope of applicability of the present invention will become
apparent from the detailed description given hereinafter. However, it should
be understood that the detailed description and specific examples, while
indicating preferred embodiments of the invention, are given by way of
illustration only, since various changes and modifications within the spirit
and scope of the invention will become apparent to those skilled in the art
from this detailed description.
Throughout this specification and the claims which follow, unless
the context requires otherwise, the word "comprise", and variations such as
"comprises" and "comprising", will be understood to imply the inclusion of a
stated integer or step or group of integers or steps but not the exclusion of
any other integer or step or group of integer or step.
BRIEF DESCRIPTION OF THE DRAWINGS
Fig. 1 shows the structure of expression plasmid pET11a085 for
expressing in E. coli, a protein consisting of an amino acid sequence starting
at the 27-position in the amino acid sequence shown in SEQ ID NO:1. The
shaded portion in the drawing shows the insert coding for the protein to be
expressed.
Fig. 2 shows the results of detection of a protein of the present
7
CA 02588614 2007-05-23
invention in blood by immunoprecipitation and Western blotting techniques.
In the drawing, the upper arrow indicates the starting point of
electrophoresis and the lower arrow indicates the end of electrophoresis.
The signal of the immunoprecipitated protein was quantified by a
densitometer. Lane 1, 0 ng of antigen protein; lane 2, 0.2 ng of antigen
protein; lane 3, 0.5 ng of antigen protein; lane 4, 1.0 ng of antigen protein;
lane 5, 2.0 ng of antigen protein; lane 6, blood from examinee 1; and lane 7,
blood from examinee 2.
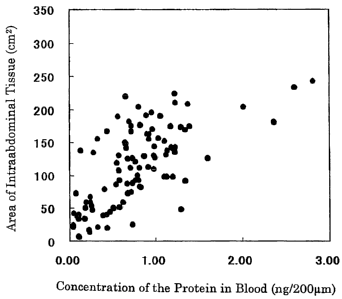
Fig. 3 is a drawing showing the correlation between the
concentration (ng/200 1) of a protein of the present invention in blood (X)
and the area (cm2) of intraabdominal adipose tissue (Y). Each plot is
derived from a different individual.
Fig. 4 shows a change in concentration of a protein of the present
invention in blood from 5 examinees as determined by enzyme
immunoassays before and after a predetermined period, as well as a change
in the area of intraabdominal adipose tissue. Respectively, lane Nos. 1 and
2 indicate the results of examinee 1 before and after the predetermined
period of 28 days; lane Nos. 3 and 4, the results of examinee 2 before and
after the predetermined period of 49 days; lane Nos. 5 and 6, the results of
examinee 3 before and after the predetermined period of 56 days; lane Nos.
7 and 8, the results of examinee 4 before and after the predetermined period
of 63 days; and lane Nos. 9 and 10, the results of examinee 5 before and
after the predetermined period of 17 days.
DETAILED DESCRIPTION OF THE INVENTION
8
CA 02588614 2007-05-23
Hereinafter, the present invention is described in detail.
In the present invention, the "amount of intraabdominal adipose
tissue" refers to the amount of adipose tissue in the abdominal rectus,
external and internal abdominal oblique muscles, abdominal transverse,
quadrate lumbar muscle, psoas major and inner portions of pyramid. As
such, the amount of intraabdominal adipose tissue may employ the total
amount (e.g. volume or weight) of intraabdominal adipose tissue or the area
of intraabdominal adipose tissue in a section of the abdomen, which is
known to be proportional to the total amount of intraabdominal adipose
tissue (Int. J. Obesity, vol. 17, p. 187, 1993). The "area of intraabdominal
adipose tissue in a section of the abdomen" refers to the area occupied by
adipose tissue in the abdomen, that is, in the abdominal rectus, external
and internal abdominal oblique muscle, abdominal transverse, quadrate
lumbar muscle, ps.oas major and pyramid, which is discerned in a taken
picture of the section of the abdomen, obtained in methods described in
"Image Analysis Viewed from Symptoms" (compiled by the Japan Medical
Association), such as computed tomography (referred to hereinafter as CT
scanning) (Visceral Fatty-Type Obesity, 1995, published by lyaku Journal
Ltd.), ultrasound examination and magnetic resonance imaging. The term
"abdomen" or "abdominal" refers to an area which is usually examined for
measuring the area of intraabdominal adipose tissue, and roughly speaking,
this site is typically above the groin and below the midriff forming a
boundary with the 'chest.
The protein used in the analytical method of the present invention
typically is a protein encompassed by the amino acid sequence shown in
9
CA 02588614 2007-05-23
SEQ ID NO:1 or protein recognized by an antibody against said protein
(hereinafter, such proteins are collectively referred to as the protein of the
present invention). More specific examples of the protein of the present
invention include a protein encompassed by the amino acid sequence shown
in SEQ ID NO:1, as well as proteins which are recognized by an antibody
against a protein encompassed by the amino acid sequence shown in SEQ
ID NO:1. The latter protein may be, for example, proteins encompassed by
the amino acid sequence shown in SEQ ID NO:1, wherein one or more
amino acids have been deleted, replaced or added. The amino acid
sequences wherein "amino acids have been deleted, replaced or added"
typically include naturally occurring variations of a protein encompassed by
the amino acid sequence shown in SEQ ID NO:1 e.g. resulting from
intracellular processing or a difference in the type of species, a difference
among individuals or tissues from which said protein was derived.
In order to analyze the amount of intraabdominal adipose tissue in
a test-animal according to the present invention, based on the relationship
between the concentration of the protein of the present invention in a body
fluid, tissue or cell from an animal, preferably an animal which is of the
same species as the test-animal, or in a prepared solution from the body
fluid, tissue or cell of such an animal and the amount of intraabdominal
adipose tissue in such an animal, the amount of intraabdominal adipose
tissue in the test-animal is usually determined from the concentration of
the protein of the present invention in a body fluid, tissue or cell of the
test-
animal or in a prepared test solution from the bodyfluid, tissue or cell of
said test-animal. The body fluids from an animal include e.g. blood, urine,
CA 02588614 2007-05-23
saliva etc.; the tissues therefrom include e.g. intraabdominal adipose tissue
such as mesenteric adipose tissue and omental adipose tissue; and the cells
therefrom include e.g. fat cells present in intraabdominal adipose tissue
such as mesenteric adipose tissue and omental adipose tissue. Among the
above-described body fluids, tissues and cells of an animal, blood may be
mentioned as a preferable example. Further, a control solution or a test
solution may be prepared by subjecting the body fluids, tissues and cells
from an animal to post-treatment such as disruption treatments which use
a crushing device such as a Teflon homogenizer or solid removing
treatments which use a centrifuge, if so desired.
The relationship between the concentration of the protein of the
present invention in body fluids, tissues or cells of an animal or in a
prepared solution from the body fluids, tissues or cells of said animal and
the amount of intraabdominal adipose tissue in said animal may be
determined, for example, as follows: The concentration(s) of the protein of
the present invention in a control solution from a control-animal, preferably
in control solutions from plural individuals of a control-animal of a single
species is determined by e.g. an immunochemical analysis described below.
After the concentration of the protein of the present invention in the control
solution is determined, the amount of intraabdominal adipose tissue in each
of the animals may be determined as e.g. the area of intraabdominal
adipose tissue in a section of the abdomen. For quantifying such an area,
techniques such as CT scanning, ultrasound examination and magnetic
resonance imaging are typically used. More specifically, photographing a
section of the abdomen by CT scanning may be conducted according to a
11
CA 02588614 2007-05-23
method described in "Visceral Fatty-Type Obesity" published in 1995 by
Iyaku Journal Ltd. The area to be subjected to photographing a section of
the abdomen is an area which is ordinarily measured for examining the
amount of intraabdominal adipose tissue and which can provide for an
accurate examination of the amount of the intraabdominal adipose tissue,
and preferably is the area of the navel. When the concentration of the
protein of the present invention in body fluids, tissues or cells of the
control-animals or in prepared solutions from the body fluids, tissues or
cells of the control-animal are plotted on the X-axis and the amounts of
intraabdominal adipose tissue, e.g. the area of intraabdominal adipose
tissue in a section of the abdomen, are plotted on the Y-axis, there appears a
direct relationship or positive correlation between the concentrations of the
protein of the present invention in the control solutions and the amounts of
intaabdominal adipose tissue. The plurality of plots thus obtained are
statistically processed whereby the concentration of the protein of the
present invention in the control solution and the amount of intraabdominal
adipose tissue are expressed as variables which are different from each
other. In this regard, the correlation therebetween, for example, may use
the linear function such as the correlation Y = aX + b. The reliability (i.e.,
coefficient of correlation) of this correlation is improved as the number of
plots is increased. The number of plots may be, more specifically, e.g. 50 to
500 or so, and by way of an example, a coefficient of correlation such as
about 0.7 may be obtained from about 100 plots.
To determine the amount of intraabdominal adipose, tissue in the
test-animal, the concentration of the protein of the present invention in
12
CA 02588614 2007-05-23
body fluids, tissues or cells of said test-animal or in or in a prepared test
solution from the body fluids, tissues or cells of the test-animal is
determined by e.g. a method described below. The concentration thus
determined is substituted for X in the above obtained correlation, e.g. the
correlation Y= aX + b, whereby Y may be calculated as the amount of
intraabdominal adipose tissue in said test-animal.
Hereinafter, the examination method of the present invention is
further described. It has been revealed that excessive accumulation of
intraabdominal adipose tissue is related closely to the onset of diseases
including metabolic disorders such as diabetes, hyperlipemia and
arteriosclerosis as well as cardiac vessel disorders such as coronary artery
diseases, angina and myocardial infarction (Visceral Fat-Type Obesity, 1995,
published by Iyaku Journal Ltd.). Further, since the concentration of the
protein of the present invention in a control solution is in a positive
relationship with the amount of intraabdominal adipose tissue as described
above, the concentration of the protein of the present invention in a control
solution is related closely to the onset of said diseases. Accordingly, (1)
the
increase or decrease of the amount of intraabdominal adipose tissue in the
same test-animal for a predetermined period is examined by use of the
analytical method of the present invention, (2) the increase or 'decrease of
the concentration of the protein of the present invention in a body fluid,
tissue or cell of the same test-animal or in a prepared test solution from the
body fluid, tissue or cell of the test-animal for a predetermined period is
examined, (3) the amount of intraabdominal adipose tissue in a test-animal
is determined by use of the analytical method of the present invention and
13
CA 02588614 2007-05-23
then compared with the amount of intraabdominal adipose tissue in a
healthy animal of the same species of said test-animal or (4) the
concentration of the protein of the present invention in a body fluid, tissue
or cell of a test-animal or in a prepared test solution from the body fluid,
tissue or cell of said test-animal is compared with the concentration of the
protein of the present invention in a body fluid, tissue or cell of a healthy
animal of the same species of said test-animal or a prepared test solution
from the body fluid, tissue or cell of said healthy animal, whereby the risk
of
onset of the diseases related closely to the increase of the amount of
intraabdominal adipose tissue or judging the recovery of a disease in the
test-animal can be predicted.
