Note: Descriptions are shown in the official language in which they were submitted.
CA 02605472 2007-10-18
WO 2006/135724
PCT/US2006/022418
ELECTROCHEMICAL CELL AND REFERENCE CELL WITH FLOWING LIQUID
JUNCTION
BACKGROUND OF THE INVENTION
Electrochemical cells form the basis of a
variety of analytical sensors. Electrochemical cells
generally have two or more electrodes of the cell and
are coupled to an analyzer that measures an
lo electrical characteristic of the cell to infer a
property of a sample within, or otherwise coupled to,
the cell. Many electrochemical cells include a
measurement electrode and a reference electrode. The
reference electrode will generally include a chamber
that houses a reference electrode fill solution. A
junction, of some sort, allows electrochemical
interaction between a sample solution and the fill
solution. Electrochemical cells can be used for
oxidation/reduction potential (ORP) sensors, pH
sensors, or other suitable sensors.
One type of junction used with reference
electrodes of electrochemical cells is known as a
liquid junction. A liquid junction uses a relatively
small passageway that is in fluidic communication
with both the sample solution and the reference fill
solution. In order to achieve a stable potential at
the liquid junction, it is generally preferred that
at least some flow of fill solution through the
passageway into the sample solution be induced. With
a "flowing" liquid junction, the reference electrode
fill solution constantly flows through the liquid
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
- 2 -
j unct ion . into the sample solution. However, in order
for the flowing liquid junction to work properly, the
fill solution must be free to flow through the
junction pore(s) to overcome the diffusion of cations
and anions in the fill solution. If the passageway
becomes blocked, ions in the fill solution will
diffuse differently. The electrical resistance of the
junction will increase. This can cause
electrochemical readings to drift and become noisy.
lo Severe fouling can sometimes completely block the
liquid junction, breaking the electrical connection
between the electrode and the sample, and making the
electrode unusable. Plugging can come from various
sources, including: suspended solids in a sample or
solids resulting from a chemical reaction involving
the fill solution. Plugged junctions are difficult to
clean.
Therefore, there is a need to provide
electrochemical cells with the longevity advantage of
liquid junctions, but without such cells being as
susceptible to fouling induced by obstructions within
the liquid junction. Such an electrochemical cell
would enjoy the advantages of longevity, stable
reference potential, and relatively low maintenance
requirements.
SUMMARY OF THE INVENTION
An electrochemical cell includes a
measuring electrode and a reference electrode. The
reference electrode includes a flowing liquid
junction between a reference fill fluid and a sample.
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
-3-
The flowing liquid junction is configured to inhibit
particles from blocking or obstructing it. In one
aspect a particle filter is provided before the
flowing liquid junction to prevent particles from the
fill fluid from entering the flowing liquid junction.
In another aspect a particle filter is provided after
the flowing liquid junction to prevent particles in
the sample from entering the flowing liquid junction.
In another aspect the flowing liquid junction has a
lo diameter that generally increases from an aperture
proximate the fill fluid to an aperture proximate the
sample. One example of such a configuration is a
tapered flowing liquid junction.
BRIEF DESCRIPTION OF THE DRAWINGS
FIG. 1 is a diagrammatic view of an
electrochemical cell environment, with which
embodiments of the present invention are particularly
useful.
FIG. 2 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with the prior
art.
FIG. 3 is a diagrammatic view of the
junction illustrated in FIG. 2 further illustrating
an obstruction to the flow passageway.
FIG. 4 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with an embodiment
of the present invention.
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
-4-
FIG. 5 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical in accordance with another embodiment
of the present invention.
FIG. 6 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with another
embodiment of the present invention.
FIG. 7 is a diagrammatic view of a flowing
lo liquid junction of a reference electrode of an
electrochemical cell in accordance with yet another
embodiment of the present invention.
FIG. 8 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
ls electrochemical cell in accordance with yet another
embodiment of the present invention.
DETAILED DESCRIPTION OF ILLUSTRATIVE EMBODIMENTS
FIG. 1 is a diagrammatic view of an
20 electrochemical cell being used to analyze pH. Those
skilled in the art will recognize that embodiments of
the present invention can be practiced with various
types of chemical analysis, and embodiments of the
present invention are certainty not limited to the
25 illustrative pH analyzer application illustrated in
FIG. 1. Meter 10 is coupled to measuring electrode
12, working electrode 14 and temperature sensor 16.
