Note: Descriptions are shown in the official language in which they were submitted.
CA 02827765 2013-08-19
WO 2012/121710 PCT/US2011/027574
CARBON DIOXIDE GAS MIXTURE PROCESSING WITH STEAM ASSISTED OIL
RECOVERY
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] None
STATEMENT REGARDING FEDERALLY SPONSORED RESEARCH OR
DEVELOPMENT
[0002] None
FIELD OF THE INVENTION
[0003] Embodiments of the invention relate to methods and systems for
processing
carbon dioxide in flue gas from oxy-fuel combustion utilizing steam assisted
oil recovery.
BACKGROUND OF THE INVENTION
[0004] Oxy-fuel combustion refers to burning of fuel in oxygen (e.g., 95%
pure oxygen)
instead of air to reduce amount of nitrogen in resulting flue gas. The flue
gas from the oxy-fuel
combustion thus contains carbon dioxide and water vapor, which can be removed
if condensed
through cooling. The oxy-fuel combustion facilitates carbon dioxide capture
since the flue gas is
almost pure carbon dioxide with trace amounts of impurities, such as oxygen
(e.g., about 0.1-2
volume percent) remaining due to equilibrium constraints as well as local
mixing conditions
during combustion.
[0005] The oxygen in the carbon dioxide makes transportation of the
carbon dioxide to a
sequestration site problematic since the oxygen causes corrosion. Common
carbon dioxide
quality specifications for pipeline transport require oxygen content to be
below 0.001 by volume.
Cryogenic distillation provides one option for removing the oxygen but
requires additional
expense and results in loss of 7-10 percent of the carbon dioxide.
[0006] Alternate approaches utilize the flue gas from the oxy-fuel
combustion. For
example, injecting the flue gas into reservoirs of natural gas helps displace
the natural gas.
However, gas phase interactions of the flue gas in the reservoirs and the
interactions not
occurring at where the reservoir is being heated limits any possible oxygen
removal from the
carbon dioxide.
[0007] Therefore, a need exists for improved methods and systems for
processing carbon
dioxide for capture.
1
CA 02827765 2013-08-19
WO 2012/121710 PCT/US2011/027574
SUMMARY OF THE INVENTION
[0008] In one embodiment, a method includes forming a mixture of flue gas
from oxy-
fuel combustion and steam generated prior to being mixed with the flue gas.
The flue gas
contains carbon dioxide with an initial concentration of oxygen that is at
least 0.1 volume
percent. Injecting the mixture into a subterranean formation heats oil in the
formation and reacts
with the oil at least some of the oxygen that is from the flue gas and is
dissolved in liquid
condensate of the steam. The method further includes recovering fluids
including the oil that is
heated and carbon dioxide from the flue gas and separating the fluids
recovered to isolate from a
liquid phase the carbon dioxide containing less than the initial concentration
of oxygen for
transporting the carbon dioxide to a sequestration site.
[0009] According to one embodiment, a method includes producing flue gas
from oxy-
fuel combustion, generating steam without contact of the steam with the flue
gas, and
introducing the steam into the flue gas to form a mixture. The flue gas
contains carbon dioxide
with quantities of oxygen greater than a transport specification. In addition,
the method includes
injecting the mixture into a subterranean formation for heating oil in the
formation, recovering
fluids including the oil that is heated and the carbon dioxide from the flue
gas, and separating the
fluids into liquids and vapors. The vapors formed of the carbon dioxide meet
the transport
specification due to removal of at least some of the oxygen by oxidation of
the oil upon the
oxygen being dissolved in condensate of the steam for liquid phase reactions
at temperatures
elevated by the steam. The method further includes transporting to a
sequestration site the
carbon dioxide obtained by the separating.
[0010] For one embodiment, a system includes a supply of flue gas from an
oxy-fuel
combustion chamber, a source of steam generated without contact of the steam
with the flue gas,
and an injection well disposed in a subterranean formation containing oil. The
injection well
couples in fluid communication with the supply of the flue gas and the source
of the steam. A
vapor-liquid separator of the system receives produced fluids heated by the
steam. The vapor-
liquid separator also outputs carbon dioxide that is in the produced fluids
from the flue gas and is
processed by liquid phase reactions between the oil heated by the steam and at
least some oxygen
that is from the flue gas and is dissolved in condensate of the steam.
2
CA 02827765 2013-08-19
WO 2012/121710 PCT/US2011/027574
BRIEF DESCRIPTION OF THE DRAWINGS
[0011] The invention, together with further advantages thereof, may best
be understood
by reference to the following description taken in conjunction with the
accompanying drawings.