For example, the amount of intraabdominal adipose tissue in a
test-animal is determined as a previous amount by the analytical method of
the present invention. After a predetermined period, for example, after a
period of 0.5 month or more, the amount of intraabdominal adipose tissue in
the same test-animal is determined and compared with the previous
amount thereof, so that the increase or decrease of the amount of
intraabdominal adipose tissue in the same test-animal may be known.
Further, by having the amount of intraabdominal adipose tissue in the
same test-animal recorded several times, for example, 3 or more times, the
change with time of the amount of intraabdominal adipose tissue in the
same test-animal may also be known. The risk of onset of the diseases
related closely to the increase of the amount of intraabdominal adipose
tissue may thereby be predicted. That is, if the amount of intraabdominal
adipose tissue in the test-animal is increased, the risk of onset of the
14
CA 02588614 2007-05-23
diseases is predicted to be higher, whereas if the amount of intraabdominal
adipose tissue in the test-animal is decreased, the risk of onset of the
diseases is predicted to be lower. Of course, the concentration of the
protein of the present invention in body fluids, tissues or cells of the test-
animal or in prepared test solutions from the body fluids, tissues or cells of
said test-animal may be directly used, in place of the amount of
intraabdominal adipose tissue of the test-animal.
The aniount of intraabdominal adipose tissue in a test-animal, as
determined by use of the analytical method of the present invention, may
also be expressed in terms of the area of intraabdominal adipose tissue in a
section of the abdomen. As such, the risk of onset of the diseases is
predicted
to be high when said area is higher than a standard area regarded as being
indicative of a high risk of onset of the diseases. In addition, the risk of
onset of the diseases is predicted to be low when said area is lower than the
standard area regarded as being indicative of a high risk of onset of the
diseases. The standard area regarded as being indicative of a high risk of
onset of the diseases is varied depending on the species, sex and age of a
test-animal and the type of disease etc., but the preferred standard area for
humans is e.g. about 90 to 130 cm2.
The amount of intraabdominal adipose tissue in a test-animal or
the concentration of the protein of the present invention in body fluids,
tissues or cells of the test-animal or a prepared solution from the body
fluids,
tissues or cells of said test-animal, as determined by use of the analytical
method of the present invention, may also be compared with an average
amount of intraabdominal adipose tissue or an average concentration of the
CA 02588614 2007-05-23
protein of the present invention in test solutions obtained from a population
composed mainly of healthy individuals of a similar species of the test-
animal, preferably an identical species (or an identical human race when
the test-animal is humans), an identical sex and a similar age of the test-
animal. If the resulting value of the test-animal is higher than the
average values, the risk of onset of the diseases is typically higher. If the
value of the test-animal is e.g. twice as high as the average value, the risk
of
onset of the diseases is predicted to be high, and if the value is about
thrice
as high as the average value, the risk of onset of the diseases is predicted
to
be extremely high, although said prediction depends on the type of disease.
The analytical method as described above is very useful for
managing the test-animals to maintain their health.
The protein of the present invention may be used in analyzing the
amount of intraabdominal adipose tissue in a test-animal, that is, an
analysis of the intraabdominal adipose tissue which comprises determining
the amount of intraabdominal adipose tissue in the test-animal from the
concentration of the protein of the present invention in a body fluid, tissue
or cell of the test-animal or in a prepared test solution from the body fluid,
tissue or cell of said test-animal, based on the positive relationship between
the concentration of the protein of the present invention and the amount of
intraabdominal adipose tissue in a control-animal, in order to analyze the
amount of intraabdominal adipose tissue in the test-animal.
The test-animal to which the analytical method of the present
invention and the examination method of the present invention as described
above are applicable includes e.g. mammals, and preferably humans and
16
CA 02588614 2007-05-23
monkeys.
A method of measuring the concentration of the protein of the
present invention in a body fluid, tissue or cell of an animal or in a
prepared
solution from the body fluid, tissue or cell of said animal, or in a body
fluid,
tissue or cell of a test-animal or in a prepared test solution from the body
fluid, tissue or cell of said test-animal may be any method being capable of
specifically identifying said protein, with examples thereof including:
1) an immunochemical analysis using an antibody against a protein
encompassed the amino acid sequence shown in SEQ ID NO:l;
2) a method wherein the supernatant, obtained by centrifugation of a
control solution or a test solution, is applied to liquid chromatography
thereby separating and fractionating proteins contained in the supernatant,
and the protein of the present invention is identified and quantified by mass
spectroscopy;
3) a method wherein a control solution or a test solution is pretreated for
removal of unnecessary proteins such as, albumin and immunoglobulin, and
then subjected to two-dimensional electrophoresis, and the components in
the control are separated and developed two-dimensionally, based on the
difference in the isoelectric points and molecular weights of proteins to
identify and quantify a spot of the protein of the present invention
(Proteome Research: New Frontiers in Functional Genomics, p. 190, 1997,
published by Springer); and
4) a method wherein molecules (DNA, RNA, protein, low-molecular
compound etc.) being capable of specifically recognizing the protein of the
present invention are selected from a molecular library synthesized at
17
CA 02588614 2007-05-23
random, and based on the specificity and affinity of the protein of the
present invention for said molecules, the protein of the present invention is
specifically separated and quantified from a control solution or a test
solution.
Among those methods described above, the immunochemical
analysis is described below in detail as a more specific example.
(1) Preparation of antigen
To prepare an antibody used in the immunochemical analysis, an
antigen is first prepared. As the antigen, a protein encompassed by the
amino acid sequence shown in e.g. SEQ ID NO:1 (referred to hereinafter as
antigen protein) may be used.
The antigen protein may be produced and obtained in a large
amount by conventional genetic engineering techniques (e.g., techniques
described by J. Sambrook, E. F. Frisch, T. Maniatis: Molecular Cloning 2nd
edition, Cold Spring Harbor Laboratory press) by using a gene coding for
the antigen protein, for example, DNA having a nucleotide sequence coding
for the amino acid sequence shown in SEQ ID NO:1, more specifically, a
DNA encompassed by the nucleotide sequence shown in SEQ ID NO:6. In
more detail, a plasmid which is capable of permitting a gene coding for the
antigen protein to be expressed in host cells is prepared and transfected into
host cells, and the resulting transformant is cultured.
Examples of such host cells may include eukaryote and procaryote
microorganisms or aninial cells such as mammalian and insect cells, and
preferably include E. coli.
The plasmid is one which can autonomously multiply and includes
18
CA 02588614 2007-05-23
genetic information for being able to replicate in host cells, and preferable
plasmids also can be easily isolated and purified from host cells, contain a
promoter capable of functioning in host cells and have a gene coding for the
antigen protein introduced into an expression vector carrying a detectable
marker. A wide variety of expression vectors are commercially available,
and for example, expression vectors carrying a promoter such as lac, trp or
tac used for expression in E. coli are commercially available from e.g.
Pharmacia and Takara Shuzo Co., Ltd. Restriction enzymes used for
introducing the gene encoding the antigen protein into the expression vector
are also commercially available from e.g. Takara Shuzo Co., Ltd. Further,
a ribosome-binding region may be ligated to a region upstream from the
gene encoding the antigen protein so that higher expression is brought
about in some cases. The ribosome-binding region is known according to a
report of Guarente, L. et al. (Cell, 20, 543 (1980)) or a report of Taniguchi
et
al. (Genetics of Industrial Microorganisms, p. 202, (1982) published by
Kodansha).
The plasmid thus obtained can be introduced.into the host cells by
usual genetic engineering techniques.
Culturing of the host cells can be carried out by conventional
techniques employed for microbial cultures. For example, the host cells
are cultured in a medium containing suitable carbon and nitrogen sources
as well as trace nutrients such as vitamins. The method for culturing may
perform in either solid or liquid medium, preferably shake culture under
aeration.
From the host cells thus obtained, the antigen protein may be
19
CA 02588614 2007-05-23
prepared by a combination of techniques usually employed in isolation and
purification of general proteins. For example, after culturing, the antigen
protein may be purified by collecting the host cells in a centrifuge, lysing
or
conducting bacteriolysis on the host cells, optionally solubilizing the
antigen
protein and performing a combination of various chromatographic steps
such as ion-exchange, hydrophobic and gel filtration chromatography.
Operations for refolding the structure of the protein may be further
conducted if necessary.
For preparing the antibody used in the immunochemical analysis
described above, an antigen prepared by the following methods may also be
used. Examples of the methods include a method wherein an antigenic
peptide containing a particular partial amino acid sequence of the amino
acid sequence of the protein of the present invention is rendered polymeric,
or a method wherein the antigenic peptide is bound directly or indirectly
with a spacer, to a high-molecular carrier to form a complex. Such
methods are typically those wherein the antigenic peptide which due to a
low molecular weight, is partially antigenic by itself, that is, an incomplete
antigen, and is converted into a complete antigen by increasing its
molecular weight. Hereinafter, the process of converting the antigenic
peptide into a complete antigen is described.
The antigenic peptide may be selected by using e.g. a method for
predicting an epitope in a protein as described in "Experimental Protocols
on Anti-Peptide Antibody" published by Shujunsha. Usually, a peptide
consisting of 10 to 20 amino acids is selected as the antigenic peptide. The
used antigenic peptide containing a particular amino acid sequence used is
CA 02588614 2007-05-23
preferably a peptide of high purity, and its synthesis and purification are
also described in the "Experimental Protocols on Anti-Peptide Antibody"
published by Shujunsha. For example, the antigenic peptide can be
purified beforehand by ordinary techniques such as high performance liquid
chromatography, if necessary.