Each of measurement electrode 12, reference electrode
14 and temperature sensor 16 are disposed within
30 sample 18. In most applications, pH is simply
determined by measuring the voltage of
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
- 5 -
electrochemical cell 20. The voltage of cell 20 is
generally directly proportional to the pH of sample
liquid 18. Meter 10 measures the voltage and uses a
temperature-dependent factor to convert the voltage
s to pH. Because cell 20 has a relatively high internal
resistance, meter 10 has a very high input impedance.
Although FIG. 1 illustrates separate measuring and
reference electrodes 12, 14, such electrodes are
sometimes combined, along with temperature sensor 16
lo within a single body. Such sensors are called
combination electrodes.
The voltage of cell 20 is the algebraic sum
of the potentials of measuring electrode 12,
reference electrode 14, and the liquid junction. The
ls potential of measuring electrode 12 depends primarily
on the pH of sample 18. The potential of reference
electrode 14 is unaffected by pH, so it provides a
stable reference voltage. The liquid junction
potential depends in a complex way on the identity
20 and concentration of the ions in sample liquid 18.
The liquid junction potential is always present, but
is usually small and relatively constant if cell 20
is properly designed. All three potentials depend on
temperature.
25 Measuring electrode 12 is generally
application-specific. In an application such as that
illustrated in FIG. 1, where pH is measured,
measuring electrode will include a thin piece of pH-
sensitive glass, which is blown onto the end of a
30 length of glass tubing. Sealed inside of electrode 12
is a solution of potassium chloride buffered at pH 7.
_
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
-6-
A piece of silver wire plated with silver chloride
contacts the solution. However, where electrochemical
cell 20 is used for other applications, measuring
electrode 12 may take any suitable form.
Reference electrode 14 includes a piece of
silver wire plated with silver chloride in contact
with a concentrated solution of potassium chloride
held in a glass or plastic tube. However, the
selection of material for the reference electrode
lo wire as well as the fill solution can also vary
depending on the application. Reference electrode 14
includes flowing liquid junction 22 which fluidically
couples fill solution 24 disposed within reference
electrode 14 to sample solution 18. The use of
flowing liquid junction 22 allows cations and anions
to pass evenly allowing reference electrode 14 to
have a fairly stable junction potential.
FIG. 2 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with the prior
art. FIG. 2 illustrates a portion of an endcap 26
having a narrow flow passageway 28 that fluidically
communicates between an interior of the reference
electrode containing fill solution 24 and an exterior
of the reference electrode exposed to sample 18. The
flowing liquid junction is in the form of a
relatively constant diameter bore, such as provided
by a capillary. It is also known to provide a
plurality of such passageways using a porous material
having a pore diameter that allows for fluidic
communication between the reference fill solution 24
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
- 7 -
and the sample solution 18. However, in both such
circumstances, the passageway itself is believed to
have a relatively constant or irregular diameter
hole(s) that extends from fill solution 24 to sample
solution 18.
FIG. 3 is a diagrammatic view illustrating
the flowing liquid junction of FIG. 2 in a clogged,
or fouled condition. Particles, or solids 30 have
aggregated within flow passageway 28 and obstructed
flow passageway 28 such that fluid communication
therethrough is no longer possible. Plugging can
generally come from at least two sources. Suspended
solids in sample 18 can flow into passageway 28 and
clog it. Additionally, plugging can be generated by
solids resulting from a chemical reaction involving
reference fill solution 24. One way that fouling has
been addressed in the past is to provide a flowing
liquid junction with a relatively large surface area.
However, to achieve a relatively long service life of
fill solution 24, the inner diameter of the flowing
junction should generally be in the range of
micrometers, or smaller. This design goal increases
the clogging problems associated with flowing liquid
junctions of the prior art.
FIG. 4 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with an embodiment
of the present invention. FIG. 4 illustrates a
flowing liquid junction having an internal passageway
28 that has a relatively constant internal diameter
as it fluidically couples reference fill solution 24
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
-8-
to sample solution 18. However, disposed within the
reference electrode, proximate an internal surface of
endcap 26 of the electrode is filter 32. Filter 32 is
a particle filter that is placed in the flow path
before passageway 28. The flow path is illustrated
diagrammatically at reference numeral 34. Filter 32
can be any suitable porous material as long as the
pore size of filter 32 is smaller than the inner
diameter of passageway 28. In a preferred embodiment,
lo filter 32 is a sub-micron porous polymer particle
filter sheet such as that provided under the trade
designation Supor supplied by Pall. Any particle
that passes through filter 32 will not be sized to
clog, or otherwise obstruct passageway 28, on its
own.
