Note: Descriptions are shown in the official language in which they were submitted.
81800293
1
COMPOUNDS AND COMPOSITIONS AS TOLL-LIKE RECEPTOR 7 AGONISTS
CROSS-REFERENCE TO RELATED APPLICATIONS
This application claims the benefit of U.S. Provisional Application No.
61,987,321,
filed 01 May 2014.
FIELD OF THE INVENTION
The invention relates to compounds which are Toll-Like Receptor 7 (TLR7)
agonists,
compositions containing such compounds and methods of using such compounds.
BACKGROUND OF THE INVENTION
Early detection of specific classes of pathogens is accomplished by the innate
immune
system with the help of pattern recognition receptors (PRRs). The detected
pathogens include
viruses, bacteria, protozoa and fungi, and each constitutively expresses a set
of class-specific,
mutation-resistant molecules called pathogen-associated molecular patterns
(PAMPs). These
molecular markers may be composed of proteins, carbohydrates, lipids, nucleic
acids or
combinations thereof, and may be located internally or externally. Examples of
PAMPs include
bacterial carbohydrates (lipopolysaccharide or LPS, mannose), nucleic acids
(bacterial or viral
DNA or RNA), peptidoglycans and lipotechoic acids (from Gram positive
bacteria), N-
formylmethionine, lipoproteins and fungal glucans.
Pattern recognition receptors have evolved to take advantage of three PAMP
qualities.
First, constitutive expression allows the host to detect the pathogen
regardless of its life cycle
stage. Second, the PAMPs are class specific, which allows the host to
distinguish between
pathogens and thereby tailor Its response. Third, mutation resistance allows
the host to
recognize the pathogen regardless of its particular strain.
Pattern recognition receptors are involved in more than just recognition of
pathogens via
their PAMPs. Once bound, pattern recognition receptors tend to cluster,
recruit other
extracellular and intracellular proteins to the complex, and initiate
signaling cascades that
ultimately impact transcription. Additionally, pattern recognition receptors
are involved in
activation of complement, coagulation, phagocytosis, inflammation, and
apoptosis functions in
response to pathogen detection.
Pattern recognition receptors (PRRs) may be divided into endocytic PRRs or
signaling
PRPIs. The signaling PRRs include the large families of membrane-bound Toll-
like receptors
(TLRs) and cytoplasmic NOD-like receptors, while the endocytic PRRs promote
the attachment,
engulfment and destruction of microorganisms by phagocytes without relaying an
intracellular
signal, are found on all phagocytes and mediate removal of apoptotic cells. in
addition,
endocytic PRRs recognize carbohydrates and include mannose receptors of
macrophages,
CA 2946408 2019-09-27
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
9
glucan receptors present on all phagocytes and scavenger receptors that
recognize charged
ligands.
SUMMARY OF THE INVENTION
Provided herein are compounds and pharmaceutical compositions thereof, which
are
agonists of toll-like receptor 7 (TLR7). Such TLR7 agonists are immune
potentiators. Also
provided herein are immunogenic compositions that contain such TLR7 agonists.
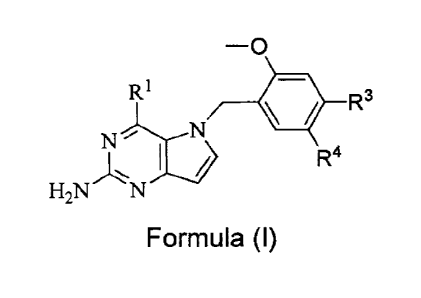
In one aspect such compounds, and the pharmaceutically acceptable salts,
individual
isomers and mixture of isomers thereof, have a structure according to Formula
(I):
-o
RI = R3
N
R4
H2N N
Formula (I)
wherein:
1111 is -NNW or -NHCHR6R9;
R3 is H, -L2F112, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -
CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
R4 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -
CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
where, when R3 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)OH, -CH=CHC(=0)0H, -
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
or when R4 is H, then R3 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2F12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
L1 is -(CH2)m-;
1_2 1S -(CH2)m-;
1-7 = -(CF12)m(CHR11CF12)m(CF12)n-;
R6 is -C3-C6alkyl;
R7 is -C1-C3alkyl;
R9 is 1_101-1;
each R11 is independently selected from -OH;
R12 is
a) an unsubstituted tetrazolyl;
b) a tetrazolyl substituted with -1_1C(=0)0H;
or
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
3
C) a 5-6 membered heterocycloalkyl having 1 to 2 heteroatoms independently
selected from N and 0 substituted with -C(=0)0H;
each m is independently selected from 1, 2, 3, and 4,
and
.. each n is independently selected from 0, 1, 2, 3, and 4.
In certain embodiments, the compound of Formula (1) is a compound of Formula
(la) or
Formula (lb):
-0 -0
RI 411 R3 R1
N =
H2N N 112N N
Formula (la) Formula (lb).
In certain embodiments of the compounds of Formula (1),
R1 is -NHRe or -NHCHReR9;
R3 is H, -0L2C(=0)0H or tetrazolyl;
R4 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -
CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H or tetrazolyl;
where, when 1713 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H or tetrazolyl;
or when R4 is H, then R3 is -0L2C(=0)0H or tetrazolyl;
L1 is -(CH2)m-;
I-2 iS -(CH2)m-;
L7= -(CH2),,(CHR11CH2)m(CH2)n-;
Re is -C3-C6alkyl;
R7 is -C1-C3alkyl; methyl
R9 is 1_10H;
each R11 is independently selected from -OH;
R12 is
a) an unsubstituted tetrazolyl;
b) a tetrazolyl substituted with -L1C(=0)0H;
or
c) a 5-6 membered heterocycloalkyl having 1 to 2 heteroatoms independently
selected from N and 0 substituted with -C(=0)0H;
each m is independently selected from 1, 2, 3, and 4,
and
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
4
each n is independently selected from 0, 1, 2, 3, and 4.
In certain embodiments of the compounds of Formula (la) or Formula (lb),
R1 is -NHRe or -NHCHReR9;
R3 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -
HN-N
)2,,-1\1µ2Ni
CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -L7C(=0)0H, -0L2C(=0)0H, or
R4 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H,
CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H, or
N-NH
N .
where, when R3 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H,
N:N
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H, or
or when R4 is H, then R3 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
HN-N!
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0Hõ
N-N1FNI
\41\i' =
L1 is -(CH2)m-;
1_2 is -(CH2)m-;
L7 = -(CH2)m(CHRliCH2WCF12)n-;
R6 is -C3-Cealkyl;
R7 is -C1-C3alkyl;
R9 is 1_10H;
each R11 is independently selected from -OH;
R12 is
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
HN-N,N N-NH
N41\iN
a) or
0
HO-1
N"--N
b) \--"N
or
N _ _2 N _ _
H )04012 H )=== co H
N _2
5 each m is independently selected from 1, 2, 3, and 4,
and
each n is independently selected from 0, 1, 2, 3, and 4.
In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb),
R1 is -NHFie or -NHCHREF19;
HN-N N-NH
I:NI
R3 is H, -0L2C(=0)0H, N4 N
R4 is H, -0L2R12,
-C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H,
HN
N-NH
32; N:N
CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, or
where, when R3 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
HN-N N-NH
N:N
C(=0)0H, -L2C(=0)0H, -L7C(=0)0H, or
HN-N, N-NH
\&'Ns'N \41\1:N
or when R4 is H, then 1:13 is -0L2C(=0)0H,
L1 is -(CH2)m-;
L2 is -(CH2)m-;
L7 = -(CH2WCHRliCH2WCF12)n-;
R6 is -C3-C6alkyl;
F17 is -Ci-C3alkyl;
R9 is Li OH;
each R11 is independently selected from -OH;
R12 is
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
6
HN-N N-NH
a) '32,,--LINIs:N or -\41\12N
0
HO-1
N-N
4 õ1
b) N
or
CO2H
each m is independently selected from 1, 2, 3, and 4,
and
each n is independently selected from 0, 1, 2, 3, and 4.
In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), L1
is -CH2-; L2 is -CH2- or -CH2CH2-; L7 = -(CF12)2(CHR11CH2)2-; R6 is -C4alkyl
or -05alkyl; R7 is
.. methyl, ethyl or propyl, and R11 is OH.
In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), R1
is ¨NHR6; R3 is H; R4 is -L2R12; L2 is -(CH2)m-; R6 is -03-C6alkyl;
R12 is
a) an unsubstituted tetrazolyl;
b) a tetrazolyl substituted with -1_1C(=0)0H;
or
c) a 5-6 membered heterocycloalkyl having 1 to 2 heteroatoms independently
selected from N and 0 substituted with -C(=0)0H,
and
.. each m is independently selected from 1, 2, 3, and 4.
In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), R1
is ¨NHR6; R3 is H; R4 is -L2R12; L2 is -CH2- or -CH2CH2-; R6 is -05alkyl; and
R12 is
HN-N N-NH
a) NciN:N or Nil'
0
N-N
N
b) N
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
7
or
CO2H N CO2H N "CO2H
or I
In certain embodiments of the compounds of Formula (1), Formula (la) and
Formula (1b), R1
is -NHF16; R3 is H; R4 is -L2R12; L2 is -CH2- or -CH2CH2-; R6 is -05alkyl; and
R12 is
HN-N N-NH
a) -% or -%
or
N CO2H N CO2H N 1CO2H
or I
In certain embodiments of the compounds of Formula (1), Formula (la) and
Formula (lb), R1
is -NFIR6; R3 is H; R4 is -L2C(=0)0H; L2 is -CH2- or -CH2CH2-; and R6 is -
05alkyl.