The method of rendering the antigenic peptide polymeric includes
e.g. the MAP (multiple antigen peptide) method devised by Tam et al. (Proc.
Natl. Acad. Sci. USA, 85, 5409, 1988). According to such a method, a lysine
residue is introduced to the C-terminus of the antigenic peptide during the
synthesis thereof and the peptide is branched sequentially by using the a-
and E-amino groups of the lysine to render the antigenic peptide polymeric
so that the antigenecity of the peptide is increased. Since various kinds of
previously branched and lysine-bound resin for MAP are commercially
available, each of the amino acids in the antigenic peptide containing a
particular partial amino acid sequence of the amino acid sequence of the
antigen protein may be sequentially added to such a resin by using
conventional peptide synthesis methods to elongate its peptide chain.
In the method wherein the antigenic peptide containing a
particular partial amino acid sequence of the amino acid sequence of the
antigen protein is bounci directly or indirectly with a spacer, to a high-
molecular carrier molecule to form a complex, the high-molecular carrier
molecule used for binding with the antigenic peptide may have reactive
groups freely available for a linking reaction with the antigenic peptide
containing a particular amino acid sequence or a compound having a spacer
bound thereto (hereinafter referred to collectively as the incomplete
21
CA 02588614 2007-05-23
, =
antigen), and be capable of conferring immunogenicity thereto or capable of
raising the original immunogenicity of the antigenic peptide by being linked
to said incomplete antigen. The macromolecular compound containing
freely available reactive amino groups is particularly preferable. An
example of such macromolecular compounds include lysine-enriched
proteins having a molecular weight of about 10,000 to 150,000. More
specifically, such macromolecular compounds include bovine serum albumin
(BSA: molecular weight 66200), human serum albumin (HSA: molecular
weight 58000), rabbit serum albumin (RSA: molecular weight 68000), goat
serum albumin (GSA: molecular weight 68000) and keyhole limpet
hemocyanin (KLH: molecular weight > 1000000). Other macromolecular
compounds which can meet the requirements described above may also be
used as carrier molecules, and such compounds include e.g. porcine
thyroglobulin, B2 microglobulin, hemocyanin, immunoglobulins, toxins
(chorea toxin, tetanus toxin, diphtheria toxin etc.), polysaccharides,
liposaccharides, natural occurring or synthetic polyadenylic acid and
polyuridylic acid, polyalanyl and polylysine polypeptides or cellular
membrane components such as formalin- or glutaraldehyde-treated
erythrocyte cellular membranes.
The method for binding the incomplete antigen to the high-
molecular carrier molecule described above may be a method in which the
region of a particular amino acid sequence in the incomplete antigen may
remain freely available so that a specific immune response is inducible, that
is, which may have the production of a specific antibody inducible. More
specifically, for example, it is preferred that (1) an incomplete antigen in
22
CA 02588614 2007-05-23
which the particular amino acid sequence is located, as much as possible, at
the most outermost surface thereof is selected and (2) the site of the
particular amino acid sequence in the selected incomplete antigen is located,
as much as possible, at the outermost surface from the high-molecular
carrier molecule.
If the reactive group in the incomplete antigen is a reactive amino
group, a reactive group of a spacer is bound to one of the reactive amino
groups in the high-molecular carrier molecule by use of e.g. a dialdehyde
such as glutaraldehyde. If the reactive group in the incomplete antigen is
a reactive SH group, the reactive group in the incomplete antigen is bound
by using e.g. oxidative reaction, to one of the reactive SH groups in the
high-molecular carrier molecule. If the reactive group in the incomplete
antigen is one of the reactive carboxyl group, the reactive group in the
incomplete antigen is bound to one of reactive amino groups in the high-
molecular carrier molecule by use of e.g. carbodiimide, preferably 1-ethyl-3-
(3-dimethylaminopropyl)carbodiimide hydrochloride. By way of an
example, an incomplete antigen having a reactive carboxyl group may be
bound to a reactive amino group in the high-molecular carrier molecule by
e.g. an active ester method described by H. Hosoda et al. in Chem. Pharm.
Bull. 31 (11), 4001-4007 (1983) or by a mixed acid anhydride method
described by B. F. Erlanger et al. in J. Biol. Chem., 234, 1090-1094 (1959).
The spacer used for when indirectly linking with the spacer is a
compound containing one or more kinds of reactive groups capable of
forming a covalent bond with a freely available reactive group in the high-
molecular carrier molecule. An example of the spacer is a compound
23
CA 02588614 2007-05-23
containing 2 to 16 cross-linking carbon atoms and having one or more
reactive groups such as an amino group, carboxyl group, maleimide group or
SH group. More specifically, a compound of the general formula
H2N(CH2)nCOOH (n is an integer of 2 to 16) is a preferable example.
Linking of the spacer to the antigenic peptide containing a particular amino
acid sequence may be conducted in a similar manner as the above-described
method for binding a reactive group in the incomplete antigen to one of the
reactive groups in the high-molecular carrier molecule.
(2) Step of immunization and sensitization of mammals and acquisition of
an antibody
The antigen thus obtained is used to immunize mammals such as
mice, hamsters, guinea pigs, chickens, rats, rabbits and dogs according to
the usual immunization and sensitization methods described by e.g. W. H.
Newsome et al. in J. Assoc. Off. Anal. Chem. 70(6), 1025-1027 (1987). The
antigen may be administered once or several times.
The antigen is administered preferably 3 or 4 times at 7- to 30-day
intervals, particularly 12- to 16-day intervals. The dose thereof is, for
example, approximately 0.05 to 2 mg of the antigen for each administration.
The administration route may be selected from subcutaneous
administration, intracutaneous administration, intraabdominal
administration, intravenous administration and intramuscular
administration, and an injection given intravenously, intraabdominally or
subcutaneously is a preferable administration form. In addition, a
combination of the subcutaneous injection and the intraabdominal injection
is particularly preferable. In such cases, the antigen is usually used after
24
CA 02588614 2007-05-23
being dissolved in a suitable buffer, for example, sodium phosphate buffer or
physiological saline containing a kind of ordinarily used adjuvant such as
complete Freund's adjuvant (a mixture of Aracel A, Bayol F and dead
tubercule bacillus), RAS [MPL (monophosphoryl lipid A) + TDM (synthetic
trehalose dicorynomycolate) + CWS (cell wall skeleton) adjuvant system] or
aluminum hydroxide. However, depending on the administration route or
conditions, the adjuvants described above may not be used. As used herein,
the "adjuvant" means a substance which upon administration with the
antigen, enhances a immune reaction unspecifically against the antigen.
After the mammal is left for 0.5 to 4 months without an
administration thereof, a small amount of blood is sampled from e.g. an ear
vein of the mammal and measured for antibody titer. When the antibody
titer is increasing, administration of the antigen is conducted repeatedly an
appropriate number of times, depending on cases. For example, the
antigen may be administered I to 5 times at a dose of 100 g to 1 mg for
each administration. One to two months after the final administration,
blood is collected in a usual manner from the immunized and sensitized
mammal, and by having the blood separated and purified by conventional
techniques such as precipitation by centrifugation or with ammonium
sulfate or polyetliylene glycol and chromatography such as gel filtration
chromatography, ion-exchange chromatography and affinity
chromatography, the antibody used in the present invention may be
obtained as a polyclonal antiserum. Further, the antiserum may be
subjected to treatment e.g. at 56 C for 30 minutes to inactivate the
complement system.
CA 02588614 2007-05-23
Further, immunocompetent B cells are isolated from the
immunized and sensitized mammal described above, and the
immunocompetent cells are fused with tumor cells capable of continuous
cell division, and the formed fusion cells are then isolated. After screening,
hybridoma cells producing the desired antibody are cloned, and the
hybridoma cells are cultured in vitro or in vivo to produce the monoclonal
antibody, whereby an antibody having high degrees of specificity and
affinity may also be produced.
(3) Quantification of the protein of the present invention by the antibody
The method of measuring the concentration of the protein of the
present invention by the antibody thus prepared is described by reference
with the following typical examples.
In accordance with the present invention, as an antibody used in
the following techniques for quantifying the protein of the present invention,
there may be used molecules containing antibody activity such as a
polyclonal or monoclonal antibody, any class and subclass of an antibody, as
well as a Fab and Fab' fragment.
(A) Immunoblotting techniques
In immunoblotting techniques, the protein of the present invention
bound to a solid carrier is recognized by an antibody against said protein
(hereinafter, said antibody is referred to as primary antibody) and the
antibody is detected. The principles and outline of such a method are
described in e.g. Antibodies - A Laboratory Manual, p. 471 (1988, Cold
Spring Harbor Laboratory).
26
CA 02588614 2007-05-23
As the solid carriers, nitrocellulose formed in the shape of a
membrane, sheet, filter etc. is generally used, but the solid carriers are not
particularly limited insofar that the protein of the present invention may
adhere well thereto and the solid carriers do not eliminate the antigenecity
of the protein of the present invention. If a nitrocellulose membrane is
used, a solution obtained by diluting the protein of the present invention at
a suitable concentration with a suitable buffer such as phosphate buffered
saline is spotted onto the nitrocellulose membrane, whereby the protein of
the present invention is bound to the nitrocellulose membrane. The
amount of the spot for quantification thereof is preferably about 1 l/3 mm2
or an amount in which the primary antibody is in excess. To permit the
protein of the present invention to be recognized by the primary antibody, it
is preferable to treat a sample containing the protein of the present
invention with e.g. 0.1 % (w/v) SDS or have 0.1%(w/v) of SDS in the buffer
used when binding to nitrocellulose membrane. Alternatively, a sample
containing the protein of the present invention is diluted with a suitable
buffer such as phosphate buffered saline and electrophoresed in acrylamide
gel of suitable concentration. The protein of the present invention after
electrophoresis is transferred onto a suitable membrane such as Hybond-N
(Amersham) by electroblotting techniques or semi-dry techniques (Bio
Experiment Illustrated 5, p. 105, published by Shujyunsha).