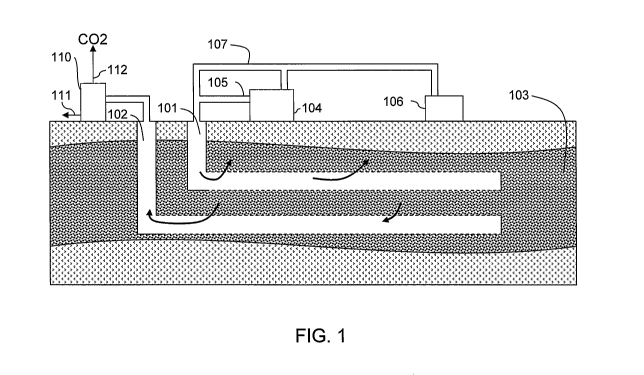
[0012] Figure 1 is a schematic of a production system for both
purification of carbon
dioxide in flue gas and steam assisted oil recovery, according to one
embodiment of the
invention.
DETAILED DESCRIPTION OF THE INVENTION
[0013] Embodiments of the invention relate to methods and systems for
processing flue
gas from oxy-fuel combustion. Steam generated without contact of the steam
with the flue gas
combines with the flue gas for injection into a formation to facilitate oil
recovery from the
formation. Fluids produced include the oil and carbon dioxide with a lower
concentration of
oxygen than present in the flue gas that is injected.
[0014] Figure 1 illustrates a system with an injection well 101 and a
production well 102
that traverse through an earth formation 103 containing petroleum products,
such as heavy oil or
bitumen. The system further includes a steam generator 104 to supply a flow of
steam 105 to the
injection well 101. The steam generator 104 and/or a separate heating unit 106
produce flue gas
107 from oxy-fuel combustion.
[0015] The oxy-fuel combustion produces the flue gas 107 containing
carbon dioxide
with at least 0.1 volume percent oxygen as a result of burning fuel in oxygen,
such as at least
about 95% by volume pure oxygen separated from air. The carbon dioxide may
make up by
volume at least about 85%, at least about 90%, or at least about 95% of the
flue gas 107.
Sources for the fuel include coal, petroleum coke, asphaltenes, methane,
natural gas and
hydrogen. To limit resulting flame temperatures to levels common during
conventional
combustion and within thermal thresholds, some cooled combustion gases may
circulate back
and be injected into a combustion chamber used for the oxy-fuel combustion.
[0016] For some embodiments, a burner heats a boiler within the steam
generator 104 for
making the steam 105 without initial contact of the flue gas 107 with the
steam 105 in the steam
generator 104 since an inside of the boiler is sealed from the burner, which
may define the
chamber for the oxy-fuel combustion. The flue gas 107 combines with the steam
105 to form a
mixture after the steam 105 is generated. The mixture passes into the
injection well 101 upon
introducing the flue gas 107 into the steam 105 between the steam generator
104 and the
3
CA 02827765 2013-08-19
WO 2012/121710 PCT/US2011/027574
injection well 101. The mixture may in some embodiments further contain a
solvent for the
products added to help mobilize the products, which are more viscous than the
solvent.
Examples of the solvent introduced into the mixture include hydrocarbons, such
as at least one of
propane, butane, pentane, hexane, heptane, naphtha, natural gas liquids and
natural gas
condensate.
[0017] In operation, the mixture makes the petroleum products mobile
enough to enable
or facilitate recovery with, for example, the production well 102. For some
embodiments, the
injection well 101 includes a horizontal borehole portion that is disposed
above (e.g., 0 to 6
meters above) and parallel to a horizontal borehole portion of the production
well 102. While
shown in an exemplary steam assisted gravity drainage (SAGD) well pair
orientation, some
embodiments utilize other configurations of the injection well 101 and the
production well 102,
which may be combined with the injection well 101 or arranged crosswise
relative to the
injection well 101, for example.
[0018] A vapor chamber develops in the formation 103 and grows as the
products are
recovered. Walls of the vapor chamber form an interface with the products
where the steam 105
condenses transferring heat to the products that then drain to the production
well 102. Since the
flue gas 107 containing the oxygen is injected into the vapor chamber during
development of the
chamber, the oxygen contacts this condensate of the steam 105 and is dissolved
in the condensate
enabling both the condensate to carry oxygen into the products and liquid
phase reaction of the
oxygen with the products. For some embodiments, effective removal of the
oxygen from the
carbon dioxide in the flue gas 107 relies on the reactions being in liquid
phase compared to
inefficient gas contact of the oxygen with the products.