In certain embodiments of the compounds of Formula (1), Formula (la) and
Formula (lb) the
compound is selected from:
5-(5-((1H-tetrazol-5-yl)methoxy)-2-methoxybenzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-2,4-diamine;
5-(5-((1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-
2,4-diamine;
(S)-2-((5-(5-((1H-tetrazol-5-yOmethyl)-2-methoxybenzyl)-2-amino-5H-pyrrolo[3,2-
d]pyrimidin-4-y1)amino)hexan-1-01;
(S)-2-((5-(5-((1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-2-amino-5H-
pyrrolo[3,2-
90 d]pyrimidin-4-yl)amino)pentan-1-ol;
5-(5-(2-(1H-tetrazol-5-ypethyl)-2-nnethoxybenzy1)-N4-pentyl-5H-pyrrolo[3,2-
d]pyriniidine-
2,4-diamine;
N4-hexy1-5-(2-methoxy-5-(1H-tetrazol-5-y1)benzyl)-5H-pyrrolo[3,2-d]pyrimidine-
2,4-
diamine;
95 (S)-2-((2-am ino-5-(2-methoxy-5-(1H-tetrazol-5-yl)benzyl)-5H-pyrrolo[3,2-
d]pyrim idin-4-
yl)amino)hexan-1-ol;
2-(5-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxybenzyl)-2H-tetrazol-2-ypacetic acid;
5-(2-methoxy-4-(1H-tetrazol-5-yl)benzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrinnidine-2,4-
30 diamine;
5-(2-methoxy-5-(1H-tetrazol-5-yl)benzyl)-N4-pentyl-5H-pyrrolo[3,2-d]pyrimidine-
2,4-
diamine;
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
8
5-(5-(2-(1H-tetrazol-5-y0propan-2-y1)-2-methoxybenzyl)-N4-pentyl-5H-
pyrrolo[3,2-
d]pyrimidine-2,4-diamine;
(S)-2-((5-(5-(2-(1H-tetrazol-5-yl)propan-2-y1)-2-methoxybenzy1)-2-amino-5H-
pyrrolo[3,2-
d]pyrimidin-4-y1)amino)hexan-1-ol;
5-(5-(difluoro(1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-N4-pentyl-5H-
pyrrolo[3,2-
d]pyrimidine-2,4-diamine;
2-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxypheny1)-2,2-difluoroacetic acid;
(3R,5R)-7-(3-((2-amino-4-(pentylam ino)-5H-pyrrolo[3,2-d]pyrim idin-5-yl)m
ethyl)-4-
methoxyphenyI)-3,5-dihydroxyheptanoic acid;
2-(34(2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenypacetic acid;
(S)-2-(3-((2-amino-4-((1-hydroxyhexan-2-ypamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-methoxyphenypacetic acid;
3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxybenzoic
acid;
(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenyl)phosphonic acid;
methyl hydrogen (3-((2-am ino-4-(pentylam ino)-5H-pyrrolo[3,2-d]pyrim idin-5-
yOmethyl)-4-
methoxyphenyl)phosphonate;
(E)-3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenypacrylic acid;
3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenyl)propanoic acid;
(S)-3-(3-((2-amino-4-((1-hydroxyhexan-2-yl)amino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-methoxyphenyl)propanoic acid;
(S)-1-(4-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yOmethyl)-3-
methoxybenzyppyrrolidine-2-carboxylic acid ;
(R)-1-(3-((2-am ino-4-(pentylam ino)-5H-pyrrolo[3,2-d]pyrim idin-5-yl)methyl)-
4-
methoxybenzyl)pyrrolidine-2-carboxylic acid, and
2-(4-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-3-
methoxyphenoxy)acetic acid.
In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb) the
compound is selected from:
2-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenyl)acetic acid ;
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
9
3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxybenzoic
acid;
5-(5-((1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-
2,4-diamine;
5-(5-(2-(1H-tetrazol-5-ypethyl)-2-methoxybenzy1)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-
2,4-diamine;
(S)-1-(4-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-3-
methoxybenzyl)pyrrolidine-2-carboxylic acid, and
(R)-1-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxybenzyl)pyrrolidine-2-carboxylic acid.
Another aspect, provided herein are methods of using compounds of Formula (I),
Formula
(la) and Formula (lb), and pharmaceutical compositions comprising such
compounds of
Formula (I), Formula (la) and Formula (lb).
Another aspect provided herein are pharmaceutical compositions that include a
.. therapeutically effective amount of a compound of Formula (I), Formula (la)
or Formula (lb), and
a pharmaceutically acceptable carrier. In certain embodiments of such
pharmaceutical
compositions, the pharmaceutical composition is formulated for intravenous
administration,
intravitrial administration, intramuscular administration, oral
administration, rectal administration
inhalation, nasal administration, topical administration, ophthalmic
administration or otic
administration. In other embodiments, the pharmaceutical compositions are in
the form of a
tablet, a pill, a capsule, a liquid, an inhalant, a nasal spray solution, a
suppository, a solution, an
emulsion, an ointment, eye drop or ear drop. In other embodiments, such
pharmaceutical
compositions further include one or more additional therapeutic agents.
Another aspect provided herein are medicaments for treating a patient with a
disease or
disorder associated with TLR7 receptor activity, and such medicaments include
a
therapeutically effective amount of a compound of Formula (I), Formula (la) or
Formula (lb).
Another aspect provided herein is the use of a compound of Formula (I),
Formula (la) or
Formula (lb) in the manufacture of a medicament for treating a disease or
disorder associated
with TLR7 activity. In certain embodiments of such uses the disease is an
infectious disease, a
.. viral infectious disease, an inflammatory disease, a respiratory disease, a
dermatological
disease, an autoimmune disease, a cell-proliferative disease or cancer. In
certain embodiments
of such uses the disease is asthma, chronic obstructive pulmonary disease
(CORD), adult
respiratory distress syndrome (ARDS), ulcerative colitis, Crohn's disease,
bronchitis, dermatitis,
actinic keratosis, basal cell carcinoma, bladder cancer, allergic rhinitis,
psoriasis, scleroderma,
urticaria, rheumatoid arthritis, multiple sclerosis, cancer, breast cancer,
HIV, hepatitis, hepatitis
C or lupus. In certain embodiments of such uses the disease is hepatitis B,
hepatitis C,
colorectal cancer or hepatocellular carcinoma.
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
Another aspect provided herein are methods for activating a TLR7 receptor,
wherein the
method includes administering to a system or a subject in need thereof, a
therapeutically
5 effective amount of a compound of Formula (I), Formula (la) or Formula
(lb), or
pharmaceutically acceptable salts or pharmaceutical compositions thereof,
thereby activating
the TLR receptor. In certain embodiments of such methods, the methods include
administering
the compound to a cell or tissue system or to a human or animal subject.
Another aspect provided herein are methods for treating a disease or disorder
associated
10 .. with TLR7 activity , wherein the method includes administering to a
system or subject in need of
such treatment an effective amount of a compound of Formula (I), Formula (la)
or Formula (lb),
or pharmaceutically acceptable salts thereof, thereby treating the disease or
disorder. In certain
embodiments of such methods, the methods include administering the compound to
a cell or
tissue system or to a human or animal subject. In certain embodiments of such
methods, the
disease or condition is an infectious disease, a viral infectious disease, an
inflammatory
disease, a respiratory disease, a dermatological disease, an autoimmune
disease, a cell-
proliferative disease or cancer. In certain embodiments of such methods, the
disease or
condition is asthma, chronic obstructive pulmonary disease (CORD), adult
respiratory distress
syndrome (ARDS), ulcerative colitis, Crohn's disease, bronchitis, dermatitis,
actinic keratosis,
basal cell carcinoma, bladder cancer, allergic rhinitis, psoriasis,
scleroderma, urticaria,
rheumatoid arthritis, multiple sclerosis, cancer, breast cancer, HIV,
hepatitis, hepatitis C or
lupus. In certain embodiments of such methods the disease is hepatitis B,
hepatitis C, colorectal
cancer or hepatocellular carcinoma.
Another aspect provided herein are methods for treating a cell-proliferative
disease,
comprising administering to a system or subject in need of such treatment an
effective amount
of a compound of Formula (I), Formula (la) or Formula (lb), or
pharmaceutically acceptable
salts thereof; wherein the cell-proliferative disease is bladder cancer,
lymphoma, osteosarcoma,
melanoma, or a tumor of breast, renal, prostate, colorectal, thyroid, ovarian,
pancreatic,
neuronal, lung, uterine or gastrointestinal tumor. In certain embodiments of
such methods the
cell proliferative disease is colorectal cancer or hepatocellular carcinoma.
In certain
embodiments of such methods the cell proliferative disease is colorectal
cancer. In certain
embodiments of such methods the cell proliferative disease is hepatocellular
carcinoma.
Another aspect provided herein are compounds for use in a method of medical
treatment,
wherein the method of medical treatment is for treating a disease associated
with TLR7
receptor activity, wherein the disease is selected from an infectious disease,
a viral infectious
disease, an inflammatory disease, a respiratory disease, a dermatological
disease, an
autoimmune disease, a cell-proliferative disease or cancer, and wherein the
compound is a
compound of Formula (I), Formula (la) or Formula (lb). In certain embodiments
of such
,81800293
11
methods, the disease or condition is asthma, chronic obstructive pulmonary
disease
(COPD), adult respiratory distress syndrome (ARDS), ulcerative colitis,
Crohn's
disease, bronchitis, dermatitis, actinic keratosis, basal cell carcinoma,
bladder
cancer, allergic rhinitis, psoriasis, scleroderma, urticaria, rheumatoid
arthritis, multiple
sclerosis, cancer, breast cancer, HIV, hepatitis, hepatitis C or lupus. In
certain
embodiments of such methods the disease is hepatitis B, hepatitis C,
colorectal
cancer or hepatocellular carcinoma.
Another aspect provided herein is use of an effective amount of a compound as
described herein for treating a disease or disorder associated with TLR7
activity, in a
system or subject in need of such treatment, wherein the disease is an
infectious
disease, a viral infectious disease, an inflammatory disease, a respiratory
disease, a
dermatological disease, an autoimmune disease, a cell-proliferative disease or
cancer.
Another aspect provided herein is a compound for use in treating a disease
associated with TLR7 receptor activity, wherein the disease is an infectious
disease,
a viral infectious disease, an inflammatory disease, a respiratory disease, a
dermatological disease, an autoimmune disease, a cell-proliferative disease or
cancer, and wherein the compound is as described herein.
DETAILED DESCRIPTION OF THE INVENTION
Definitions
The term "alkyl," as used herein, refers to a saturated branched or straight
chain
hydrocarbon. In certain embodiments such alkyl groups are optionally
substituted. As
used herein, the terms "C1-C3alkyl", "Cl-Caalkyl", "C1-C8alkyl", "C1-C8alkyl",
"C1-C7alkyl" and "C1-C8alkyl" refer to an alkyl group containing at least 1,
and at most
3, 4, 5, 6, 7 or 8 carbon atoms, respectively. If not otherwise specified, an
alkyl group
generally is a C1-C8 alkyl. Non-limiting examples of alkyl groups as used
herein
include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, t-
butyl, n-pentyl,
isopentyl, hexyl, heptyl, octyl, nonyl, decyl and the like.
CA 2946408 2019-09-27
. ,81800293
11a
The term "heteroatom," as used herein, refers to nitrogen (N), oxygen (0) or
sulfur (S) atoms.
The term "heterocycloalkyl," as used herein refers to a to saturated 3-6
membered monocyclic hydrocarbon ring structure, a to saturated 5-6 membered
monocyclic hydrocarbon ring structure, a saturated 6-9 membered fused bicyclic
hydrocarbon ring structure, or a saturated 10-14 membered fused tricyclic
hydrocarbon ring structure, wherein one to four of the ring carbons of the
hydrocarbon ring structure are replaced by one to four groups independently
selected
from -0-, -NR-, or -S-, wherein R is hydrogen, C1-C4alkyl or an amino
protecting
group.