To prevent the antibody from adhering nonspecifically to sites not
containing the protein of the present invention on the nitrocellulose
membrane with the protein of the present invention thus spotted or on the
nitrocellulose membrane to which the protein of the present invention was
27
CA 02588614 2007-05-23
, =
transferred by electrophoresis, the nitrocellulose membrane to which the
protein of the present invention has been bound is incubated for about 20
minutes to 24 hours at room temperature to 37 C with a solution
containing high-molecular carrier molecules not recognized by the primary
antibody, that is, gelatin, skimmed milk or high-molecular carrier molecules
(e.g. serum albumin from a different kind of animal such as goat, cattle)
which among the high-molecular carrier molecules usable in the step of
converting the antigenic peptide containing a particular amino acid
sequence into a complete antigen, are not used in production of the primary
antibody, so that the surface of the nitrocellulose membrane is covered with
the high-molecular carrier molecules. After incubation, the nitrocellulose
membrane is washed to remove the above high-molecular carrier molecules
in a free state. The nitrocellulose membrane thus prepared is mixed with
a prepared solution containing the primary antibody and then incubated for
about 10 minutes to 3 hours at room temperature to 37 C under stirring.
The prepared solution containing the primary antibody refers to a prepared
solution in which the primary antibody can be present in a free form in
distilled water or a solution such as a buffer and saline. In this manner,
the protein of the present invention is recognized by the primary antibody.
Hereinafter, the method of detecting the antibody is described.
If the primary antibody has been labeled with an enzyme such as
peroxidase, alkaline phosphatase, (3-D-galactosidase, glucose oxidase,
glucoamylase, carboxylic anhydrase, acetylcholine esterase, lysozyme,
malate dehydrogenase and glucose-6-phosphate dehydrogenase, the
primary antibody in a free form is removed by washing after incubation, a
28
CA 02588614 2007-05-23
substrate for the above labeled enzyme is allowed to act on the enzyme and
the reaction is measured by use of coloration so that the primary antibody
may be detected. For example, if the primary antibody is labeled with a
peroxidase, a combination of hydrogen peroxide as the substrate and
diaininobenzidine or o-phenylene diamine as the coloring reagent yields
brown or yellow coloration, and thus the concentration of the protein of the
present invention can be determined by quantifying the absorption at a
wavelength corresponding to said coloration. For an alternative method of
detecting a peroxidase-labeled antibody-antigen complex, an ECL detection
system (Clin. Chem. vol. 25, p. 1531, 1979) (Amersham) by which a signal
from the target antigen may be detected on an X-ray film by
chemiluminescence is commercially available. In such a method, the
signal detected on the X-ray film can be quantified with a densitometer. In
the case of the primary antibody labeled with glticose oxidase, e.g. 2,2'-
acido-di-(3-ethylbenzothiazoline-6-sulfonic acid (ABTS) is used. In the
case in which the primary antibody is labeled with biotin, streptoavidin
having affinity for biotin may be used to detect a signal from the antigen by
a similar coloration reaction to that of the primary antibody labeled with an
enzyme.
Further, when an enzyme-labeled secondary antibody recognizing
and binding to the primary antibody is used, the primary antibody in a free
form is removed by washing after incubation, and then the membrane is
incubated with the secondary antibody. Said secondary antibody labeled
with an enzyme, for example, is an antibody against the primary antibody
and is labeled with an enzyme such as peroxidase, alkaline phosphatase, ~i-
29
CA 02588614 2007-05-23
D-galactosidase, glucose oxidase, glucoamylase, carboxylic anhydrase,
acetylcholine esterase, lysozyme, malate dehydrogenase and glucose-6-
phosphate dehydrogenase, or is an antibody against the primary antibody
and is labeled with biotin. More specifically, when a rabbit anti-serum is
used as the primary antibody, the secondary antibody is preferably e.g.
peroxidase-labeled anti-rabbit immunoglobulin (IgG) donkey
immunoglobulin (IgG) or anti-rabbit immunoglobulin (IgG) goat
immunoglobulin (IgG). The anti-rabbit IgG donkey IgG or the anti-rabbit
IgG goat IgG is on the market and easily available. As the method of
detecting these secondary antibodies, there may be mentioned a similar
method of detecting the labeled primary antibody as described above.
Further, 1251 labeled-protein A[Amersham] may also be used as the
secondary antibody. This method makes use of the ability of protein A to
bind to the antibody, so that the signal may be detected on an X-ray film and
quantified by a densitometer.
(B) Separation by immunoprecipitation
In a method of immunoprecipitation, the protein of the present
invention is recognized by the primary antibody, the resulting immune
complex containing the antibody and the protein of the present invention is
purified to separate the protein of the present invention, and techniques
such as gel electrophoresis, enzyme activity measurement and
immunoblotting techniques are utilized for quantification of the protein of
the present invention.
First, a sample containing the protein of the present invention is
mixed with the primary antibody against the protein of the present
CA 02588614 2007-05-23
invention for e.g. about 1 to 24 hours at 4 C under stirring whereby an
immune complex thereof is formed. The mixing ratio of the sample to the
primary antibody is e.g. about 1: 8, which shall however vary, depending on
the amount of the protein of the present invention. Preferably, the sample
is previously treated with 0.1 % (w/v) SDS.
Then, a secondary reagent which is capable of binding specifically
to the primary antibody and separating the primary antibody and the
protein of the present invention bound specifically to the primary antibody
from the solution is added if necessary to the formed immune complex, and
the resulting mixture is incubated whereby a complex containing the
immune complex and the secondary reagent is formed and then recovered.
The secondary reagent includes, for example, antibody-binding proteins
located on bacterial cell walls, such as protein A and protein G, or anti-
immunoglobulin antibodies. If a secondary reagent previously bound to
insoluble carriers is used, the complex containing the immune complex and
the secondary reagent may be recovered very easily by centrifugation and
washing. Alternatively, without substantially using the secondary reagent,
a primary antibody bound directly to insoluble carriers may be added to a
sample solution containing the protein of the present invention, and the
protein of the present invention thus rendered insoluble may be recovered.
The insoluble carriers may have a very wide variety of designs and shapes
depending on the specifically intended purposes of use. For example,
beads, dishes, spheres, plates, small rods, cells, small bottles, small tubes,
fibers and nets may be mentioned as the insoluble carrier. More specific
examples include beads (e.g., Sepharose, Bio-Gel etc.) made of
31
CA 02588614 2007-05-23
polysaccharides such as agarose and microtiter plates made of transparent
plastic materials such as polyvinyl chloride or polystyrene, as well as small
spheres, tubes or rods made of polystyrene and polystyrene latex. For
example, agarose-based beads such as cyanogen bromide-activated
Sepharose and Affi-Gel, cellulose-based beads and polyacrylamide-based
beads are commercially available, and functional groups on the beads
thereof have previously been activated so that the secondary reagent or the
primary antibody may be bound thereto directly by a coupling reaction
(Affinity Chromatography, published by Pharmacia, or Nature, vol. 214, p.
1302, 1967). Further, protein A and protein G bound previously to
agarose-based beads are also commercially available.
Subsequently, the protein of the present invention is released from
the recovered complex by procedures such as heat treatment or elution with
a low-pH buffer. Then, the protein of the present invention in a free form
can be detected and quantified by techniques such as gel electrophoresis,
enzyme activity measurement and immunoblotting techniques.
(C) Enzyme immunoassays
Enzyme immunoassays include e.g. a sandwich method and
competitive assay. In the sandwich method, a sample containing the
protein of the present invention is allowed to react with the primary
antibody bound to solid carriers, free components not bound to the solid
carriers are removed by washing and the amount of the protein of the
present invention which has formed an antigen-antibody complex on the
solid carriers is quantified with a labeled secondary antibody or a labeled
antibody specifically binding to the secondary antibody, whereby the
32
CA 02588614 2007-05-23
concentration of the protein of the present invention in the sample is
determined.
In the competitive assay, an antigen or a primary antibody bound
to solid carriers are reacted such that the antigen is reacted competitively
with a sample containing the protein of the present invention and the
primary antibody and the primary antibody is reacted competitively with
the sample containing the protein of the present invention and the free
competitive antigen. Thereafter, free components not bound to the solid
carriers are removed by washing. The amount of the antibody or
competitive antigen which has formed an antigen-antibody complex on the
solid carriers is quantified through the enzyme molecule labeled with said
antibody or antigen or through the labeled secondary antibody specifically
binding to the primary antibody, whereby the concentration of the protein of
the present invention in the sample is determined.
The principles and detailed procedures of such techniques are
described in "Seikagaku Jikkenho 11 (Biochemical Experimental Method
11) (Zbkyo Kagaku Dojin Ltd.) and Method in Enzymology, vol. 70
(Academic Press).
The sandwich assay is further described by reference with the
following typical example.
The primary antibody may be bound directly to solid carriers or
indirectly thereto with a spacer or a high-molecular carrier molecule not
recognized by the secondary antibody and the labeled antibody specific to
the secondary antibody. As used herein, the "high-molecular carrier
molecule not recognized by the secondary antibody and the labeled antibody
33
CA 02588614 2007-05-23
specific to the secondary antibody" refers to a high-molecular carrier
molecule which among the high-molecular carrier molecules usable in the
step of converting the antigenic peptide containing a particular amino acid
sequence of the antigen to a complete antigen, is not used in production of
the secondary antibody and the labeled antibody specific to the secondary
antibody. Further, in cases wherein the primary antibody is to be bound
with a spacer or a high-molecular carrier not recognized by the secondary
antibody and the labeled antibody specific to the secondary antibody, the
spacer of high-molecular carrier may be bound in a similar manner to that
described in the step of converting the antigenic peptide containing the
particular amino acid sequence to a complete antigen.
The solid carriers used for direct or indirect binding of the primary
antibody include ordinarily used materials such as polystyrene, polyacryl,
polycarbonate, polymethacrylate, TeflonTM, nitrocellulose membrane, filter
paper, dextran, glass, agarose, ferrite and latex (natural rubber). The solid
carriers may assume a very wide variety of designs and shapes depending
on specifically intended purposes of use. For example, dishes, spheres,
plates, small rods, cells, small bottles, small tubes, fibers, nets, gels and
column resins may be mentioned as designs thereof. By way of more
specific examples, microtiter plates made of transparent plastic materials
such as polyvinyl chloride or polystyrene, small spheres, tubes or rods made
of polystyrene and polystyrene latex can be mentioned as the solid carrier.
For binding the primary antibody to the solid carriers directly or
indirectly with a spacer or a high-molecular carrier molecule not recognized
by the secondary antibody and the labeled antibody specific to the
34
CA 02588614 2007-05-23
secondary antibody (referred to hereinafter as coating), a non-covalent and
covalent method can be applied.
