Note: Descriptions are shown in the official language in which they were submitted.
CA 02802245 2013-01-16
P8120/Ha
Sulzer Metco AG, CH-5610 Wohlen (Switzerland)
Plasma spray method
The invention relates to a plasma spray method in accordance with claim
1.
It is known that nitride nanopowder or carbide nanopowder can be
manufactured using suitable liquid or gaseous starting materials, so-
called precursors. In this connection, a nanopowder should be understood
as a powder having a grain size of approximately 1 nm to 11.1m. Suitable
precursors, for example titanium tetrachloride or tetrakis (dimethylamino)
titanium, are very expensive and usually very toxic or dangerous. The
precursors are vaporized for manufacturing the nanopowder and form
nanopowder in a reactive chemical gas phase deposition process (a so-
called CVD process). It is also possible to manufacture a nitride coating or
a carbide coating on a substrate using a comparable process.
In addition, so-called plasma spray gas phase deposition processes (so-
called PS-PVD processes) are known by means of which films can be
generated on a substrate from a starting material in powder form. For this
purpose, the starting material is introduced into a plasma and thus
converted into the gas phase and is deposited from the gas phase onto the
substrate as a thin film. Thermal barrier coatings are produced in this
manner, for example. In this respect, yttria stabilized zirconia (abbreviated
YSZ) is, for example used as a non-metallic, inorganic starting material.
CA 02802245 2013-01-16
2
Against this background, it is the object of the invention to propose a
plasma spray method which can serve as a starting point for an
inexpensive manufacture of metal nanopowder, nitride nanopowder or
carbide nanopowder or of metal films, nitride films or carbide films. This
object is satisfied in accordance with the invention by a plasma spray
method having the features of claim 1.
In the plasma spray method in accordance with the invention, a starting
material is introduced into a plasma jet generated by a plasma generator
at a process pressure of at most 1000 Pa, in particular at most 400 Pa.
The starting material contains a metal or silicon oxide which vaporizes in
the plasma jet and is reduced in so doing. After the reduction, the metal or
silicon which had formed the metal or silicon oxide in the starting material
is thus present in pure form or in almost pure form.
The term "process pressure" should be understood as that pressure at
which the process runs, that is at which the plasma jet is formed. Since
the required process pressure is much smaller than the atmospheric
pressure, the whole process takes place in a closed process chamber in
which the process pressure can be set.
The starting material in this respect is composed of in particular 95% to
100% metal oxide, particularly preferably 100% of metal oxide, of a single
metal or of different metals, in particular of zirconium oxide (zirconia),
hafnium oxide or titanium oxide. In addition to the metal oxide, the
starting material can, for example, be composed of other oxides, for
example silicon oxide.
CA 02802245 2013-01-16
3
In the method in accordance with the invention, the starting material
containing metal or silicon oxide is injected and thus introduced into a
plasma jet, for example an argon-helium plasma, generated by a plasma
torch known per se. The starting material is in this respect in particular
introduced into the plasma jet as a powder. It can, for example, be
introduced as a loose powder by means of a carrier gas. The carrier gas is,
for example, a noble gas, a noble gas mixture or an inert gas. Examples
for carrier gases are argon or a helium-argon mixture. It is, however, also
possible that the starting material is introduced into the plasma jet in a
suspension, that is as a dispersed powder in a liquid, for example in
ethanol,
The plasma gas thereby arising expands in a nozzle of the plasma torch
due to the high temperature of the plasma from 10,000 to 20,000 Kelvin
and accelerates to supersonic speed. Due to the named low process
pressure, an expansion of the plasma gas takes place into a process
chamber at low pressure, with a long, large-area plasma jet with
expansion zones and compression zones arising. The plasma jet in
particular has a length between 1 and 2.5 m. The metal or silicon oxide
contained in the starting material vaporizes in the plasma jet due to the
high temperature and to the low pressure. In this respect, an oxygen loss,
that is a reduction of the metal or silicon oxide, takes place due to the low
partial pressure of the oxygen so that the metal or silicon is present in
pure form or in almost pure form in the plasma flow after the reduction.