Non-limiting examples of heterocycloalkyl groups, as used herein, include
aziridinyl, aziridin-1-yl, aziridin-2-yl, aziridin-3-yl, oxiranyl, oxiran-2-
yl, oxiran-3-yl,
thiiranyl, thiiran-2-yl, thiiran-3-yl, azetadinyl, azetadin-1-yl, azetadin-2-
yl, azetadin-3-
yl, oxetanyl, oxetan-2-yl, oxetan-3-yl, oxetan-4-yl, thietanyl, thietan-2-yl,
thietan-3-yl,
thietan-4-yl, pyrrolidinyl, pyrrolidin-1 -yl, pyrrolidin-2-yl, pyrrolidin-3-
yl, pyrrolidin-4-yl,
pyrrolidin-5-yl, tetrahydrofuranyl, tetrahydrofuran-2-yl, tetrahydrofuran-3-
yl,
tetrahydrofuran-4-yl, tetrahydrofuran-5-yl, tetrahydrothienyl, tetrahydrothien-
2-yl,
tetrahydrothien-3-yl, tetrahydrothien-4-yl, tetrahydrothien-5-yl, piperidinyl,
piperidin-1-
yl, piperidin-2-yl, piperidin-3-yl, piperidin-4-yl, piperidin-5-yl, piperidin-
6-yl,
tetrahydropyranyl, tetrahydropyran-2-yl, tetrahydropyran-3-yl, tetrahydropyran-
4-yl,
tetrahydropyran-5-yl, tetrahydropyran-6-yl, tetrahydrothiopyranyl,
tetrahydrothiopyran-
2-yl, tetrahydrothiopyran-3-yl, tetrahydrothiopyran-4-yl, tetrahydrothiopyran-
5-yl,
tetrahydrothiopyran-6-yl, piperazinyl, piperazin-1-yl, piperazin-2-yl,
piperazin-3-yl,
piperazin-4-yl, piperazin-5-yl, piperazin-6-yl, morpholinyl, morpholin-2-yl,
morpholin-3-
yl, morpholin-4-yl, morpholin-5-yl,
CA 2946408 2019-09-27
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
12
morpholin-6-yl, thiomorpholinyl, thiomorpholin-2-yl, thiomorpholin-3-yl,
thiomorpholin-4-yl,
thiomorpholin-5-yl, thiomorpholin-6-yl, oxathianyl, oxathian-2-yl, oxathian-3-
yl, oxathian-5-yl,
oxathian-6-yl, dithianyl, dithian-2-yl, dithian-3-yl, dithian-5-yl, dithian-6-
yl, azepanyl, azepan-1-yl,
azepan-2-yl, azepan-3-yl, azepan-4-yl, azepan-5-yl, azepan-6-yl, azepan-7-yl,
oxepanyl,
oxepan-2-yl, oxepan-3-yl, oxepan-4-yl, oxepan-5-yl, oxepan-6-yl, oxepan-7-yl,
thiepanyl,
thiepan-2-yl, thiepan-3-yl, thiepan-4-yl, thiepan-5-yl, thiepan-6-yl, thiepan-
7-yl, dioxolanyl,
dioxolan-2-yl, dioxolan-4-yl, dioxolan-5-yl, thioxanyl, thioxan-2-yl, thioxan-
3-yl, thioxan-4-yl,
thioxan-5-yl, dithiolanyl, dithiolan-2-yl, dithiolan-4-yl, dithiolan-5-yl,
pyrrolinyl, pyrrolin-1-yl,
pyrrolin-2-yl, pyrrolin-3-yl, pyrrolin-4-yl, pyrrolin-5-yl, imidazolinyl,
imidazolin-1-yl, imidazolin-3-yl,
imidazolin-4-yl, imidazolin-5-yl, imidazolidinyl, imidazolidin-1-yl,
imidazolidin-2-yl, imidazolidin-3-
yl, imidazolidin-4-yl, imidazolidin-4-yl, pyrazolinyl, pyrazolin-1-yl,
pyrazolin-3-yl, pyrazolin-4-yl,
pyrazolin-5-yl, pyrazolidinyl, pyrazolidin-1-yl, pyrazolidin-2-yl, pyrazolidin-
3-yl, pyrazolidin-4-yl,
pyrazolidin-5-yl, hexahydro-1,4-diazepinyl, dihydrofuranyldihydropyranyl,
1,2,3,6-
tetrahydropyridinyl, 2H-pyranyl, 4H-pyranyl, dihydropyranyl, dihydrothienyl,
dihydrofuranyl, 3-
azabicyclo[3.1.0]hexanyl, 3-azabicyclo[4.1.0]heptanyl, pyrrolidiny1-2-one,
piperidiny1-3-one
piperidiny1-2-one, piperidiny1-4-one, and 2H-pyrroly1
The term "acceptable" with respect to a formulation, composition or
ingredient, as used
herein, means having no persistent detrimental effect on the general health of
the subject being
treated.
90 The term "administration" or "administering" of the subject compound
means providing a
compound of Formula (1) or a pharmaceutically acceptable salt thereof to a
subject in need of
treatment.
The term "cancer," as used herein refers to an abnormal growth of cells which
tend to
proliferate in an uncontrolled way and, in some cases, to metastasize
(spread). The types of
cancer include, but is not limited to, solid tumors (such as those of the
bladder, bowel, brain,
breast, endometrium, heart, kidney, lung, lymphatic tissue (lymphoma), ovary,
pancreas or
other endocrine organ (thyroid), prostate, skin (melanoma) or hematological
tumors (such as
the leukemias).
The term "carrier," as used herein, refers to chemical compounds or agents
that facilitate
the incorporation of a compound described herein into cells or tissues.
The terms "co-administration" or "combined administration" or the like as used
herein are
meant to encompass administration of the selected therapeutic agents to a
single patient, and
are intended to include treatment regimens in which the agents are not
necessarily
administered by the same route of administration or at the same time.
The term "dermatological disorder," as used herein refers to a skin disorder.
Such
dermatological disorders include, but are not limited to, proliferative or
inflammatory disorders of
the skin such as, atopic dermatitis, bullous disorders, collagenoses, contact
dermatitis eczema,
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
13
Kawasaki Disease, rosacea, Sjogren-Larsso Syndrome, actinic keratosis, basal
cell carcinoma
and urticaria.
The term "diluent," as used herein, refers to chemical compounds that are used
to dilute a
compound described herein prior to delivery. Diluents can also be used to
stabilize compounds
described herein.
The terms "effective amount" or "therapeutically effective amount," as used
herein, refer to a
sufficient amount of a compound described herein being administered which will
relieve to some
extent one or more of the symptoms of the disease or condition being treated.
The result can be
reduction and/or alleviation of the signs, symptoms, or causes of a disease,
or any other
desired alteration of a biological system. For example, an "effective amount"
for therapeutic
uses is the amount of the composition comprising a compound as disclosed
herein required to
provide a clinically significant decrease in disease symptoms. An appropriate
"effective" amount
in any individual case may be determined using techniques, such as a dose
escalation study.
The terms "enhance" or "enhancing," as used herein, means to increase or
prolong either in
potency or duration a desired effect. Thus, in regard to enhancing the effect
of therapeutic
agents, the term "enhancing" refers to the ability to increase or prolong,
either in potency or
duration, the effect of other therapeutic agents on a system. An "enhancing-
effective amount,"
as used herein, refers to an amount adequate to enhance the effect of another
therapeutic
agent in a desired system.
90 The term "excipient" refers to any essentially accessory substance that
may be present in
the finished dosage form. For example, the term "excipient" includes vehicles,
binders,
disontegrants, fillers (diluents), lubricants, suspending/dispersing agents,
and the like.
The terms "fibrosis" or "fibrosing disorder," as used herein, refers to
conditions that follow
acute or chronic inflammation and are associated with the abnormal
accumulation of cells
and/or collagen and include but are not limited to fibrosis of individual
organs or tissues such as
the heart, kidney, joints, lung, or skin, and includes such disorders as
idiopathic pulmonary
fibrosis and cryptogenic fibrosing alveolitis.
The term "iatrogenic," as used herein, means a condition, disorder, or disease
created or
worsened by medical or surgical therapy.
The term "immunologically effective amount," as used herein, means that the
administration
of a sufficient amount to an individual, either in a single dose or as part of
a series, that is
effective for treatment or prevention of an immunological disease or disorder.
This amount
varies depending upon the health and physical condition of the individual to
be treated, age, the
taxonomic group of individual to be treated (e.g. non-human primate, primate,
etc.), the capacity
of the individual's immune system to synthesize antibodies, the degree of
protection desired,
the treating doctor's assessment of the medical situation, and other relevant
factors. It is
expected that the amount will fall in a relatively broad range that can be
determined through
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
14
routine trials.
An "immunological response" or "immune response" to an antigen or composition,
as used
herein, refers to the development in a subject of a humoral and/or cellular
immune response to
the antigen or composition.
Immune responses include innate and adaptive immune responses. Innate immune
responses are fast-acting responses that provide a first line of defense for
the immune system.
In contrast, adaptive immunity uses selection and clonal expansion of immune
cells having
somatically rearranged receptor genes (e.g., T- and B-cell receptors) that
recognize antigens
from a given pathogen or disorder (e.g., a tumor), thereby providing
specificity and
immunological memory. Innate immune responses, among their many effects, lead
to a rapid
burst of inflammatory cytokines and activation of antigen-presenting cells
(APCs) such as
macrophages and dendritic cells. To distinguish pathogens from self-
components, the innate
immune system uses a variety of relatively invariable receptors that detect
signatures from
pathogens, known as pathogen-associated molecular patterns, or PAMPs. The
mechanism
behind this potentiation of the immune responses has been reported to involve
pattern-
recognition receptors (PRRs), which are differentially expressed on a variety
of immune cells,
including neutrophils, macrophages, dendritic cells, natural killer cells, B
cells and some
nonimmune cells such as epithelial and endothelial cells. Engagement of PRRs
leads to the
activation of some of these cells and their secretion of cytokines and
chemokines, as well as
maturation and migration of other cells. In tandem, this creates an
inflammatory environment
that leads to the establishment of the adaptive immune response. PRRs include
nonphagocytic
receptors, such as Toll-like receptors (TLRs) and nucleotide-binding
oligomerization domain
(NOD) proteins, and receptors that induce phagocytosis, such as scavenger
receptors,
mannose receptors and 13-glucan receptors. Dendritic cells are recognized as
some of the most
important cell types for initiating the priming of naive CD4+ helper T (TH)
cells and for inducing
CD8+ T cell differentiation into killer cells. TLR signaling has been reported
to play an important
role in determining the quality of these helper T cell responses, for
instance, with the nature of
the TLR signal determining the specific type of TH response that is observed
(e.g., TH1 versus
TH2 response). A combination of antibody (humoral) and cellular immunity are
produced as
part of a TH1-type response, whereas a TH2-type response is predominantly an
antibody
response.