FIG. 5 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with another
embodiment of the present invention. FIG. 5 bears
many similarities to FIG. 4, and reference numerals
are numbered similarly. In the embodiment illustrated
in FIG. 5, a second porous particle filter 36 is
disposed proximate an external surface of endcap 26.
Filter 36 is in contact with sample solution 18 and
with passageway 28. Filter 36 may be identical to
filter 32, or may be of any other suitable design, as
long as it also has a pore size that is smaller than
the inner diameter of passageway 28. Again, it is
preferred that filter 36 be formed of a sub-micron
porous polymer sheet material such as Supor .
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
-9-
FIG. 6 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with another
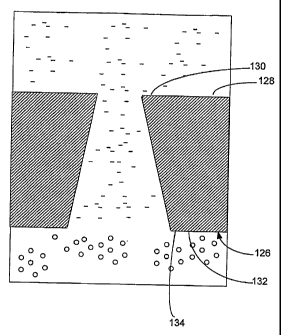
embodiment of the present invention. Endcap 126 is
s similar to endcap 26 (shown in FIG. 1) but is given a
different reference numeral because its physical
configuration proximate the flowing liquid junction
is different. In particular, an internal surface 128
of endcap 126 has an aperture 130 of a first
lo diameter. Additionally, endcap 126 has an external
surface 132 having an aperture 134 with a diameter
that is larger than aperture 130. Preferably, the
internal diameter varies linearly between aperture
130 and aperture 134 to create a tapered flowing
ls liquid junction. However, other configurations can be
practiced in accordance with embodiments of the
present invention as long as the diameter of the
internal passageway at any point beyond aperture 130
is always larger than aperture 130. In a preferred
20 embodiment, however, the flowing liquid junction is a
tapered flowing liquid junction. Since the wall of
the junction is tapered, aperture 134 is larger than
aperture 130. Accordingly, if any particle manages to
enter through aperture 130, the tapered wall makes it
25 relatively easy for the flowing junction to
essentially perform self-cleaning.
FIG. 7 is a diagrammatic view of flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with another
30 embodiment of the present invention. The embodiment
illustrated in FIG. 7 bears many similarities to that
CA 02605472 2007-10-18
WO 2006/135724 PCT/US2006/022418
-10-
illustrated in FIG. 6, and like components are
numbered similarly. FIG. 7 illustrates the flowing
liquid junction also including a particle filter 32
disposed proximate internal surface 128. Filter 32
can be identical to that described above with respect
to FIGS. 4 and 5. Filter 32 has a pore size that is
selected to be smaller than the diameter of aperture
130. Thus, filter 32 helps to ensure that no
particles that are larger than aperture 130 even
reach aperture 130. However, if such as a particle
does manage to get through filter 32, the tapered
design of the flowing liquid junction helps ensure
that the particle is swept cleanly through the
passageway into the sample 18.
FIG. 8 is a diagrammatic view of a flowing
liquid junction of a reference electrode of an
electrochemical cell in accordance with yet another
embodiment of the present invention. The embodiment
illustrated in FIG. 8 bears many similarities to that
described with respect to FIG. 7, and like components
are numbered similarly.
The primary difference between the
embodiment of FIG. 8 and that of FIG. 7 is that the
embodiment illustrated in FIG. 8 utilizes yet another
filter 136 disposed proximate surface 132.
Preferably, filter 136 is a sub-micron porous
polymeric sheet material, such as Suporc), that has a
maximum pore size that is smaller than the diameter
of aperture 130.
Embodiments of the present invention are
believed to provide the benefits of the extended
CA 02605472 2012-12-10
-11-
operating lifetimes associated with reference
electrodes that use flowing liquid junctions, while
also providing the stability of the reference
potential enjoyed by such flowing liquid junctions.
Moreover, embodiments of the present invention are
less susceptible to clogging, or similar forms of
fouling than liquid junctions of the prior art.