The starting material is in particular supplied and is thus introduced into
the plasma at a comparatively low feed rate. The supply rate in particular
lies in a range between 0.1 and 5 g/min. It can thus be achieved that the
total metal or silicon oxide or almost the total metal oxide introduced is
completely reduced.
CA 02802245 2013-01-16
4
The total flow rate of the process gate in particular amounts to between 50
and 200 SLPM (standard liters per minute) and particularly preferably to
90 to 120 SLPM.
In an embodiment of the invention, an additional reactant is introduced
into the plasma so that a reaction can take place between the reduced
metal or silicion oxide and the reactant to form a reaction product. The
reactant in particular contains nitrogen and/or carbon so that the pure
metal or silicon arising on the reduction can react to form a metal or
silicon nitride and/or a metal or silicon carbide. It is thus advantageously
possible to manufacture metal or silicon nitride and/or metal or silicon
carbide from a very inexpensive and non-dangerous starting material in
the form of metal or silicon oxide powder.
Depending on the partial pressure of the elements in the reaction to the
reaction product, M0xNy or MO.Cy or MN y or MC, arise, where M stands
for the metal forming the metal oxide, for example zirconium or titanium.
The named partial pressures can be influenced by means of process
parameters such as the process pressure, type of process gas and the
current for generating the plasma, the flow rate of the process gas or the
supply rate of the starting material. A large reduction is in particular
achieved by a high current as well as by a low powder conveying rate.
The reactant containing nitrogen can, for example, be pure gaseous
nitrogen or air. The reactant containing carbon can, for example, be
gaseous as carbon dioxide or methane or can be in solid form as starch or
as a polymer.
CA 02802245 2013-01-16
The pure metal, the silicon or the reaction product arising on the
reduction can be deposited from the plasma jet. It can be deposited, for
example, in the form of metal or silicon nanopowder. The deposition in
particular takes place at a comparatively small spacing from a discharge
5 nozzle for the plasma jet. The spacing in particular amounts to between
100 and 400 mm. The named range is characterized in that, on the one
hand, the reduction of the metal or silicon oxide is fully or at least almost
fully completed and, on the other hand, the metal or silicon particles have
not yet entered into any other bonds. The named spacing is in particular
advantageous when metal or silicon nanopowder is to be manufactured.
The plasma spray method in accordance with the invention allows an
inexpensive manufacture of metal or silicon nanopowder, nitride
nanopowder or carbide nanopowder. In addition, with corresponding
process conditions, nanopowders can thus be manufactured from non-
meltable nitride compounds or carbide compounds such as silicon nitride
(Si3N4).
A method is thus proposed for manufacturing a metal or silicon powder, a
metal or silicon nitride powder or a metal or silicon carbide powder in
which a starting material in the form of a metal or silicon oxide is used
which is introduced at a process pressure of at most 1000 Pa into a
plasma flow which is generated by a plasma generator and in which the
starting material is vaporized and in so doing reduced and arising metal
particles, metal nitride particles or metal carbide particles are deposited
from the process jet as powder.
The deposition as a nanopowder in particular takes place when the
plasma jet can form without impacting a barrier, for example in the form
of a substrate. To promote the deposition as a nanopowder, the plasma jet
CA 02802245 2013-01-16
6
can also be directly cooled in a defined region, for example by means of a
gas flow; the metal or silicon can therefore be quenched so-to-say and the
formation of metal or silicon nanopowder can thus be promoted. The gas
flow is, for example, a gas flow from a noble gas (e.g. argon), from a noble
gas mixture (e.g. argon-helium mixture) or from an inert gas and is
oriented transversely to the plasma jet, for example. An electrostatic filter
can also be used for promoting the deposition.
The grain size of the arising nanopowder can in particular be influenced
by the type of the plasma gas which has an influence on the plasma
temperature and the plasma speed and by the process pressure which has
an influence on the condensation of the nanopowder. The grain size in
particular becomes smaller as the current increases and the powder
conveying rate decreases.
The pure metal or silicon or the reaction product created in the reduction
can also be deposited from the plasma jet as a film on a substrate. A
substrate should be understood in this connection as a workpiece to be
coated, for example a turbine blade. For this purpose, the plasma jet is
directed to the substrate so that a film of the pure metal or silicon or of
the reaction product is formed on the substrate. The plasma spray method
in accordance with the invention thus makes possible an inexpensive
metal or silicon coating, nitride coating or carbide coating of a substrate.