A "humoral immune response" refers to an immune response mediated by antibody
molecules, while a "cellular immune response" refers to an immune response
mediated by T-
lymphocytes and/or other white blood cells. One important aspect of cellular
immunity involves
an antigen-specific response by cytolytic T-cells ("CTLs"). CTLs have
specificity for peptide
antigens that are presented in association with proteins encoded by the major
histocompatibility
complex (MHC) and expressed on the surfaces of cells. CTLs help induce and
promote the
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
intracellular destruction of intracellular microbes, or the lysis of cells
infected with such
microbes. Another aspect of cellular immunity involves an antigen-specific
response by helper
1-cells. Helper T-cells act to help stimulate the function, and focus the
activity of, nonspecific
effector cells against cells displaying peptide antigens in association with
MHC molecules on
5 their surface. A "cellular immune response" also refers to the production
of cytokines,
chemokines and other such molecules produced by activated 1-cells and/or other
white blood
cells, including those derived from CD4+ and CD8+ 1-cells.
The term "inflammatory disorders," as used herein, refers to those diseases or
conditions
that are characterized by one or more of the signs of pain (dolor, from the
generation of noxious
10 substances and the stimulation of nerves), heat (calor, from
vasodilatation), redness (rubor,
from vasodilatation and increased blood flow), swelling (tumor, from excessive
inflow or
restricted outflow of fluid), and loss of function (functio laesa, which may
be partial or complete,
temporary or permanent). Inflammation takes many forms and includes, but is
not limited to,
inflammation that is one or more of the following: acute, adhesive, atrophic,
catarrhal, chronic,
15 cirrhotic, diffuse, disseminated, exudative, fibrinous, fibrosing, focal,
granulomatous,
hyperplastic, hypertrophic, interstitial, metastatic, necrotic, obliterative,
parenchymatous, plastic,
productive, proliferous, pseudomembranous, purulent, sclerosing, seroplastic,
serous, simple,
specific, subacute, suppurative, toxic, traumatic, and/or ulcerative.
Inflammatory disorders
further include, without being limited to those affecting the blood vessels
(polyarteritis, temporal
arthritis); joints (arthritis: crystalline, osteo-, psoriatic, reactive,
rheumatoid, Reiter's);
gastrointestinal tract (Disease,); skin (dermatitis); or multiple organs and
tissues (systemic lupus
erythematosus).
The terms "ocular disease" or "ophthalmic disease," as used herein, refer to
diseases which
affect the eye or eyes and potentially the surrounding tissues as well. Ocular
or ophthalmic
.. diseases include, but are not limited to, conjunctivitis, retinitis,
scleritis, uveitis, allergic
conjuctivitis, vernal conjunctivitis, papillary conjunctivitis and
cytomegalovirus (CMV) retinitis.
The term "pharmaceutically acceptable," as used herein, refers a material,
such as a carrier
or diluent, which does not abrogate the biological activity or properties of
the compounds
described herein. Such materials are administered to an individual without
causing undesirable
biological effects or interacting in a deleterious manner with any of the
components of the
composition in which it is contained.
The term "pharmaceutically acceptable salt," as used herein, refers to a
formulation of a
compound that does not cause significant irritation to an organism to which it
is administered
and does not abrogate the biological activity and properties of the compounds
described herein.
The terms "combination" or "pharmaceutical combination," as used herein mean a
product
that results from the mixing or combining of more than one active ingredient
and includes both
fixed and non-fixed combinations of the active ingredients. The term "fixed
combination" means
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
16
that the active ingredients, by way of example, a compound of Formula (I) and
an additional
therapeutic agent, are both administered to a patient simultaneously in the
form of a single
entity or dosage. The term "non-fixed combination" means that the active
ingredients, by way of
example, a compound of Formula (I) and an additional therapeutic agent, are
both administered
to a patient as separate entities either simultaneously, concurrently or
sequentially with no
specific time limits, wherein such administration provides therapeutically
effective levels of the 2
compounds in the body of the patient. The latter also applies to cocktail
therapy, e.g. the
administration of 3 or more active ingredients.
The terms "composition" or "pharmaceutical composition," as used herein,
refers to a
mixture a compound of Formula (I), Formula (la) or Formula (lb), or
pharmaceutically
acceptable salt thereof, with at least one and optionally more than one other
pharmaceutically
acceptable chemical components, such as carriers, stabilizers, diluents,
dispersing agents,
suspending agents, thickening agents, and/or excipients.
The term "respiratory disease," as used herein, refers to diseases affecting
the organs that
.. are involved in breathing, such as the nose, throat, larynx, trachea,
bronchi, and lungs.
Respiratory diseases include, but are not limited to, asthma, adult
respiratory distress syndrome
and allergic (extrinsic) asthma, non-allergic (intrinsic) asthma, acute severe
asthma, chronic
asthma, clinical asthma, nocturnal asthma, allergen-induced asthma, aspirin-
sensitive asthma,
exercise-induced asthma, isocapnic hyperventilation, child-onset asthma, adult-
onset asthma,
cough-variant asthma, occupational asthma, steroid-resistant asthma, seasonal
asthma,
seasonal allergic rhinitis, perennial allergic rhinitis, chronic obstructive
pulmonary disease,
including chronic bronchitis or emphysema, pulmonary hypertension,
interstitial lung fibrosis
and/or airway inflammation and cystic fibrosis, and hypoxia.
The term "subject" or "patient," as used herein, encompasses mammals and non-
mammals.
Examples of mammals include, but are not limited to, humans, chimpanzees, apes
monkeys,
cattle, horses, sheep, goats, swine; rabbits, dogs, cats, rats, mice, guinea
pigs, and the like.
Examples of non-mammals include, but are not limited to, birds, fish and the
like. Frequently
the subject is a human, and may be a human who has been diagnosed as in need
of treatment
for a disease or disorder disclosed herein.
The term "TLR7 agonist," as used herein, refers to a compound which activates
a TLR7
receptor.
The term "TLR7 disease" or a "disease or disorder associated with TLR7
activity," as used
herein, refers to any disease state associated with a toll-like receptor. Such
diseases or
disorders include, but are not limited to, an infectious disease, a viral
infectious disease, an
inflammatory disease, a respiratory disease, a dermatological disease, an
autoimmune disease,
a cell-proliferative disease and cancer, such as, by way of example only,
asthma, chronic
obstructive pulmonary disease (COPD), adult respiratory distress syndrome
(ARDS), ulcerative
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
17
colitis, Crohn's disease, bronchitis, dermatitis, actinic keratosis, basal
cell carcinoma, bladder
cancer, allergic rhinitis, psoriasis, scleroderma, urticaria, rheumatoid
arthritis, multiple sclerosis,
cancer, breast cancer, lymphoma, osteosarcoma, melanoma, breast cancer, renal
cancer,
prostate cancer, colorectal cancer, thyroid cancer, ovarian cancer, pancreatic
cancer, neuronal
cancer, lung, uterine cancer gastrointestinal cancer,HIV, hepatitis, hepatitis
B, hepatitis C,
hepatocellular carcinoma or lupus.
The term "therapeutically effective amount," as used herein, refers to any
amount of a
compound which, as compared to a corresponding subject who has not received
such amount,
results in improved treatment, healing, prevention, or amelioration of a
disease, disorder, or
side effect, or a decrease in the rate of advancement of a disease or
disorder. The term also
includes within its scope amounts effective to enhance normal physiological
function.
The terms "treat," "treating" or "treatment," as used herein, refers to
methods of alleviating,
abating or ameliorating a disease or condition symptoms, preventing additional
symptoms,
ameliorating or preventing the underlying metabolic causes of symptoms,
inhibiting the disease
or condition, arresting the development of the disease or condition, relieving
the disease or
condition, causing regression of the disease or condition, relieving a
condition caused by the
disease or condition, or stopping the symptoms of the disease or condition
either
prophylactically and/or therapeutically.
The compound names were obtained using Chem Draw Ultra 10.0 (CambridgeSoft )
or
JChem version 5.2.2 (ChemAxon).
Description of the Preferred Embodiments
Provided herein are compounds and pharmaceutical compositions thereof, which
are
agonists of toll-like receptor-7 (TLR7). Also are compounds, pharmaceutical
compositions and
methods for the treatment of diseases and/or disorders associated with TLR7
activity.
The TLR7 agonists of the invention are compounds having the structure of
Formula (I), and
pharmaceutically acceptable salts, individual isomers and mixture of isomers
thereof:
¨o
4. R3
N N
R4
H,N
Formula (I)
wherein:
Al is ¨NHFie or ¨NHCHR61:19;
R3 is H, -L2R12, -0L2R12, -C(CH3)2R12, -R(=0)(OH)2, -R(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -
CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
18
R4 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -
CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -L7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
where, when R3 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
or when R4 is H, then R3 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
L1 is -(CH2)rn-;
L2 is -(CH2)rn-;
L7 = -(CF12)m(CHR11CH2)m(CH2)n-;
R6 is -C3-C6alkyl;
R7 is -C1-C3alkyl;
R9 is Li OH;
each R11 is independently selected from -OH;
R12 is
a) an unsubstituted tetrazolyl;
b) a tetrazolyl substituted with -Li C(=0)0H;
or
c) a 5-6 membered heterocycloalkyl having 1 to 2 heteroatoms independently
selected from N and 0 substituted with -C(=0)0H;
each m is independently selected from 1, 2, 3, and 4,
and
each n is independently selected from 0, 1, 2, 3, and 4.
95 In certain
embodiments, the TLR7 agonists of the invention are compounds having the
structure of Formula (la) or Formula (lb):
-0 -0
R1 R3 R1
N 41R4
H2N N 1-12N N
Formula (la) Formula (lb),
where R1, R3 and R4 are as defined herein.
The compounds of Formula (I), Formula (la) and Formula (lb) provided herein,
and the
pharmaceutically acceptable salts thereof, also includes all suitable isotopic
variations of such
compounds, and pharmaceutically acceptable salts. An isotopic variation of a
compound or a
pharmaceutically acceptable salt thereof is defined as one in which at least
one atom is
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
19
replaced by an atom having the same atomic number but an atomic mass different
from the
atomic mass usually found in nature. Examples of isotopes that may be
incorporated into the
compounds and pharmaceutically acceptable salts thereof include but are not
limited to
isotopes of hydrogen, carbon, nitrogen and oxygen such as 2H5 3H5 1105 1305
1405 15N5 1705 1805
35s, 18.-,
r 36CI and 1231. Certain isotopic variations of the compounds and
pharmaceutically
acceptable salts thereof, for example, those in which a radioactive isotope
such as 3H or 14C is
incorporated, are useful in drug and/or substrate tissue distribution studies.
In particular
examples, 3H and 14C isotopes may be used for their ease of preparation and
detectability. In
other examples, substitution with isotopes such as 2H may afford certain
therapeutic
advantages resulting from greater metabolic stability, such as increased in
vivo half-life or
reduced dosage requirements. Isotopic variations of the compounds, and
pharmaceutically
acceptable salts
and isomers thereof, are prepared by conventional procedures using appropriate
isotopic
variations of suitable reagents.