For covalent binding, the solid carriers are, for example, previously
activated in a conventional manner with glutaraldehyde or cyanogen
bromide.
The coating solution used therein includes e.g. about 10 mM
phosphate buffer (pH 7.4) containing 140 mM sodium chloride, or PBS (140
mM NaCl, 2.7 mM KCl, 10 mM Na2HPO4, 1.8 mM KH2PO4 (pH 7.4)).
Preferable coating conditions are that the concentration of the primary
antibody in the coating solution is preferably e.g. about 0.05 g/ml tolOO
g/ml and the coating time is from several hours to several days, and
preferably e.g. about 6 hours to 24 hours. The coating temperature thereof
is e.g. 4 to 37G. To avoid non-specific binding, a solution containing 0.1~-
5% bovine serum albumin, gelatin or skim milk is added to the thus
obtained primary antibody bound directly or indirectly to the solid carrier
before a sample containing the protein of the present invention is added to
the thus obtained primary antibody bound directly or indirectly to the solid
carrier.
A sample containing the protein of the present invention is added
to the thus obtained primary antibody bound directly or indirectly to the
solid carrier and then incubated at 4 to 37 C. After being incubated for
about several minutes to several days, preferably 2 hours to overnight, the
solid carrier is washed. Then, a'solution containing the secondary
antibody is added thereto and incubated at 4 to 37 C for about 10 minutes
to overnight. If the secondary antibody has been labeled with biotin or an
CA 02588614 2007-05-23
enzyme such as peroxidase, alkaline phosphatase, (3-D-galactosidase,
glucose oxidase, glucoamylase, carboxylic anhydrase, acetylcholine esterase,
lysozyme, malate dehydrogenase or glucose-6-phosphate dehydrogenase,
then the amount of the secondary antibody bound to the solid carrier may
be directly measured by the method described above in the immunoblotting
techniques.
If an enzyme- or biotin-labeled antibody which can recognize the
secondary antibody and specifically bind thereto is used, then the free
secondary antibody is removed by washing, and the solid carrier is
incubated with a solution of said labeled antibody at 4 to 37 C. After
incubation of about 10 minutes to overnight, the solid carrier is washed and
the amount of the labeled antibody bound to the solid carrier after washing
is determined. Said labeled antibody includes an antibody labeled with e.g.
biotin or an enzyme such as peroxidase, alkaline phosphatase, (3-D-
galactosidase, glucose oxidase, glucoamylase, carboxylic anhydrase,
acetylcholine esterase, lysozyme, malate dehydrogenase or glucose-6-
phosphate dehydrogenase.
In cases of using the labeled antibody specific to the secondary
antibody, the primary and secondary antibody are usually of a different
class or derived from a different animal species to substantially avoid the
binding of labeled antibody specific to the secondary antibody to the
primary antibody bound to the solid carrier. For example, when a mouse
monoclonal antibody is used as the primary antibody bound to the solid
carrier, a rabbit polyclonal antibody can be used as the secondary antibody
and a peroxidase-labeled donkey or goat anti-rabbit IgG can be used as the
36
CA 02588614 2007-05-23
labeled antibody specific to the secondary antibody.
In the method described above, a calibration curve is previously
prepared using various diluted solutions containing the protein of the
present invention at known concentrations. Then, a sample containing the
protein of the present invention at a concentration not known is measured,
and the concentration of the protein of the present invention in the sample
is determined, based on the calibration curve.
(D) Radioimmunoassays
The basic principles of the radioimmuno assays are substantially
the same as in enzyme immunoassays. By way of an example, a sample
containing the protein of the present invention is added to a reaction
solution containing known amounts of a labeled antigen and an antibody,
such that there is a competitive reaction. Then, the produced antigen-
antibody complex and the antigen in a free form are separated from each
other, and either of them is quantified and compared with a calibration
curve whereby the concentration of the protein of the present invention in
the -sample solution is quantified (New Lecture on Experiments in
Biochemical Chemistry 12, published by Tokyo Kagaku Dojin, and Method
in Enzymology, vol. 70, published by Academic Press).
Usually, the antigen used in radioimmunoassays are labeled with
125I, and the introduction of 1251 to the antigenic protein may be conducted
according to the method of Bolton-Hunter (Biochem. J., vol. 133, p. 529,
1973) or the chloramine T method.
The measurement is usually conducted by using a gamma counter,
and in a similar manner as in enzyme immunoassays, a calibration curve is
37
CA 02588614 2007-05-23
previously prepared using solutions containing the antigen at known
concentrations. Then, a sample containing the protein of the present
invention at a concentration not known is measured, and the concentration
of the protein of the present invention in =the sample may be determined,
based on the calibration curve.
Hereinafter, a kit of the present invention is described. The kit for
carrying out the analytical method of the present invention and the
examination method of the present invention may be prepared. The kit
may comprise the protein of the present invention as a standard reagent for
analyzing the amount of intraabdominal adipose tissue in an animal, and
preferably, the kit when used for carrying out the analytical method of the
present invention by using immunochemical analysis also comprises an
antibody against the protein of the present invention.
Further, the kit of the present invention may comprise the
following constitutional ingredients. That is, the constitutional
ingredients which may be contained for the sandwich method in enzyme
immunoassays typically include (1) a solid carrier, (2) a reagent containing
an antibody against the protein of the present invention, that is, the
primary antibody, (3) a reagent containing the secondary antibody and (4) a
reagent containing a labeled antibody against the secondary antibody, that
is, a reagent containing an antibody which is labeled with an enzyme and
which can recognize the secondary antibody and can specifically bind
thereto. Additionally, as needed, (5) a substrate compound for the enzyme
used in labeling the antibody, (6) a standard reagent containing a protein
encompassed by the amino acid sequence shown in SEQ ID NO:1 or a
38
CA 02588614 2007-05-23
protein which can be recognized by an antibody against said protein, (7) a
buffer, (8) additives such as a detergent and high-molecular carriers for
preventing non-specific adhering and formation of aggregates and (9) a
pipette, a reaction vessel or a calibration curve may also be included
therein.
The solid carrier in (1) may have the primary antibody in (2)
previously bound thereto directly or indirectly with a spacer or a high-
molecular carrier molecule not recognized by the secondary antibody and
the labeled antibody specific to the secondary antibody.
The reagent containing the primary antibody in (2), the secondary.
antibody in (3) or the labeled antibody in (4) may be provided in the form of
a solution in a buffer or water, or may be provided as a lyophilized product
to be dissolved just before use. Further, (2), (3) and (4) may contain a
stabilizer such as bovine serum albumin at a final concentration of about
0.1 to 10 %(W/V) when dissolved, and if so desired, a detergent such as
Tween 20 may also be contained at a final concentration of 0.1 to 2%(W/V)
when dissolved..
The buffer in (7) may be a buffer which may be used to dilute a
body fluid, tissue or cell of a control-animal or in a prepared solution from
the body fluid, tissue or cell of the control animal or a body fluid, tissue
or
cell of a test-animal or in a prepared test solution from the body fluid,
tissue
or cell of the test-animal, as well as which may be used to wash the solid
carriers and to dissolve and dilute the ingredients in (2), (3), (4) and (5)
above.
Further, the analytical method of the present invention and the
39
CA 02588614 2007-05-23
examination method of the present invention may also be conducted by use
of a combination of devices. By way of an example, in the analytical
method of the present invention and the examination method of the present
invention using immunochemical analysis for measurement of the
concentration of the proteiil of the present invention, there are used devices
produced to include (1) a thermostatic chamber for incubating a body fluid,
tissue or cell of a control-animal or a prepared solution from the body fluid,
tissue or cell of said control-animal or a body fluid, tissue or cell of a
test-
animal or in a prepared solution from the body fluid, tissue or cell of said
test-animal, together with the antibody-containing reagent, at a
temperature in which the protein of the present invention in the control
solution or in the test solution can react with the antibody, (2) a detector
selected from a densitometer for detecting a signal in immunoblotting
techniques or immunoprecipitation, a photometer for measuring the
absorbance or fluorescence of a reaction solution in enzyme immunoassays
and a y-counter used in radioimmunoassays, (3) computer software for
calculating the amount of intraabdominal adipose tissue in a test-animal
from the measurement of the concentration of the protein of the present
invention in the test solution obtained in (2), * based on the positive
relationship between the concentration of the protein of the present
invention in the control solution and the amount of intraabdominal adipose
tissue, and (4) a computer for executing calculation in (3).
EXAMPLES
Hereinafter, the present, invention is described in more detail by
CA 02588614 2007-05-23
reference to the Examples, which however are not intended to limit the
present invention.
Example 1. Isolation of a gene coding for a protein encompassed by the
amino acid sequence shown in SEQ ID NO:1
1.0 g of whole RNA prepared from human intraabdoininal adipose
tissue by the guanidine thiocyanate (GTC)/cesium chloride (CsCI) method
(Chirgwin, J. M. et al., Biochemistry, 18, 5294, 1979) was used as a
template and was mixed with oligo dT primers included in a cDNA
synthesis kit (Takara Shuzo Co., Ltd.). 50 U of MMTV reverse
transcriptase was added thereto in the presence of 1 mM dNTP, and then
the mixture was incubated at room temperature for 10 minutes, and then at
42 C for 15 minutes and at 99 C for 5 minutes whereby single-stranded
cDNA was synthesized.
Then, 55 cycles of PCR, each of the cycles including an incubation
at 94 C for 1 minute, 55 C for 1 minute and 72 C for 2 minutes, were
conducted in which 2.0 ng of said single-stranded cDNA were used as a
template and 20 pmol of each of the oligonucleotides encompassed by the
nucleotide sequences shown in SEQ ID NO: 2 or 3 were used as primers in
the presence of 200 M dNTP, 1.5 mM MgC12 and 1 U of DNA polymerase
(Perkin Elmer). The resulting PCR reaction product was subjected to 1 %
agarose gel electrophoresis (electrophoresis buffer; Tris-boric acid buffer
(Nakalai Tesque), and an about 1.5 kbp DNA band was recovered from the
gel and cloned into a HincII site in plasmid vector pUC118 (Takara Shuzo
Co.) Ltd.) by the method described by J. Sambrook, E. F. Fritsch and T.