In addition, on the setting of corresponding process parameters using the
method in accordance with the invention coatings of non-meltable nitride
compounds or carbide compounds such as silicon nitride (S13N4) can be
manufactured. This is not possible or is only possible with limitations with
known spray methods for coating.
CA 02802245 2013-01-16
=
7
A method is thus proposed for manufacturing a film on a substrate using
a plasma spray method in which a starting material in the form of a metal
oxide is used which is introduced at a process pressure of at most
1000 Pa into a plasma flow which is generated by a plasma generator and
in which the starting material is vaporized and in so doing reduced and
arising metal particles, metal nitride particles or metal carbide particles
are deposited as a film on s substrate.
The films produced in particular have a thickness between 50 nm and
500 pm and can be deposited over a large area in both a dense and a
porous form. The porous films in particular have a columnar design. The
design and the property of the film as well as the film growth can be
influenced via the named process parameters, with in particular a higher
powder conveying rate resulting in rather porous films.
Further advantages, features and details of the invention result with
reference to the following description from embodiments and with
reference to drawings in which elements which are the same or have the
same function are provided with identical reference numerals.
There are shown:
Fig. 1 a schematic representation of a plasma spray apparatus for
manufacturing nanopowder; and
Fig. 2 a schematic representation of a plasma spray apparatus for
producing a film on a substrate.
In accordance with Fig. 1, a plasma spray apparatus 11 suitable for
carrying out a method in accordance with the invention has a plasma
CA 02802245 2013-01-16
8
generator 12 known per se and having a plasma torch, not shown in more
detail, for producing a plasma. A process jet 13 is generated in a manner
known per se from a starting material P, a process gas mixture G and
electrical energy E using the plasma generator 12. The feeding of these
components E, G and P is symbolized by the arrows 14, 15, 16 in Fig. 1.
The generated plasma jet 13 exits the plasma generator through an outlet
nozzle 17 and transports the starting material P in the form of the plasma
jet 13 in which material particles 18 are dispersed in a plasma. This
transport is symbolized by an arrow 19.
The process gas G for the production of the plasma is preferably a mixture
of inert gases, in particular a mixture of argon, hydrogen and helium.
The plasma spray apparatus 11 is arranged in a process chamber 20 in
which a defined process pressure can be set by means of pumps, not
shown. On the carrying out of the method in accordance with the
invention, a process pressure of less than 1000 Pa, in particular between
100 and 400 Pa, is set. Due to the named process pressure, a
comparatively long plasma jet having a length between 1 and 2.5 m is
produced.
Specifically, a plasma spray gas deposition method (PS-PVD) is carried out
using the plasma spray apparatus 11 shown in Fig. 1. In this method, the
starting material P which is composed of titanium oxide (Ti02, zirconia
(Zr02), hafnium oxide (Hf203) or silica (Si02) in powder form is introduced
into the argon-helium plasma generated by the plasma generator 12, and
thus introduced into the plasma jet 13, by means of a carrier gas, for
example in the form of argon.
CA 02802245 2013-01-16
9
The plasma gas thereby arising expands in and after the exit from the
outlet nozzle 17 of the plasma generator 12 due to the high temperature of
the plasma of 10,000 to 20, 000 Kelvin and accelerates to supersonic
speed. The metal oxide contained in the starting material P vaporizes in
the plasma jet 13 due to the high temperature and to the low pressure. In
this respect, an oxygen loss, that is a reduction of the metal oxide, takes
place due to the low partial pressure of the oxygen so that the metal is
present in pure form or in almost pure form in the plasma flow after the
reduction.
The starting material P is supplied at a comparatively low supply rate. The
supply rate in particular lies in a range between 0.1 and 5 g/min.
The current set for the generation of the plasma in this respect has a
current between approx. 1000 and 3000 A, in particular between 2200
and 3000 A.
The starting material P is injected into the plasma as a powder jet with a
conveying gas, preferably argon or a helium-argon mixture. The flow rate
of the conveying gas preferably amounts to 5 to 40 SLPM (standard liters
per minute), in particular to 10 to 25 SLPM.