Processes for Making Compounds of Formula (I)
General procedures for preparing compounds of Formula (I), and subformulae
thereof, are
described in the Examples, infra. In the reactions described, reactive
functional groups, for
example hydroxy, amino, imino, thio or carboxy groups, where these are desired
in the final
product, may be protected to avoid their unwanted participation in the
reactions. Conventional
protecting groups may be used in accordance with standard practice (see e.g.,
T.W. Greene
and P. G. M. Wuts in "Protective Groups in Organic Chemistry," John Wiley and
Sons, 1991).
In certain embodiments, the compounds of Formula (I), and subformulae thereof,
are
prepared as a pharmaceutically acceptable acid addition salt by reacting the
free base form of
.. the compound of Formula (I), Formula (la) or Formula (lb) with a
pharmaceutically acceptable
organic acid or inorganic acid. In other embodiments, a pharmaceutically
acceptable base
addition salt of compounds of Formula (I), Formula (la) or Formula (lb) is
prepared by reacting
the free acid form of the compound of Formula (I), Formula (la) or Formula
(lb) with a
pharmaceutically acceptable organic base or inorganic base. Alternatively, the
salt forms of the
compounds of Formula (I), Formula (la) or Formula (lb) are prepared using
salts of the starting
materials or intermediates. In certain embodiments, the compounds of Formula
(I), Formula (la)
or Formula (lb) are in the form of other salts including, but not limited to,
oxalates and
trifluoroacetates. In certain embodiments, hem isalts of acids and bases are
formed, for
example, hemisulphate and hemicalcium salts.
Such pharmaceutically acceptable acid addition salts of compounds of Formula
(I), Formula
(la) or Formula (lb) include, but are not limited to, a hydrobromide,
hydrochloride, sulfate,
nitrate, succinate, maleate, formate, acetate, adipate, besylatye,
bicarbonate/carbonate,
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
propionate, fumarate, citrate, tartrate, lactate, benzoate, salicylate,
glutamate, aspartate, p-
toluenesulfonate, benzenesulfonate, nnethanesulfonate, ethanesulfonate,
naphthalenesulfonate
(e.g. 2-naphthalenesulfonate), hexanoate salt, bisulphate/sulphate, borate,
camsylate,
cyclamate, edisylate, esylate, gluceptate, gluconate, glucuronate,
hexafluorophosphate,
5 hibenzate, hydrochloride/chloride, hydrobromide/bromide, hydroiodide/iodide,
isethionate,
lactate, malate, malonate, mesylate, methylsulphate, naphthylate, 2-napsylate,
nicotinate,
orotate, oxalate, palm itate, pamoate, phosphate/hydrogen phosphate/dihydrogen
phosphate,
pyroglutamate, saccharate, stearate, tannate, tosylate, trifluoroacetate and
xinofoate salts.
The organic acid or inorganic acids used to form certain pharmaceutically
acceptable acid
10 addition salts of compounds of Formula (I), Formula (la) or Formula (lb)
include, but are not
limited to, hydrobromic, hydrochloric, sulfuric, nitric, phosphoric, succinic,
maleic, formic, acetic,
propionic, fumaric, citric, tartaric, lactic, benzoic, salicylic, glutamic,
aspartic, p-toluenesulfonic,
benzenesulfonic, methanesulfonic, ethanesulfonic, naphthalenesulfonic such as
2-
naphthalenesulfonic, or hexanoic acid.
15 Such pharmaceutically acceptable base addition salt of a compound of
Formula (I), Formula
(la) or Formula (lb) include, but are not limited to, aluminium, arginine,
benzathine, calcium,
choline, diethylamine, diolamine, glycine, lysine, magnesium, meglumine,
olamine, potassium,
sodium, tromethamine and zinc salts.
In certain embodiments, the free acid or free base forms of the compounds of
Formula (I)
20 are prepared from the corresponding base addition salt or acid addition
salt from, respectively.
For example a compound Formula (I), Formula (la) or Formula (lb) in an acid
addition salt form
is converted to the corresponding free base by treating with a suitable base
(by way of example
only, an ammonium hydroxide solution, a sodium hydroxide, and the like). For
example, a
compound of Formula (I), Formula (la) or Formula (lb) in a base addition salt
form is converted
to the corresponding free acid by treating with a suitable acid (by way of
example only,
hydrochloric acid).
In certain embodiments, compounds of Formula (I), Formula (la) or Formula (lb)
are
prepared as protected derivatives using methods known to those of ordinary
skill in the art. A
detailed description of the techniques applicable to the creation of
protecting groups and their
removal can be found in T. W. Greene, "Protecting Groups in Organic
Chemistry," 3rd edition,
John Wiley and Sons, Inc., 1999.
In certain embodiments, compounds of Formula (I), Formula (la) and Formula
(lb) are
prepared as their individual stereoisomers. In other embodiments, the
compounds of Formula
(I), Formula (la) and Formula (lb) are prepared as their individual
stereoisomers by reacting a
racemic mixture of the compound with an optically active resolving agent to
form a pair of
diastereoisomeric compounds, separating the diastereomers and recovering the
optically pure
enantionners. In certain embodiments, resolution of enantiomers is carried out
using covalent
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
21
diastereomeric derivatives of the compounds of Formula (I), Formula (la) or
Formula (lb), or by
using dissociable complexes (e.g., crystalline diastereomeric salts).
Diastereomers have
distinct physical properties (e.g., melting points, boiling points,
solubility, reactivity, etc.) and are
readily separated by taking advantage of these dissimilarities. In certain
embodiments, the
.. diastereomers are separated by chromatography, or by separation/resolution
techniques based
upon differences in solubility. The optically pure enantiomer is then
recovered, along with the
resolving agent, by any practical means that would not result in racemization.
A more detailed
description of the techniques applicable to the resolution of stereoisomers of
compounds from
their racemic mixture can be found in Jean Jacques, Andre Collet, Samuel H.
Wilen,
"Enantiomers, Racemates and Resolutions," John Wiley And Sons, Inc., 1981.
A non-limiting example of a synthetic scheme used to make compounds of Formula
(I) is
illustrated in reaction scheme (I) below.
Scheme (I)
Me0 ilk Fe
Me0 Ai R3 Me0 Ati R3
CI lir VIT:511 Br R4 CI "F R4 "F R4
N),x5s,
R-NH2 R,NH
I-12N N ii õk / /
alkylation H2N N SNAr H2N N
Scheme (I) illustrates the synthesis of pyrrolopyrimidines of Formula (I)
using a two-step
scheme starting with commercially available 4-chloro-5H-pyrrolo[3,2-
d]pyrimidin-2-amine.
Alkylation at the N-5 position by a benzylbromide (or benzylchloride) analog
gives the
corresponding 5-benzyl derivatives, where R3 and R4 are as defined herein.
Subsequent SNAr
substitution of the chloro group by an alkyl amine derivative affords the
corresponding 4,5-
disubstituted pyrrolopyrimidine, where R is R6 or ¨CHR6R9 and R6 and R9 are as
defined herein.
Pharmacology and Utility
When a foreign antigen challenges the immune system it responds by launching a
protective response that is characterized by the coordinated interaction of
both the innate and
acquired immune systems. These two interdependent systems fulfill two mutually
exclusive
requirements: speed (contributed by the innate system) and specificity
(contributed by the
adaptive system).
The innate immune system serves as the first line of defense against invading
pathogens,
holding the pathogen in check while the adaptive responses are matured. It is
triggered within
minutes of infection in an antigen-independent fashion, responding to broadly
conserved
patterns in the pathogens (though it is not non-specific, and can distinguish
between self and
pathogens). Crucially, it also generates the inflammatory and co-stimulatory
milieu (sometimes
referred to as the danger signal) that potentiates the adaptive immune system
and steers (or
polarizes it) towards the cellular or humoral responses most appropriate for
combating the
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
22
infectious agent. The development of TLR modulators for therapeutic targeting
of innate
immunity has been reviewed (see Nature Medicine, 2007, 13, 552-559; Drug
Discovery Today:
Therapeutic Stategies, 2006, 3, 343-352 and Journal of Immunology, 2005, 174,
1259-1268).
The adaptive response becomes effective over days or weeks, but ultimately
provides the
fine antigenic specificity required for complete elimination of the pathogen
and the generation of
immunologic memory. It is mediated principally by T and B cells that have
undergone germline
gene rearrangement and are characterized by specificity and long-lasting
memory. However, it
also involves the recruitment of elements of the innate immune system,
including professional
phagocytes (macrophages, neutrophils etc.) and granulocytes (basophils,
eosinophils etc.) that
engulf bacteria and even relatively large protozoal parasites. Once an
adaptive immune
response has matured, subsequent exposure to the pathogen results in its rapid
elimination due
to highly specific memory cells have been generated that are rapidly activated
upon subsequent
exposure to their cognate antigen.
Autoimmune diseases, are defined by (i) humoral or autoantibody response to a
self
antigen (by way of example only, Graves' primary hyperthyroidism with
antibodies to the TSH
receptor), or (ii) cellular response wherein immune cells destroy nonimmune
cells from which
the self-antigen is derived (by way of example only, the thyrocyte
(Hashimoto's thyroiditis) or
pancreatic -islet cell (Type 1 diabetes). Many autoimmune diseases are a
combination of both
phenomena, for instance, Hashimoto's and Type 1 diabetes also have auto-
antibodies, anti-
thyroid peroxidase (TPO) or anti-glutamic acid decarboxylase (GAD)/Islet Cell.
Autoimmune
diseases often have an inflammatory component including, but not limited to,
increases in
adhesion molecules (by way of example only, vascular cell adhesion molecule-1
(VCAM-1), and
altered leukocyte adhesion to the vasculature such as, by way of example only,
colitis, systemic
lupus, systemic sclerosis, and the vascular complications of diabetes.
Toll-like receptors (TLRs) are type-I transmembrane proteins characterized by
an
extracellular N-terminal leucine-rich repeat (LRR) domain, followed by a
cysteine-rich region, a
TM domain, and an intracellular (cytoplasmic) tail that contains a conserved
region named the
Toll/IL-1 receptor (TIR) domain. TLRs are pattern recognition receptors (PRR)
that are
expressed predominantly on immune cells including, but not limited to,
dendritic cells, T
lymphocytes, macrophages, monocytes and natural killer cells. The LLR domain
is important for
ligand binding and associated signaling and is a common feature of PRRs. The
TIR domain is
important in protein-protein interactions and is associated with innate
immunity. The TIR domain
also unites a larger IL-1 RiTLR superfamily that is composed of three
subgroups. Members of
the first group possess immunoglobin domains in their extracellular regions
and include IL-1
and IL-18 receptors and accessory proteins as well as ST2. The second group
encompasses
the TLRs. The third group includes intracellular adaptor proteins important
for signaling.
TLRs are a group of pattern recognition receptors which bind to pathogen-
associated
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
23
molecular patterns (RAMPS) from bacteria, fungi, protozoa and viruses, and act
as a first line of
defense against invading pathogens. TLRs are essential to induce expression of
genes
involved in inflammatory responses, and TLRs and the innate immune system are
a critical step
in the development of antigen-specific acquired immunity.