"Maniatis in "Molecular Cloning Second Edition", Cold Spring Harbor
41
CA 02588614 2007-05-23
Laboratory Press (1989). The DNA thus cloned was sequenced with a
373A DNA Sequencer manufactured by Applied Biosystems, in which Taq
Dye Primer Cycle Sequencing Kit and Taq Dye Deoxy Terminator Cycle
Sequencing Kit (Applied Biosystems) were also used. The DNA comprised
the nucleotide sequence shown in SEQ ID NO:6, and said nucleotide
sequence coded the amino acid sequence shown in SEQ ID NO:1.
Example 2. Preparation of protein standard (I) encompassing a partial
sequence of the amino acid sequence shown in SEQ ID NO:1
30 Cycles of PCR, each of the cycles including an incubation at 94
C for 1 minute, 60 C for 1 minute and 72 C for 2 minutes were conducted,
wherein the DNA cloned in Example 1 was used as a template and
oligonucleotides encompassed by the nucleotide sequences shown in SEQ ID
NO: 4 or 5 were used as primers. As such, a DNA consisting of a nucleotide
sequence between the 96- and 1493-positions in the nucleotide sequence
shown in SEQ ID NO:6 was amplified. The amplified DNA was digested
with Ndel and BamHI and subcloned to the NdeI and BamHI sites in
.expression vector pET11a (Novagen) to give expression plasmid pET11a085
(Fig. 1) for expressing a protein having Met added to the amino-terminus of
an amino acid sequence starting at amino acid 27 in the amino acid
sequence shown in SEQ ID NO:1.
Then, the expression plasmid pET11a085 was transformed into E.
coli DE3 (Novagen).
The resulting transformant was cultured at 37 C until the O.D.
600 of the culture reached 0.6, and IPTG was added as an inducer to a final.
42
CA 02588614 2007-05-23
concentration of 1 mM, and the transformant was further cultured
overnight. Then, the transformant was harvested by centrifugation, then
suspended in 100 mM Tris-HCl buffer (pH 7.6) containing 5 mM EDTA-2Na,
mM DTT and 1 mM PMSF (referred to hereinafter as buffer A) and
disrupted by soilication (5 minutesx3 times, under cooling on ice), and this
disrupted solution was centrifuged at 12,000xg for 15 minutes at 4 C
whereby the precipitate was recovered as an inclusion body fraction.
The inclusion body fraction was suspended in buffer A to which
urea had been added to a final concentration of 2 M, followed by sonication
(5 minutes, under cooling on ice). The resulting suspension was
centrifuged at 12,000xg for 15 minutes at 4 C, and buffer A to which urea
had been added to a final concentration of 4 M was added to the resulting
precipitate, and the procedure described above was repeated. Further, a
similar procedure was repeated using buffer A to which urea had been
added to a final concentration of 6 M, and the resulting precipitate was
suspended in 20 mM Tris-HCl buffer (pH 8.5) containing 2 mM DTT and 8
M urea and were then centrifuged at 2,000xg for 15 minutes at 4 C, and the
supernatant was recovered. The. resulting supernatant was subjected to
gel filtration chromatography on Hi Load Superdex 200 pg (column, ~16
mmx60 cm (Pharmacia); flow rate, 1.0 ml/min.; detection, 280 nm). A peak
fraction eluted at a retention time of from 45 to 55 minutes was collected
and concentrated with CentriconTM (fractionation molecular weight of
30,000, a product of Grace Japan (formerly Amicon Ltd.)) and then
subjected to Mono Q HR10/10 ion-exchange chromatography (column, ~10
mmx10 cm (Pharmacia); flow rate, 1.0 ml/min.; 1 M NaCl gradient;
43
CA 02588614 2007-05-23
detection, 280 nm). A fraction eluted with 100 to about 200 mM NaCI was
collected and concentrated to 1 mg/ml protein with CentriconTM
(fractionation molecular weight of 30,000, a product of Grace Japan). The
fraction obtained in the procedure described above were analyzed by SDS-
polyacrylamide gel electrophoresis and subsequent silver staining. As a
result, a single band was detected.
100 mM Tris-HC1 (pH 8.5) was added to the thus obtained fraction
under gentle stirring until the final concentration of urea therein was
reduced to 6 M. Gentle stirring was further continued at room
temperature overnight. Then, said fraction was centrifuged at 18,000xg
for 20 minutes at 4 C, the supernatant was then recovered, and 20 mM
Tris-HC1 buffer (pH 8.5) containing 2 M urea, 4 mM reduced glutathione
and 0.4 mM oxidized glutathione was added thereto until the final
concentration of urea therein was reduced to 2.5 M. This solution was
introduced into a dialysis tube with a cut-off molecular weight of 25,000 and
dialyzed at 4 C for about 8 hours against a 1000-fold excess volume of 20
mM Tris-HCl buffer (pH 8.5) containing 2 mM urea, 4 mM reduced
glutathione and 0.4 mM oxidized glutathione. The dialyzate was further
dialyzed at 4 C overnight against a 1000-fold excess volume of 20 mM
Tris-HCl buffer (pH 8.5) containing 2 mM reduced glutathione and 0.2 mM
oxidized glutathione and then dialyzed at 4 C overnight against a 1000-fold
excess volume of 20 mM Tris-HCl buffer (pH 8.5). An aliquot of the
fraction obtained in the procedure described above was analyzed by reverse
phase chromatography, and as a result, a single peak was detected, The
resulting protein was designated protein standard (I).
44
CA 02588614 2007-05-23
Example 3. Preparation of an antibody against a protein standard (I)
(encompassed by the amino acid sequence shown in SEQ ID NO:1)
The protein standard (I) prepared in the method of Example 2 was
used as an antigen to immunize rabbits, and an antibody was obtained from
the animals.
In the first immunization, 1.0 mg of the protein standard prepared
in the method described above was mixed with Freund's adjuvant and
administered subcutaneously into each rabbit. Thereafter, the antigen
was administered, respectively, 4 times at 2 week intervals in a similar
manner. One week after the final administration of the antigen, a small
amount of serum was obtained from an ear vein of each of the rabbits and
measured for antibody titer. Thereafter, the antigen was additionally
administered once and blood was collected from each of the rabbits. The
blood was centrifuged to give a serum fraction which was then dialyzed
overnight against a 100-fold excess volume of 50 mM sodium phosphate
buffer (pH 7.0) at 4 C. The dialyzed serum was subjected to protein A
(Pharmacia) column chromatography, and the adsorbed fraction was eluted
with 100 mM citrate buffer (pH 4.0), and immediately the eluate was
neutralized with 1 M Tris-HCl (pH 9.0), so that an antibody solution was
obtained.
Example 4. Immobilization of the antibody onto carriers
The antibody obtained in the method of Example 3 was bound to
activated cyanogen bromide-Sepharose beads according to a method
CA 02588614 2007-05-23
described in Affinity Chromatography (published by Pharmacia). Specific
procedures_ are as follows: The antibody was diluted with a buffer
comprising 150 mM NaHCO$ and 500 mM NaCl (pH 8.3) (referred to
hereinafter as buffer B) to amount to 2.5 mg protein/ml, then applied to a
PD-10 column (Pharmacia) and eluted with buffer B. 0.5 g of activated
cyanogen bromide-Sepharose beads (Pharmacia) were swollen with 1 mM
HCl previously cooled on ice and then gelled by equilibrating the beads with
200 ml of buffer B containing 1mM HC1. Then, 1.75 ml of the above eluate
was added to and mixed with the gel, and the suspension was stirred, at 4 C
overnight. This mixed suspension was filtered on a Buchner funnel in
which the filtrate was removed under suction. The remaining beads were
suspended in 3.0 ml of 1 M ethanolamine containing 500 mM NaCl (pH 8.3)
and stirred at 4 C for 6 hours to block the remaining active groups. Then,
the beads were washed with 3.0 ml of buffer B with suction on a Buchner
funnel and then with 3.0 ml of a buffer containing 100 mM acetic acid and
500 mM NaC1(pH 4.0) (referred to hereinafter as buffer C). The operation
of alternatingly washing with buffer B and then with buffer C was repeated
times, and the beads were washed several times with a lysis buffer (50
mM Tris-HC1 (pH 7.6), 150 mM NaCl, 1 % NP-40, 0.02 % NasN), suspended
in the lysis buffer at a 1: 1 ratio of the gel slurry to the buffer and then
stored at 4 C. The antibody in the filtrate after immobilization was not
detected by quantification, confirming that the antibody was completely
immobilized onto the beads.
Example 5. Measurement of the concentration of the protein of the present
46
CA 02588614 2007-05-23
invention in a sample solution by immunoprecipitation and Western
blotting techniques
Serum fractions were prepared from human blood samples by
centrifugation (3,000xg, 10 min., 4 C) and were then used as test solutions.
l of the antibody-immobilized beads prepared in Example 4 were added
to 200 l of the test solutions, respectively, and were gently stirred at 4 C
for 4 hours. The beads were precipitated by centrifugation at 12,000xg for
5 seconds at 4 C to remove the supernatant, and 500 l lysis buffer was
added, respectively, to the beads which were then gently stirred and then
centrifuged at 12,000xg for 5 seconds at 4 C to remove the supernatant.
The procedure above of washing the beads was repeated 4 times, and then
500 l distilled water was added, repectively, to the beads, and a similar
washing procedure as above was repeated 3 times. After centrifugation,
the beads were essentially deprived of liquid, and 75 ~t1 of 2 % aqueous
acetic acid was added, respectively, to the beads which were then gently
stirred. The beads were precipitated by centrifugation at 12,000xg for 5
seconds at 4 C, and the supernatant was recovered, and the same
extraction procedure was conducted by adding 2 % acetic acid again to the
beads. The resulting eluates having 2 % acetic acid were collected, and
bovine serum albumin protein was added to said eluates as a carrier and to
a final concentration of 150 jig/ml. Subsequently, 75 l of a TCA solution
was further added, respectively, thereto and the mixtures were left on ice
overnight. Such samples were centrifuged at 15,000xg for 20 minutes at 4
C, and the precipitated protein was recovered, washed with 500 l ice-cold
acetone, respectively, and dried.
47
CA 02588614 2007-05-23
The resulting precipitates were dissolved in a solution containing
50 mM Tris and 0.04 N aqueous NaOH. After an equal volume of SDS
sample buffer was added thereto, the dissolved protein (the protein of the
present invention) was subjected to a 10 to 20% SDS-PAGE gel (Bio-Craft
Ltd.). The protein in the gel after electrophoresis was transferred onto a
Hybond-N membrane (Amersham) by electroblotting techniques (transfer
buffer; 25 mM Tris, 192 mM glycine, 20 % methanol, 4 C, 80 V, 1.5 hours).