[0019] In some embodiments, this oxidation of the products further
depends on
temperature at which the oxygen contacts the products since oxygen uptake by
the products
increases with rising temperature. The reactions for some embodiments occur at
temperatures
that are elevated by the steam 105 and may be above about 100 C, above about
150 C or above
about 200 C. Injection of the flue gas 107 through a separate well and
outside of the vapor
chamber formed by the steam 105 heating the products tends to keep the oxygen
in gas phase
and insulated from thermal heating by the steam 105 due to physical separation
of the oxygen
from the condensate. Likewise, injection of the flue gas 107 after stopping
injection of the steam
105 prevents ability of the oxygen to be dissolved in the condensate and
carried into the products
4
CA 02827765 2013-08-19
WO 2012/121710 PCT/US2011/027574
at as high a temperature as possible. While helpful for processing the carbon
dioxide in the flue
gas 107, the oxidation of the products lacks influence on recovery due to only
trace amounts of
the oxygen in the flue gas 107.
[0020] As a benefit to recovery, the carbon dioxide from the flue gas 107
also dissolves
into the products reducing viscosity of the products to facility production.
The formation 103
retains some amount of the carbon dioxide from the flue gas 107. Pore space
opened from one
barrel of produced oil stores about 8 kilograms of the carbon dioxide. Unlike
loss of carbon
dioxide associated with cleaning of the flue gas 107 above ground to remove
oxygen, the carbon
dioxide being held in the formation 103 remains sequestered without requiring
any additional
treatment to be captured.
[0021] Fluid recovered from the production well 102 enters into a
separator 110 for
separation of a liquid phase 111 from a vapor phase 112. The liquid phase 111
includes the
petroleum products and water, which may be separated from the products and
recycled along
with any solvent removed from the products. The carbon dioxide from the flue
gas 107 forms
the vapor phase 112 and may make up by volume at least about 90%, or at least
about 95% of the
vapor phase 112. Lack of substantial quantities of nitrogen in the flue gas
107 due to the oxy-
fuel combustion limits nitrogen amounts in the vapor phase 112 ensuring that
the carbon dioxide
therein remains concentrated for desired capture and sequestration.
[0022] Due to the oxidation reactions discussed herein, the vapor phase
112 contains a
lower concentration of the oxygen than is present in the flue gas 107 prior to
introduction into
the injection well 101. In some embodiments, the flue gas 107 with the at
least 0.1 volume
percent oxygen before being introduced into the injection well 101 prevents
the flue gas 107
from meeting transport specifications. For example, oxygen content of above
0.001% by volume
in the flue gas 107 as produced from the oxy-fuel combustion may reduce to
below 0.001% by
volume in the vapor phase 112 and thereby be below the transport
specifications.
[0023] This reduction in oxygen content processes the carbon dioxide to
enable
transporting the carbon dioxide without further oxygen removal from the vapor
phase 112. For
some embodiments, transport of the carbon dioxide includes compressing the
vapor phase 112
that is then passed through a pipeline. The pipeline may carry the carbon
dioxide to a
sequestration facility such as a geologic reservoir distant from the formation
103 in which the
products are recovered.
CA 02827765 2013-08-19
WO 2012/121710 PCT/US2011/027574
[0024] In some embodiments, injection of the flue gas 107 from the oxy-
fuel combustion
into a depleted hydrocarbon reservoir passes the flue gas 107 into contact
with unrecovered
petroleum products that react with the oxygen from the flue gas 107. Such
oxidation scrubs
oxygen from the flue gas 107 leaving the carbon dioxide that may be
subsequently recovered for
transporting even though no hydrocarbons are also produced while recovering
the carbon
dioxide. If only such scrubbing of the flue gas 107 and not recovering of the
petroleum products
is desired, some embodiments may inject the flue gas 107 without mixing the
flue gas 107 with
the steam 105.
[0025] The preferred embodiment of the present invention has been
disclosed and
illustrated. However, the invention is intended to be as broad as defined in
the claims below.
Those skilled in the art may be able to study the preferred embodiments and
identify other ways
to practice the invention that are not exactly as described herein. It is the
intent of the inventors
that variations and equivalents of the invention are within the scope of the
claims below and the
description, abstract and drawings are not to be used to limit the scope of
the invention.
6