Adaptive (humoral or cell-mediated) immunity is associated with the TLR signal
mechanism
of innate immunity. Innate immunity is a protective immune cell response that
functions rapidly
to fight environmental insults including, but not limited to, bacterial or
viral agents. Adaptive
immunity is a slower response, which involves differentiation and activation
of naive T
lymphocytes into T helper 1 (Th1) or T helper 2 (Th2) cell types. Th1 cells
mainly promote
cellular immunity, whereas Th2 cells mainly promote humoral immunity. Though
primarily a host
protective system, pathologic expression of the innate immunity signals
emanating from the
TLR pathway are implicated in initiating autoimmune-inflammatory diseases.
All TLRs appear to function as either a homodimer or heterodimer in the
recognition of a
specific, or set of specific, molecular determinants present on pathogenic
organisms including
bacterial cell-surface lipopolysaccharides, lipoproteins, bacterial flagellin,
DNA from both
bacteria and viruses and viral RNA. The cellular response to TLR activation
involves activation
of one or more transcription factors, leading to the production and secretion
of cytokines and
co-stimulatory molecules such as interferons, TNF-, interleukins, MIP-1 and
MCP-1 which
contribute to the killing and clearance of the pathogenic invasion.
90 TLR spatial expression is coincident with the host's environmental
interface. While only a
few other Toll-like proteins have been cloned in Drosophila, the human TLR
family is composed
of at least 11 members, TLR1 through TLR11, that elicit overlapping yet
distinct biological
responses due to differences in cellular expression and signaling pathways
they initiate. Each
of the TLRs is expressed on a different subset of leukocytes and each of the
TLRs is specific in
its expression patterns and PAMP sensitivities and detects different subsets
of pathogens
allowing vigilant surveillance by the immune system.
Toll-like Receptor 7 (TLR7)
TLR7 maps to human chromosome Xp22, and the TLR7 sequence encodes a 1049 (aa)
protein containing 27 N-terminal LRRs with a calculated molecular weight of
121 kDa. TLR7 is
most closely related to TLR8 and TLR9 with 43% and 36% overall (aa) sequence
identity,
respectively.
In vivo, TLR7 mRNA is expressed in lung, placenta, spleen, lymph node, and
tonsil. TLR7
m RNA expression is highest in monocytes, B cells, and plasmocytoid dendritic
cells. In vitro,
TLR7 m RNA expression is upregulated in THP-1 cells upon PMA-induced
differentiation. TLR7
is highly upregulated by exposure to IL-6 and to a slightly lesser extent by
autocrine IFN-y, IL-
113. TLR7 m RNA expression in THP-1 cells is elevated after exposure to both
Gram-positive
and Gram-negative bacteria. Ex vivo, expression of TLR7 is elevated after
exposure to both
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
24
Gram-positive and Gram-negative bacteria in monocytes and to a greater degree
in
granulocytes. TLR7 is expressed in the endosome. The role of TLR7, is to
detect the presence
of "foreign" single-stranded RNA within a cell, as a means to respond to viral
invasion. TLR7 is
a structurally highly conserved protein which recognizes guanosine- or uridine-
rich, single-
stranded RNA (ssRNA) from viruses such as human immunodeficiency virus,
vesicular
stomatitis virus and influenza virus. Thus, through activation of dendritic
cells and other antigen-
presenting cells, TLR7 engagement and resulting cytokine production is
expected to activate
diverse innate and acquired immune response mechanisms leading to the
destruction of
pathogens, infected cells or tumor cells.
Compounds of Formula (I), Formula (la) or Formula (lb), pharmaceutically
acceptable salts
and isomers thereof, pharmaceutical compositions, and/or combinations are
agonists of toll-like
receptor 7 activity, and are used in the treatment of diseases and/or
disorders associated with
such TLR7 receptors.
In certain embodiments, the compounds of Formula (I), Formula (la) or Formula
(lb),
pharmaceutically acceptable salts and isomers thereof, pharmaceutical
compositions, and/or
combinations are used in the treatment of respiratory diseases and/or
disorders including, but
not limited to, asthma, bronchial asthma, allergic asthma, intrinsic asthma,
extrinsic asthma,
exercise-induced asthma, drug-induced asthma (including aspirin and NSAID-
induced) and
dust-induced asthma, chronic obstructive pulmonary disease (COPD); bronchitis,
including
infectious and eosinophilic bronchitis; emphysema; bronchiectasis; cystic
fibrosis; sarcoidosis;
farmer's lung and related diseases; hypersensitivity pneumonitis; lung
fibrosis, including
cryptogenic fibrosing alveolitis, idiopathic interstitial pneumonias, fibrosis
complicating anti-
neoplastic therapy and chronic infection, including tuberculosis and
aspergillosis and other
fungal infections; complications of lung transplantation; vasculitic and
thrombotic disorders of
the lung vasculature, and pulmonary hypertension; antitussive activity
including treatment of
chronic cough associated with inflammatory and secretory conditions of the
airways, and
iatrogenic cough; acute and chronic rhinitis including rhinitis medicamentosa,
and vasomotor
rhinitis; perennial and seasonal allergic rhinitis including rhinitis nervosa
(hay fever); nasal
polyposis; acute viral infection including the common cold, and infection due
to respiratory
syncytial virus, influenza, coronavirus (including SARS) and adenovirus.
In certain embodiments, the compounds of Formula (I), Formula (la) or Formula
(lb),
pharmaceutically acceptable salts and isomers thereof, pharmaceutical
compositions, and/or
combinations are used in the treatment of dermatological disorders including,
but not limited to,
psoriasis, atopic dermatitis, contact dermatitis or other eczematous
dermatoses, and delayed-
type hypersensitivity reactions; phyto- and photodermatitis; seborrhoeic
dermatitis, dermatitis
herpetiformis, lichen planus, lichen sclerosus et atrophica, pyoderma
gangrenosum, skin
sarcoid, basal cell carcinoma, actinic keratosis, discoid lupus erythematosus,
pemphigus,
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
pemphigoid, epidermolysis bullosa, urticaria, angioedema, vasculitides, toxic
erythemas,
cutaneous eosinophilias, alopecia areata, male-pattern baldness, Sweet's
syndrome, Weber-
Christian syndrome, erythema multiforme; cellulitis, both infective and non-
infective;
panniculitis;cutaneous lymphomas, non-melanoma skin cancer and other
dysplastic lesions;
5 drug-induced disorders including fixed drug eruptions.
In certain embodiments, the compounds of Formula (I), Formula (la) or Formula
(lb),
pharmaceutically acceptable salts and isomers thereof, pharmaceutical
compositions, and/or
combinations are used in the treatment of ocular diseases and/or disorders
including, but not
limited to, blepharitis; conjunctivitis, including perennial and vernal
allergic conjunctivitis; iritis;
10 anterior and posterior uveitis; choroiditis; autoimmune, degenerative or
inflammatory disorders
affecting the retina; ophthalmitis including sympathetic ophthalmitis;
sarcoidosis; infections
including viral, fungal, and bacterial.
In certain embodiments, the compounds of Formula (I), Formula (la) or Formula
(lb),
pharmaceutically acceptable salts and isomers thereof, pharmaceutical
compositions, and/or
15 combinations are used in the treatment of other auto-immune and allergic
disorders including,
but not limited to, rheumatoid arthritis, irritable bowel syndrome, systemic
lupus erythematosus,
multiple sclerosis, Hashimoto's thyroiditis, Crohn's disease, inflammatory
bowel disease (IBD),
Graves' disease, Addison's disease, diabetes mellitus, idiopathic
thrombocytopaenic purpura,
eosinophilic fasciitis, hyper-IgE syndrome, antiphospholipid syndrome and
Sazary syndrome.
90 In certain embodiments, the compounds of Formula (I), Formula (la) or
Formula (lb),
pharmaceutically acceptable salts and isomers thereof, and pharmaceutical
compositions are
used in the treatment of cancer including, but not limited to, bladder,
prostate, breast, colorectal,
liver, hepatocellular carcinoma, lung, ovarian, pancreatic, bowel and colon,
stomach, skin and
brain tumors and malignancies affecting the bone marrow (including the
leukaemias) and
25 lymphoproliferative systems, such as Hodgkin's and non-Hodgkin's
lymphoma; including the
prevention and treatment of metastatic disease and tumor recurrences, and
paraneoplastic
syndromes. In certain embodiments, the compounds of Formula (I), Formula (la)
or Formula
(lb), pharmaceutically acceptable salts and isomers thereof, and
pharmaceutical compositions
are useful as modulators of toll-like receptor activity, and are used in the
treatment of
neoplasias including, but not limited to, basal cell carcinoma, squamous cell
carcinoma, actinic
keratosis, melanoma, carcinomas, sarcomas, leukemias, renal cell carcinoma,
Kaposi's
sarcoma, myelogeous leukemia, chronic lymphocytic leukemia and multiple
myeloma.
In certain embodiments, the compounds of Formula (I), Formula (la) or Formula
(lb),
pharmaceutically acceptable salts and isomers thereof, pharmaceutical
compositions, and/or
combinations are used in the treatment of infectious diseases including, but
not limited to, viral
infectious diseases such as genital warts, common warts, plantar warts,
respiratory syncytial
virus (RSV), hepatitis B, hepatitis C, Dengue virus, herpes simplex virus (by
way of example
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
26
only, HSV-I, HSV-II, CMV, or VZV), molluscum contagiosum, vaccinia, variola,
lentivirus, human
immunodeficiency virus (HIV), human papillonna virus (HPV), cytonnegalovirus
(CMV), varicella
zoster virus (VZV), rhinovirus, enterovirus, adenovirus, coronavirus (e.g.,
SARS), influenza,
para-influenza, mumps virus, measles virus, papovavirus, hepadnavirus,
flavivirus, retrovirus,
arenavirus (by way of example only, LCM, Junin virus, Machupo virus, Guanarito
virus and
Lassa Fever) and filovirus (by way of example only, ebola virus or marbug
virus).
In certain embodiments, the compounds of Formula (I), Formula (la) or Formula
(lb),
pharmaceutically acceptable salts and isomers thereof, pharmaceutical
compositions, and/or
combinations are used in the treatment of bacterial, fungal, and protozoal
infections including,
but not limited to, tuberculosis and mycobacterium avium, leprosy;
pneumocystis carnii,
cryptosporidiosis, histoplasmosis, toxoplasmosis, trypanosome infection,
leishmaniasis,
infections caused by bacteria of the genus Escherichia, Enterobacter,
Salmonella,
Staphylococcus, Klebsiella, Proteus, Pseudomonas, Streptococcus, and
Chlamydia, and fungal
infections such as candidiasis, aspergillosis, histoplasmosis, cryptococcal
meningitis.
Administration and Pharmaceutical Compositions
For the therapeutic uses of compounds of Formula (I), Formula (la) and Formua
(lb), or
pharmaceutically acceptable salts and isomers thereof, such compounds are
administered in
therapeutically effective amounts either alone or as part of a pharmaceutical
composition.