The membrane thus prepared was washed with TTBS buffer (20 mM Tris-
HCl (pH 7.4), 150 mM NaCI, 0.05 % Tween 20, 0.05 % Na3N) and then
incubated in TTBS buffer containing 3 % gelatin at 37 C for 1 hour.
Thereafter, the membrane was incubated at 37 C for 1 hour in a solution
containing a 1,000-fold dilution of the antibody obtained in Example 3,
TTBS buffer and 1% bovine serum albumin. Then, the membrane was
washed 3 times with TTBS buffer at room temperature for 5 minutes and
then incubated at 37 C for 1 hour in a solution prepared by 1000-fold
dilution of donkey anti-rabbit IgG antibody labeled with horseradish
peroxidase, with TTBS containing 1 %bovine serum albumin. Thereafter,
the membrane was washed 3 times with TTBS buffer at room temperature
for 5 minutes and subjected to ECL detection system (Amersham). The
menbrane was exposed to a Hyperfirm ECL film _(Amersham), and a
fluorescent signal (in the vicinity of 50 Kda) on the Hyperfirm ECL film
(Amersham) was detected (Fig. 2).
The intensity of the fluorescent signal thus obtained was
quantified with a densitometer. The concentration of the protein of the
present invention in each of the test solutions was calculated from the
48
CA 02588614 2007-05-23
signal intensity of each of the samples, based on a calibration curve
previously prepared using 0.1 to 5 ng of the protein standard (I) obtained in
Example 2.
Example 6. Measurement of the concentration of the protein of the present
invention in a test solution by enzyme immunoassays
A coating solution was prepared by adding the antibody obtained in
Example 3 to PBS (140 mM NaCl, 2.7 mM KCl, 10 mM Na2HP04 and 1.8
mM KH2PO4 (pH 7.4)) to a concentration of 20 g/ml therein. The coating
solution was put into wells of a 96-well polystyrene microtiter plate at a
volume of 150 l/well and then incubated at 4 C overnight. After the
coating solution was removed, each of the wells was washed twice with 300
l of PBS, and the microtiter plate was turned upside down and gently
tapped against a paper towel to remove the solution in the wells of the plate.
Then, PBS containing 1%(W/V) bovine serum albumin was added to the
wells of the microtiter plate at a volume of 150 l/well and incubated at 4 C
overnight for blocking.
After the blocking solution was removed, the microtiter plate was
washed twice with 300 l of a washing solution (50 mM Na2PO4 - Na2HPO4,
150 mM NaCl, pH 7.4, 2.0 % Tween 20). Subsequently, serum fractions
were prepared from human blood samples by centrifugation (3,000xg, 10
minutes, 4 C) and were then used as test solutions. 120 l of a reaction
solution prepared by adding 1%(W/V) bovine serum albumin to the
washing solution was added to each of the wells, and then 30 [t1 of the test
solutions were added to each of the wells, repectively, and incubated at room
49
CA 02588614 2007-05-23
temperature overnight. Theri, the reaction solution was removed, and
each of the wells was washed 3 times with 300 l of the washing solution.
Subsequently, an antibody Fab' fragment obtained by digesting the
antibody obtained in Example 3 with pepsin and the subsequent reduction
thereof, was labeled with peroxidase according to the maleimide hinge
method (described in Enzyme Immunoassays, 3rd edition, published by
Igakushoin). 150 l of an antibody reaction solution (50 mM Na2PO4 -
Na2HPO4, 150 mM NaC1, pH 7.4, 2.0 % Tween 20, 1 % (w/v) normal rabbit
serum, 0.067 % (w/v) 4-aminoantipyrine) containing the resulting
peroxidase-labeled antibody Fab' fragment at a concentration of 5 g/ml
was added to each of the wells and incubated at 4 C for 2 hours. After the
resulting antibody reaction solution was removed, each of the wells was
washed 4 times with 300 l of washing solution.
Then, 50 mM phosphate-25 mM citrate buffer (pH 4.8) containing
1.0 mg/ml o-phenylene diamine and 0.017 %(V/V) hydrogen peroxide was
prepared just before use, and said buffer was added to the wells of the
microtiter plate at a volume of 150 l/well. Then, the microtiter plate was
covered with aluminum foil and iricubated at room temperature for 30
minutes. Thereafter, the reaction was terminated by adding 50 l of 2 N
sulfuric acid, and the coloration on the microtiter plate was measured for
the difference of absorbance between the wavelengths of 492 nm and 595
nm in a multi-scanning spectrophotometer (Bio-Rad).
Diluted solutions of protein standard (I) prepared at concentrations
of 0 to 100 ng/ml were used to prepare a calibration curve for the difference
in absorbance for protein standard (I), and the concentration of the protein
CA 02588614 2007-05-23
of the present invention in each of the test solutions was determined, based
on the calibration curve.
Example 7. Confirmation (1) of the correlation between the concentration
of the protein of the present invention and the amount of intraabdominal
adipose tissue
200 l of serum fractions were prepared by centrifuging (3,000xg,
minutes, 4 C) blood samples from 100 persons and were used as control
solutions. The concentration of the protein of the present invention in each
of the resulting control solutions was determined by the method described
in Example 5.
The area of intraabdominal adipose tissue (cmz) determined for
each of the 100 persons by computed tomography of a section of the
abdomen using CT scanning was plotted on the Y-axis and the
concentration of the protein of the present invention (ng/200 l) in each of
the control solutions on the X-axis (Fig. 3). From the plots thereof, the
coefficient of correlation between X and Y was determined to be about 0.7,
and the correlation between X and Y, that is, Y = 78.8X + 51.4, was thus
obtained.
Example 8. Confirmation (2) of the correlation between the concentration
of the protein of the present invention and the amount of intraabdominal
adipose tissue
The concentration of the protein of the present invention in each
blood sample from 5 examinees was measured before and after a
51
CA 02588614 2007-05-23
predetermined period of time by the enzyme immunoassay described in
Example 6. The changes in the concentration of the protein of the present
invention before and after the predetermined period of time agreed with the
changes in the area of intraabdominal fat determined, respectively, by
tomography of a section of the abdomen by CT scanning before and after the
predetermined period of time. That is, an examinee showing a reduction in
the area of intraabdominal fat also showed a reduction in the concentration
of the protein of the present invention in blood and an examinee showing an
increase in the area of intraabdoniinal fat also showed an increase in the
concentration of the protein of the present invention (Fig. 4).
Example 9. Measurement of the protein of the present invention in blood
from patients with diseases related closely to an increase in intraabdominal
adipose tissue
The concentrations of the protein of the present invention in each
blood sample collected from 38 patients with diabetes and 14 patients with
coronary artery diseases were determined according to the method
described in Example 5. The results are shown in Table 1.
Table 1. Concentration of the protein of the present invention in blood
Blood donors Concentration of the protein
of the present invention in
blood (average; ng/ml)
Patients with diabetes 14.7
Patients with coronary artery diseases 9.8
As illustrated hereinbefore, a method for analyzing the amount of
intraabdominal adipose tissue, which is easily and rapidly feasible with
52
CA 02588614 2007-05-23
satisfactory accuracy, can be provided according to the present invention.
53
CA 02588614 2007-05-23
SEQUENCE LISTING FREE TEXT
SEQ ID NO:2 shows an oligonucleotide primer designed to amplify
the antigen gene.
SEQ ID NO:3 shows an oligonucleotide primer designed to amplify
the antigen gene.
SEQ ID NO:4 shows an oligonucleotide primer designed to amplify
the antigen gene.
SEQ ID NO:5 shows an oligonucleotide primer designed to amplify
the antigen gene.
54
CA 02588614 2007-05-23
SEQUENCE LISTING
<110> Sumitomo Chemical Co., Ltd.