The process gas for the generation of the plasma is preferably a mixture of
inert gases, in particular a mixture of argon Ar, helium He and hydrogen
H. In practice, a total gas flow between 50 and 200 SLPM, in particular 90
to 120 SLPM has proven itself. Of this in particular approximately 1/3 is
argon and 2/3 helium. In addition, a portion of up to 10 SLPM hydrogen
is conceivable.
CA 02802245 2013-01-16
To promote a deposition of the created metal particles in the form of a
nanopowder, the plasma jet 13, and thus also the metal particles 18
contained in the plasma jet 13, are directly cooled by a gas flow 21
oriented transversely to the plasma flow 13 and the metal is thus
5 quenched so-to-say. A condensation of the gaseous metal particles 18 is
triggered and nanopowder 22 is formed which collects in a collection
apparatus 23.
The gas flow 21 in this respect has a spacing D1 from the exit nozzle 17
10 between 100 and 400 mm, in particular 150 mm.
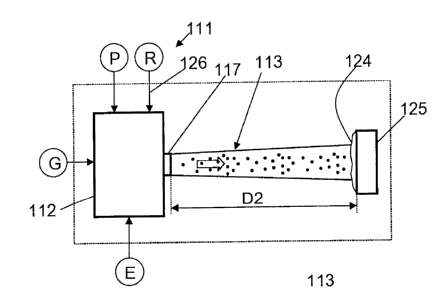
A plasma spray apparatus 111 for generating a film 124 on a substrate
125 is shown in Fig. 2. The design of the plasma spray apparatus 111
corresponds in large parts to the plasma spray apparatus 11 of Fig. 1 so
that mainly the differences of the two plasma spray apparatus will be
looked at.
A reactant R, whose infeed is symbolized by an arrow 126, is supplied to a
plasma generator 112 of the plasma spray apparatus 111 in addition to
the starting material P which is in turn titanium oxide (Ti02) or zirconia
(Zr02) in powder form. The reactant in particular contains nitrogen or
carbon so that the pure metal arising on the reduction of the metal oxide
can react to form a metal nitride and/or a metal carbide. The reactant
containing nitrogen can, for example, be pure gaseous nitrogen. The
reactant containing carbon can, for example, be gaseous as carbon dioxide
or methane or can be in solid form as starch or as a polymer. When the
reactant is supplied in solid form, this is also done using a transport gas.
In this respect, it can be the same transport gas as for the starting
material P or a transport gas differing therefrom.
CA 02802245 2013-01-16
=
11
After the above-described reduction of the metal oxide of the starting
material P, a reaction of the pure metal with the reactant R to form a
reaction product takes place in the plasma flow 113. If one looks at the
total method, the oxygen bound in the metal oxide is replaced with
nitrogen or carbon of the reactant R to gain the reaction product.
Depending on the partial pressure of the elements on the reaction to the
reaction product, M0xNy or MO,Cy or MN y or MC, arise, where M stands
for the metal forming the metal oxide, that is zirconium or titanium. The
named partial pressures can be influenced by means of process
parameters such as the process pressure, type of process gas and the
current for generating the plasma, the flow rate of the process gas or the
supply rate of the starting material.
The substrate 125 is arranged at a spacing D2 from an exit nozzle 117 of
the plasma generator 112. The spacing D2 can be selected as larger than
the spacing D1 in Fig. 1; it in particular amounts to between 500 and
2000 mm. The named reaction product is deposited at the substrate 125
as a film 124. The film 124 can have a thickness between 50 nm and
500 pm and can be made both dense and porous. The design and the
property of the film as well as the film growth can be influenced via the
named process parameters.
It is naturally also possible that, in the generation of nanopowder in
accordance with Fig. 1, a reactant is supplied and thus a nitride
nanopowder or carbide nanopowder is generated. It is equally possible to
dispense with the supply of a reactant in the generation of a film on a
substrate in accordance with Fig. 2 and thus to generate a film of pure
metal. To achieve satisfactory results, specific adaptations of the process
parameters will be necessary under certain circumstances.