Accordingly, such pharmaceutical compositions, comprise a compound of Formula
(I), Formula
(la) or Formula (lb), or pharmaceutically acceptable salt thereof, and one or
more
pharmaceutically acceptable carriers, diluents, or excipients. In addition,
such compounds and
compositions are administered singly or in combination with one or more
additional therapeutic
agents. The method of administration of such compounds and compositions
include, but are not
limited to, oral administration, rectal administration, parenteral,
intravenous administration,
intravitreal administration, intramuscular administration, inhalation,
intranasal administration,
topical administration, ophthalmic administration or otic administration.
The therapeutically effective amount will vary depending on, among others, the
disease
indicated, the severity of the disease, the age and relative health of the
subject, the potency of
the compound administered, the mode of administration and the treatment
desired. In certain
embodiments, the daily dosage of a compound of Formula (I), Formula (la) or
Formula (lb),
satisfactory results are indicated to be obtained systemically at daily
dosages of from about
0.03 to 2.5mg/kg per body weight. In certain embodiments, the daily dosage of
a compound of
Formula (I), Formula (la) or Formula (lb), administered by inhalation, is in
the range from 0.05
micrograms per kilogram body weight (pg/kg) to 100 micrograms per kilogram
body weight
(pg/kg). In certain embodiments, the quantit of a compound of Formula (I),
Formula (la) or
Formula (lb) per dose administered by inhalation, is in the range from 10 ng
to 500 ng. In other
embodiments, the daily dosage of a compound of Formula (I), Formula (la) or
Formula (lb),
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
27
administered orally, is in the range from 0.01 micrograms per kilogram body
weight (pg/kg) to
100 milligrams per kilogram body weight (mg/kg). An indicated daily dosage in
the larger
mammal, e.g. humans, is in the range from about 0.5mg to about 100mg of a
compound of
Formula (I), conveniently administered, e.g. in divided doses up to four times
a day or in
controlled release form. In certain embodiment, unit dosage forms for oral
administration
comprise from about 1 to 50 mg of a compound of Formula (I), Formula (la) or
Formula (lb).
Other aspects provided herein are processes for the preparation of
pharmaceutical
composition which comprise a compound of Formula (I), Formula (la) or Formula
(lb), or
pharmaceutically acceptable salt thereof. In certain embodiments, such
processes include
admixing a compound of the Formula (I), Formula (la) or Formula (lb), or
pharmaceutically
acceptable salts thereof, with one or more pharmaceutically acceptable
carriers, diluents or
excipients. In certain embodiments, the pharmaceutical compositions comprising
a compound
of Formula (I), Formula (la) or Formula (lb), in free form or in a
pharmaceutically acceptable salt
form, in association with at least one pharmaceutically acceptable carrier,
diluent or excipient
are manufactured by mixing, granulating and/or coating methods. In other
embodiments, such
compositions are optionally contain excipients, such as preserving,
stabilizing, wetting or
emulsifying agents, solution promoters, salts for regulating the osmotic
pressure and/or buffers.
In other embodiments, such compositions are sterilized.
Oral Dosage Forms
90 In certain embodiments, the pharmaceutical compositions containing a
compound of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
administered orally as discrete dosage forms, wherein such dosage forms
include, but are not
limited to, capsules, gelatin capsules, caplets, tablets, chewable tablets,
powders, granules,
syrups, flavored syrups, solutions or suspensions in aqueous or non-aqueous
liquids, edible
foams or whips, and oil-in-water liquid emulsions or water-in-oil liquid
emulsions.
The capsules, gelatin capsules, caplets, tablets, chewable tablets, powders or
granules,
used for the oral administration of a compound of Formula (I), Formula (la) or
Formula (lb), or
pharmaceutically acceptable salt thereof, are prepared by admixing a compound
of Formula (I),
Formula (la) or Formula (lb), or pharmaceutically acceptable salt thereof,
together with at least
one excipient using conventional pharmaceutical compounding techniques. Non-
limiting
examples of excipients used in oral dosage forms described herein include, but
are not limited
to, binders, fillers, disintegrants, lubricants, absorbents, colorants,
flavors, preservatives and
sweeteners.
Non-limiting examples of such binders include, but are not limited to, corn
starch, potato
starch, starch paste, pre-gelatinized starch, or other starches, sugars,
gelatin, natural and
synthetic gums such as acacia, sodium alginate, alginic acid, other alginates,
tragacanth, guar
gum, cellulose and its derivatives (by way of example only, ethyl cellulose,
cellulose acetate,
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
28
carboxym ethyl cellulose calcium, sodium carboxymethylcellulose, methyl
cellulose,
hydroxypropyl methylcellulose and microcrystalline cellulose), magnesium
aluminum silicate,
polyvinyl pyrrolidone and combinations thereof.
Non-limiting examples of such fillers include, but are not limited to, talc,
calcium carbonate
(e.g., granules or powder), microcrystalline cellulose, powdered cellulose,
dextrates, kaolin,
mannitol, silicic acid, sorbitol, starch, pre-gelatinized starch, and mixtures
thereof. In certain
embodiments, the binder or filler in pharmaceutical compositions are present
in from about 50
to about 99 weight percent of the pharmaceutical composition or dosage form.
Non-limiting examples of such disintegrants include, but are not limited to,
agar-agar, alginic
acid, sodium alginate, calcium carbonate, sodium carbonate, microcrystalline
cellulose,
croscarmellose sodium, crospovidone, polacrilin potassium, sodium starch
glycolate, potato or
tapioca starch, pre-gelatinized starch, other starches, clays, other algins,
other celluloses,
gums, and combinations thereof. In certain embodiments, the amount of
disintegrant used in
the pharmaceutical compositions is from about 0.5 to about 15 weight percent
of disintegrant,
while in other embodiments the amount is from about 1 to about 5 weight
percent of
disintegrant.
Non-limiting examples of such lubricants include, but are not limited to,
sodium stearate,
calcium stearate, magnesium stearate, stearic acid, mineral oil, light mineral
oil, glycerin,
sorbitol, mannitol, polyethylene glycol, other glycols, sodium lauryl sulfate,
talc, hydrogenated
vegetable oil (by way of example only, peanut oil, cottonseed oil, sunflower
oil, sesame oil, olive
oil, corn oil, and soybean oil), zinc stearate, sodium oleate, ethyl oleate,
ethyl laureate, agar,
silica, a syloid silica gel (AEROSIL 200, manufactured by W.R. Grace Co. of
Baltimore, Md.), a
coagulated aerosol of synthetic silica (marketed by Degussa Co. of Plano,
Tex.), CAB-0-SIL (a
pyrogenic silicon dioxide product sold by Cabot Co. of Boston, Mass.) and
combinations
thereof. In certain embodiments, the amount of lubricants used in the
pharmaceutical
compositions is in an amount of less than about 1 weight percent of the
pharmaceutical
compositions or dosage forms.
Non-limiting examples of such diluents include, but are not limited to,
lactose, dextrose,
sucrose, mannitol, sorbitol, cellulose, glycine or combinations thereof.
In certain embodiments, tablets and capsules are prepared by uniformly
admixing a
compound of Formula (I), Formula (la) or Formula (lb), or pharmaceutically
acceptable salt
thereof, with liquid carriers, finely divided solid carriers, or both, and
then shaping the product
into the desired presentation if necessary. In certain embodiments, tablets
are prepared by
compression. In other embodiments, tablets are prepared by molding.
In certain embodiments, a compound of Formula (I), Formula (la) or Formula
(lb), or
pharmaceutically acceptable salt thereof, is orally administered as a
controlled release dosage
form. Such dosage forms are used to provide slow or controlled-release of a
compound of
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
29
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof.
Controlled release is obtained using, for example, hydroxypropylnnethyl
cellulose, other polymer
matrices, gels, permeable membranes, osmotic systems, multilayer coatings,
microparticles,
liposomes, microspheres, or a combination thereof. In certain embodiments,
controlled-release
dosage forms are used to extend activity of the compound of Formula (I),
reduce dosage
frequency, and increase patient compliance.
Administration of compounds of Formula (I) as oral fluids such as solution,
syrups and
elixirs are prepared in unit dosage forms such that a given quantity of
solution, syrups or elixirs
contains a predetermined amount of a compound of Formula (I). Syrups are
prepared by
dissolving the compound in a suitably flavored aqueous solution, while elixirs
are prepared
through the use of a non-toxic alcoholic vehicle. Suspensions are formulated
by dispersing the
compound in a non-toxic vehicle. Non-limiting examples of excipients used in
as oral fluids for
oral administration include, but are not limited to, solubilizers,
emulsifiers, flavoring agents,
preservatives, and coloring agents. Non-limiting examples of solubilizers and
emulsifiers
.. include, but are not limited to, water, glycols, oils, alcohols,
ethoxylated isostearyl alcohols and
polyoxy ethylene sorbitol ethers. Non-limiting examples of preservatives
include, but are not
limited to, sodium benzoate. Non-limiting examples of flavoring agents
include, but are not
limited to, peppermint oil or natural sweeteners or saccharin or other
artificial sweeteners.
Parenteral Dosage Forms
In certain embodiments pharmaceutical compositions containing a compound of
Formula (I),
Formula (la) or Formula (lb), or pharmaceutically acceptable salt thereof, are
administered
parenterally by various routes including, but not limited to, subcutaneous,
intravenous (including
bolus injection), intramuscular, and intraarterial.
Such parenteral dosage forms are administered in the form of sterile or
sterilizable
injectable solutions, suspensions, dry and/or lyophylized products ready to be
dissolved or
suspended in a pharmaceutically acceptable vehicle for injection
(reconstitutable powders) and
emulsions. Vehicles used in such dosage forms include, but are not limited to,
Water for
Injection USP; aqueous vehicles such as, but not limited to, Sodium Chloride
Injection, Ringer's
Injection, Dextrose Injection, Dextrose and Sodium Chloride Injection, and
Lactated Ringer's
Injection; water-miscible vehicles such as, but not limited to, ethyl alcohol,
polyethylene glycol,
and polypropylene glycol; and non-aqueous vehicles such as, but not limited
to, corn oil,
cottonseed oil, peanut oil, sesame oil, ethyl oleate, isopropyl myristate, and
benzyl benzoate.
Transdermal Dosage Forms
In certain embodiments pharmaceutical compositions containing a compound of
Formula (I),
Formula (la) or Formula (lb), or pharmaceutically acceptable salt thereof, are
administered
transdemally. Such transdermal dosage forms include "reservoir type" or
"matrix type" patches,
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
which are applied to the skin and worn for a specific period of time to permit
the penetration of a
desired amount of a compound of Formula (I). By way of example only, such
transdermal
devices are in the form of a bandage comprising a backing member, a reservoir
containing the
compound optionally with carriers, optionally a rate controlling barrier to
deliver the compound
5 to the skin of the host at a controlled and predetermined rate over a
prolonged period of time,
and means to secure the device to the skin. In other embodiments, matrix
transdermal
formulations are used.