<120> A method for analyzing the amount of intraabdominal adipose tissue
<130> 557496
<140> PCT/JPOO/02210
<141> 2000-04-06
<150> JP 11/103858
<151> 1999-04-12
<160> 6
<210> 1
<211> 491
<212> PRT
<213> Homo sapiens
<400> 1
Met Asn Pro Ala Ala Glu Ala Glu Phe Asn Ile Leu Leu Ala Thr Asp
1 5 10 15
Ser Tyr Lys Val Thr His Tyr Lys G1n Tyr Pro Pro Asn Thr Ser Lys
20 25 30
Val Tyr Ser Tyr Phe Glu Cys Arg Glu Lys Lys Thr Glu Asn Ser Lys
35 40 45
Leu Arg Lys Val Lys Tyr Glu Glu Thr Val Phe Tyr Gly Leu Gin Tyr
50 55 60
Ile Leu Asn Lys Tyr Leu Lys Gly Lys Val Val Thr Lys Glu Lys Ile
65 70 75 80
Gln Glu Ala Lys Asp Val Tyr Lys Glu His Phe Gln Asp Asp Val Phe
85 90 95
Asn Glu Lys Gly Trp Asn Tyr Ile Leu Glu Lys Tyr Asp Gly His Leu
100 105 110
Pro Ile Glu Ile Lys Ala Val Pro Glu Gly Phe Val Ile Pro Arg Gly
115 120 125
Asn Val Leu Phe Thr Val Glu Asn Thr Asp Pro Glu Cys Tyr Trp Leu
130 135 140
Thr Asn Trp Ile Glu Thr Ile Leu Val Gln Ser Trp Tyr Pro Ile Thr
145 150 155 160
Val Ala Thr Asn Ser Arg Glu Gln Lys Lys Ile Leu Ala Lys Tyr Leu
165 170 175
Leu Glu Thr Ser Gly Asn Leu Asp Gly Leu Glu Tyr Lys Leu His Asp
180 185 190
Phe Gly Tyr Arg Gly Val Ser Ser Gln Glu Thr Ala Gly Ile Gly Ala
195 200 205
Ser Ala His Leu Val Asn Phe L,ys Gly Thr Asp Thr Val Ala Gly Leu
210 215 220
Ala Leu Ile Lys Lys Tyr Tyr Gly Thr Lys Asp Pro Val Pro Gly Tyr
225 230 235 240
Ser Val Pro Ala Ala Glu His Ser Thr Ile Thr Ala Trp Gly Lys Asp
245 250 255
His Glu Lys Asp Ala Phe Glu His Ile Val Thr Gln Phe Ser Ser Val
260 265 270
Pro Val Ser Val Val Ser Asp Ser Tyr Asp Ile Tyr Asn Ala Cys Glu
275 280 285
CA 02588614 2007-05-23
Lys Ile Trp Gly Glu Asp Leu Arg His Leu Ile Val Ser Arg Ser Thr
290 295 300
Gln Ala Pro Leu Ile Ile Arg Pro Asp Ser Gly Asn Pro Leu Asp Thr
305 310 315 320
Val Leu Lys Val Leu Glu Ile Leu Gly Lys Lys Phe Pro Val Thr Glu
325 330 335
Asn Ser Lys Gly Tyr Lys Leu Leu Pro Pro Tyr Leu Arg Val Ile Gln
340 345 350
Gly Asp Gly Val Asp Ile Asn Thr Leu Gln Glu Ile Val Glu Gly Met
355 360 365
Lys Gln Lys Met Trp Ser Ile Glu Asn Ile Ala Phe Gly Ser Gly Gly
370 375 380
Gly Leu Leu Gln Lys Leu Thr Arg Asp Leu Leu Asn Cys Ser Phe Lys
385 390 395 400
Cys Ser Tyr Val Val Thr Asn Gly Leu Gly Ile Asn Val Phe Lys Asp
405 410 415
Pro Val Ala Asp Pro Asn Lys Arg Ser Lys Lys Gly Arg Leu Ser Leu
420 425 430
His Arg Thr Pro Ala Gly Asn Phe Val Thr Leu Glu Glu Gly Lys Gly
435 440 445
Asp. Leu Glu Glu Tyr Gly Gln Asp Leu Leu His Thr Val Phe Lys Asn
450 455 460
Gly Lys Val Thr Lys Ser Tyr Ser Phe Asp Glu Ile Arg Lys Asn Ala
465 470 475 480
Gln Leu Asn Ile Glu Leu Glu Ala Ala His His
485 490
<210> 2
<211> 24
<212> DNA
<213> Artificial Sequence
<220>
<223> Designed oligonucleotide primer to amplify antigen gene
<400> 2
ctgtcctccg gcccgagatg aatc 24
<210> 3
<211> 30
<212> DNA
<213> Artificial Sequence
<220>
<223> Designed oligonucleotide primer to amplify antigen gene
<400> 3
cacaacacac acccagtcat aaagcctaat 30
<210> 4
<211> 27
<212> DNA
<213> Artificial Sequence
<220>
<223> Designed oligonucleotide primer to amplify antigen gene
<400> 4
tataaacata tgccacccaa cacaagc 27
56
CA 02588614 2007-05-23
. ~' <210> 5
<211> 26
<212> DNA
<213> Artificial Sequence
<220>
<223> Designed oligonucleotide primer to amplify antigen gene
<400> 5
cagtcaggat ccctaatgat gtgctg 26
<210> 6
<211> 1518
<212> DNA
<213> Homo sapiens
<220>
<221> CDS
<222> (18)..(1493)
<400> 6
ctgtcctccg qcccgag atg aat cct gcg gca gaa gcc gag ttc aac atc 50
Met Asn Pro Ala Ala Glu Ala Glu Phe Asn Ile
1 5 10
ctc ctg gcc acc gac tcc tac aag gtt act cac tat aaa caa tat cca 98
Leu Leu Ala Thr Asp Ser Tyr Lys Val Thr His Tyr Lys Gln Tyr Pro
15 20 25
ccc aac aca agc aaa gtt tat tcc tac ttt gaa tgc cgt gaa aag aag 146
Pro'Asn Thr Ser Lys Val Tyr Ser Tyr Phe Glu Cys Arg Glu Lys Lys
30 35 40
aca gaa aac tcc aaa tta agg aag gtg aaa tat gag gaa aca gta ttt 194
Thr Glu Asn Ser Lys Leu Arg Lys Val Lys Tyr Glu Glu Thr Val Phe
45 50 55
tat ggg ttg cag tac att ctt aat aag tac tta aaa ggt aaa gta gta 242
Tyr Gly Leu Gln Tyr Ile Leu Asn Lys Tyr Leu Lys Gly Lys Val Val
60 65 70 75
acc aaa gag aaa atc cag gaa gcc aaa gat gtc tac aaa gaa cat ttc 290
Thr Lys Glu Lys Ile Gln Glu Ala Lys Asp Val Tyr Lys Glu His Phe
80 85 90
caa gat gat gtc ttt aat gaa aag gga tgg aac tac att ctt gag aag 338
Gln Asp Asp Val Phe Asn Glu Lys Gly Trp Asn Tyr Ile Leu Glu Lys
95 100 105
tat gat ggg cat ctt cca ata gaa ata aaa gct gtt cct gag ggc ttt 386
Tyr Asp Gly His Leu Pro Ile Glu Ile Lys Ala Val Pro Glu Gly Phe
110 115 120
gtc att ccc aga gga aat gtt ctc ttc acg gtg gaa aac aca gat cca 434
Val Ile Pro Arg Gly Asn Val Leu Phe Thr Val Glu Asn Thr Asp Pro
125 130 135
gag tgt tac tgg ctt aca aat tgg att gag act att ctt gtt cag tcc 482
Glu Cys Tyr Trp Leu Thr Asn Trp Ile Glu Thr Ile Leu Val Gln Ser
140 145 150 155
tgg tat cca atc aca gtg gcc aca aat tct aga gag cag aag aaa ata 530
Trp Tyr Pro Ile Thr Val Ala Thr Asn Ser Arg Glu Gln Lys Lys Ile
160 165 170
ttg gcc aaa tat ttg tta gaa act tct ggt aac tta gat ggt ctg gaa 578
Leu Ala Lys Tyr Leu Leu Glu Thr Ser Gly Asn Leu Asp Gly Leu Glu
175 180 185
57
CA 02588614 2007-05-23
tac aag tta cat gat ttt ggc tac aga gga gtc tct tcc caa gag act 626
Tyr Lys Leu His Asp Phe Gly Tyr Arg Gly Val Ser Ser Gln Glu Thr
190 195 200
gct ggc ata gga gca tct gct cac ttg gtt aac ttc aaa gga aca gat 674
Ala Gly Ile Gly Ala Ser Ala His Leu Val Asn Phe Lys Gly Thr Asp
205 210 215
aca gta gca gga ctt qct cta att aaa aaa tat tat gga acg aaa gat 722
Thr Val Ala Gly Leu Ala Leu Ile Lys Lys Tyr Tyr Gly Thr Lys Asp
220 225 230 235
cct gtt cca ggc tat tct gtt cca gca gca gaa cac agt acc ata aca 770
Pro Val Pro Gly Tyr Ser Val Pro Ala Ala Glu His Ser Thr Ile Thr
240 245 250
gct tgg ggg aaa gac cat gaa aaa gat gct ttt gaa cat att gta aca 818
Ala Trp Gly Lys Asp His Glu Lys Asp Ala Phe Glu His Ile Val Thr
255 260 265
cag ttt tca tca gtg cct gta tct gtg gtc agc gat agc tat gac att 866
Gln Phe Ser Ser Val Pro Val Ser Val Val Ser Asp Ser Tyr Asp Ile
270 275 280
tat aat gcg tgt gag aaa ata tgg ggt gaa gat cta aga cat tta ata 914
Tyr Asn Ala Cys Glu Lys Ile Trp Gly Glu Asp Leu Arg His Leu Ile
285 290 295
gta tcg aga agt aca cag gca cca cta ata atc aga cct gat tct gga 962
Val Ser Arg Ser Thr Gln Ala Pro Leu Ile Ile Arg Pro Asp Ser Gly
300 305 310 315
aac cct ctt gac act gtg tta aag gtt ttg gag att tta ggt aag aag 1010.
Asn Pro Leu Asp Thr Val Leu Lys Val Leu Glu Ile Leu Gly Lys Lys
320 325 330
ttt cct gtt act gag aac tca aag ggt tac aag ttg ctg cca cct tat 1058
Phe Pro Val Thr Glu Asn Ser Lys Gly Tyr Lys Leu Leu Pro Pro Tyr
335 340 345
ctt aga gtt att caa ggg gat gga gta gat att aat acc tta caa gag 1106
Leu Arg Val Ile Gln Gly Asp Gly Val Asp Ile Asn Thr Leu Gln Glu
350 355 360
att gta gaa ggc atg aaa caa aaa atg tgg agt att gaa aat att gcc 1154
Ile Val Glu Gly Met Lys Gln Lys Met Trp Ser Ile Glu Asn Il.e Ala
365 370 375 .
ttc ggt tct ggt gga ggt ttg cta cag aag ttg aca aga gat. ctc ttg 1202
Phe Gly Ser Gly Gly Gly Leu Leu Gln Lys Leu Thr Arg Asp Leu Leu
380 385 390 395
aat tgt tcc ttc aag tgt agc tat gtt gta act aat ggc ctt ggg att 1250
Asn Cys Ser Phe Lys Cys. Ser Tyr Val Val Thr Asn Gly Leu Gly Ile
400 405 410
aac gtc ttc aag gac cca gtt gct gat ccc aac aaa agg tcc aaa aag 1298
Asn Val Phe Lys Asp Pro Val Ala Asp Pro Asn Lys Arg Ser Lys Lys
415 420 425
ggc cga tta tct tta cat agg acg cca gca ggg aat ttt gtt aca ctg 1346
Gly Arg Leu Ser Leu His Arg Thr Pro Ala Gly Asn Phe Val Thr Leu
430 435 440
gag.gaa gga aaa gga gac ctt gag gaa tat ggt cag gat ctt ctc cat 1394
Glu Glu Gly Lys Gly Asp Leu Glu Glu Tyr Gly Gln Asp Leu Leu His
445 450 455
act gtc ttc aag aa.t ggc aag gtg aca aaa agc tat tca ttt gat gaa 1442
Thr Val Phe Lys Asn Gly Lys Val Thr Lys Ser Tyr Ser Phe Asp Glu
460 465 470 475
ata aga aaa aat gca cag ctg aat att gaa ctg gaa gca gca cat cat 1490
Ile Arg Lys Asn Ala Gln Leu Asn Ile Glu Leu Glu Ala Ala His His
480 485 490
tag gctttatgac tgggtgtgtg ttgtg 1518
58