Formulations for transdermal delivery of a compound of Formula (I) include an
effective
amount of a compound of Formula (I), a carrier and an optional diluent. A
carrier includes, but
10 is not limited to, absorbable pharmacologically acceptable solvents to
assist passage through
the skin of the host, such as water, acetone, ethanol, ethylene glycol,
propylene glycol, butane-
1,3-diol, isopropyl myristate, isopropyl palmitate, mineral oil, and
combinations thereof.
In certain embodiments, such transdermal delivery systems include penetration
enhancers
to assist in delivering one or more compounds of Formula (I) to the tissue.
Such penetration
15 enhancers include, but are not limited to, acetone; various alcohols
such as ethanol, oleyl, and
tetrahydrofuryl; alkyl sulfoxides such as dimethyl sulfoxide; dimethyl
acetamide; dimethyl
formamide; polyethylene glycol; pyrrolidones such as polyvinylpyrrolidone;
Kollidon grades
(Povidone, Polyvidone); urea; and various water-soluble or insoluble sugar
esters such as
Tween 80 (polysorbate 80) and Span 60 (sorbitan monostearate).
90 In other embodiments, the pH of such a transdermal pharmaceutical
composition or dosage
form, or of the tissue to which the pharmaceutical composition or dosage form
is applied, is
adjusted to improve delivery of one or more compounds of Formula (I). In other
embodiments,
the polarity of a solvent carrier, its ionic strength, or tonicity are
adjusted to improve delivery. In
other embodiments, compounds such as stearates are added to advantageously
alter the
25 hydrophilicity or lipophilicity of one or more compounds of Formula (I)
so as to improve delivery.
In certain embodiments, such stearates serve as a lipid vehicle for the
formulation, as an
emulsifying agent or surfactant, and as a delivery-enhancing or penetration-
enhancing agent. In
other embodiments, different salts, hydrates s of the compounds of Formula (I)
are used to
further adjust the properties of the resulting composition.
30 Topical Dosage Forms
In certain embodiments a compound of Formula (I), Formula (la) or Formula
(lb), or
pharmaceutically acceptable salt thereof, is administered by topical
application of
pharmaceutical composition containing a compound of Formula (I), Formula (la)
or Formula
(lb), or pharmaceutically acceptable salt thereof, in the form of lotions,
gels, ointments solutions,
emulsions, suspensions or creams. Suitable formulations for topical
application to the skin are
aqueous solutions, ointments, creams or gels, while formulations for
ophthalmic administration
are aqueous solutions. Such formulations optionally contain solubilizers,
stabilizers, tonicity
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
31
enhancing agents, buffers and preservatives.
Such topical formulations include at least one carrier, and optionally at
least one diluent.
Such carriers and diluents include, but are not limited to, water, acetone,
ethanol, ethylene
glycol, propylene glycol, butane-1,3-diol, isopropyl myristate, isopropyl
palmitate, mineral oil,
and combinations thereof.
In certain embodiments, such topical formulations include penetration
enhancers to assist in
delivering one or more compounds of Formula (I) to the tissue. Such
penetration enhancers
include, but are not limited to, acetone; various alcohols such as ethanol,
oleyl, and
tetrahydrofuryl; alkyl sulfoxides such as dimethyl sulfoxide; dimethyl
acetamide; dimethyl
formamide; polyethylene glycol; pyrrolidones such as polyvinylpyrrolidone;
Kollidon grades
(Povidone, Polyvidone); urea; and various water-soluble or insoluble sugar
esters such as
Tween 80 (polysorbate 80) and Span 60 (sorbitan monostearate).
In certain embodiments pharmaceutical compositions containing a compound of
Formula (I),
Formula (la) or Formula (lb), or pharmaceutically acceptable salt thereof, are
administered by
inhalation. Dosage forms for inhaled administration are formulated as aerosols
or dry powders.
Aerosol formulations for inhalation administration comprise a solution or fine
suspension of a
compound of Formula (I), Formula (la) or Formula (lb), or pharmaceutically
acceptable salt
thereof, in a pharmaceutically acceptable aqueous or non-aqueous solvent. In
addition, such
pharmaceutical compositions optionally comprise a powder base such as lactose,
glucose,
trehalose, mannitol or starch, and optionally a performance modifier such as L-
leucine or
another amino acid, and/or metals salts of stearic acid such as magnesium or
calcium stearate.
In certain embodiments, compounds of Formula (I) are be administered directly
to the lung
by inhalation using a Metered Dose Inhaler ("MDI"), which utilizes canisters
that contain a
suitable low boiling propellant, e.g., dichlorodifluoromethane,
trichlorofluoromethane,
dichlorotetrafluoroethane, carbon dioxide or other suitable gas, or a Dry
Powder Inhaler (DPI)
device which uses a burst of gas to create a cloud of dry powder inside a
container, which is
then be inhaled by the patient. In certain embodiments, capsules and
cartridges of gelatin for
use in an inhaler or insufflator are formulated containing a powder mixture of
a compound of
Formula (I) and a powder base such as lactose or starch. In certain
embodiments, compounds
of Formula (I) are delivered to the lung using a liquid spray device, wherein
such devices use
extremely small nozzle holes to aerosolize liquid drug formulations that can
then be directly
inhaled into the lung. In other embodiments, compounds of Formula (I) are
delivered to the lung
using a nebulizer device, wherein a nebulizers creates an aerosols of liquid
drug formulations
by using ultrasonic energy to form fine particles that can be readily inhaled.
In other
embodiments, compounds of Formula (I) are delivered to the lung using an
electrohydrodynamic ("EHD") aerosol device wherein such EHD aerosol devices
use electrical
energy to aerosolize liquid drug solutions or suspensions.
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
32
In certain embodiments, the pharmaceutical composition containing a compound
of Formula
(I), Formula (la) or Formula (lb), or pharmaceutically acceptable salts
thereof, described herein,
also contain one or more absorption enhancers. In certain embodiments, such
absorption
enhancers include, but are not limited to, sodium glycocholate, sodium
caprate, N-laury1-13-D-
maltopyranoside, EDTA, and mixed micelles.
In certain embodiments the pharmaceutical composition containing a compound of
Formula
(I), Formula (la) or Formula (lb), or a pharmaceutically accceptable salt
thereof, are
administered nasally. The dosage forms for nasal administration are formulated
as aerosols,
solutions, drops, gels or dry powders.
In certain embodiments the pharmaceutical composition containing a compound of
Formula
(I), Formula (la) or Formula (lb), or a pharmaceutically accceptable salt
thereof, are
administered rectally in the form of suppositories, enemas, ointment, creams
rectal foams or
rectal gels. In certain embodiments such suppositories are prepared from fatty
emulsions or
suspensions, cocoa butter or other glycerides.
In certain embodiments the pharmaceutical composition containing a compound of
Formula
(I), Formula (la) or Formula (lb), or a pharmaceutically accceptable salt
thereof, are
administered opthamically as eye drops. Such formulations are aqueous
solutions that
optionally contain solubilizers, stabilizers, tonicity enhancing agents,
buffers and preservatives.
In certain embodiments the pharmaceutical composition containing a compound of
Formula
(I), Formula (la) or Formula (lb), or a pharmaceutically accceptable salt
thereof, are
administered otically as ear drops. Such formulations are aqueous solutions
that optionally
contain solubilizers, stabilizers, tonicity enhancing agents, buffers and
preservatives.
In certain embodiments the pharmaceutical composition containing a compound of
Formula
(I), Formula (la) or Formula (lb), or a pharmaceutically accceptable salt
thereof, are formulated
as a depot preparation. Such formulations are administered by implantation
(for example
subcutaneously or intramuscularly) or by intramuscular injection. In certain
embodiments, such
formulations include polymeric or hydrophobic materials (for example, as an
emulsion in an
acceptable oil) or ion exchange resins, or as sparingly soluble derivatives,
for example, as a
sparingly soluble salt.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for oral administration for the treatment of viral infectious diseases
and/or disorders
associated with TLR7 activity.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for intramuscular administration for the treatment of viral infectious
diseases and/or
disorders associated with TLR7.
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
33
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for oral administration for the treatment of infectious diseases
and/or disorders
associated with TLR7.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for oral administration for the treatment of bacterial diseases and/or
disorders
associated with TLR7.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for oral administration for the treatment of fungal diseases and/or
disorders associated
with TLR7.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
.. adapted for oral administration for the treatment of cancer associated with
TLR7.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for intravenous administration for the treatment of cancer associated
with TLR7.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for administration as eye drops for the treatment of ophthalmic
diseases and/or
disorders associated with TLR7.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for topical administration for the treatment of dermatological
diseases and/or disorders
associated with TLR7.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for topical administration for the treatment of actinic keratosis. In
a further embodiment,
the pharmaceutical compositions comprising a compound of Formula (I), Formula
(la) or
Formula (lb), or pharmaceutically acceptable salt thereof, are adapted for
topical administration
as a cream for the treatment of actinic keratosis.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for topical administration for the treatment of basal cell carcinoma.
In a further
embodiment, the pharmaceutical compositions comprising a compound of Formula
(I), Formula
(la) or Formula (lb), or pharmaceutically acceptable salt thereof, are adapted
for topical
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
34
administration as a cream for the treatment of basal cell carcinoma.
In a further embodiment, the pharmaceutical compositions comprising a compound
of
Formula (I), Formula (la) or Formula (lb), or pharmaceutically acceptable salt
thereof, are
adapted for administration by inhalation for the treatment of respiratory
diseases and/or
disorders associated with TLR7. In certain embodiments, the respiratory
disease is allergic
asthma.
Provided herein are compounds of Formula (I), Formula (la) or Formula (lb),
and
pharmaceutically acceptable salts thereof, and the pharmaceutical composition
containing a
compound of Formula (I), Formula (la) or Formula (lb), or a pharmaceutically
accceptable salt
thereof, and/or pharmaceutically acceptable salts thereof, for use in
activating TLR7 activity,
and thereby are used to in the prevention or treatment of diseases and/or
disorders associated
with TLR7 activity. Such compounds of Formula (I) and pharmaceutically
acceptable salts
thereof, and pharmaceutical compositions are agonists of TLR7.
Also are methods for the treatment of a subject suffering from a disease
and/or disorder
associated with TLR7 activity, wherein the methods include administering to
the subject an
effective amount of a compound of Formula (I) or a pharmaceutically acceptable
salt thereof,
either alone or as part of a pharmaceutical composition as described herein.
Provided herein is the use of a compound of Formula (I), or a pharmaceutically
acceptable
salt thereof, in the preparation of a medicament for the treatment of a
disease or disorder
associated with TLR7 activity.
Combination Treatment
In certain embodiments, a compound of Formula (I), or a pharmaceutically
acceptable salt
thereof, or a pharmaceutical composition containing a compound of Formula (I),
Formula (la) or
Formula (lb), or pharmaceutically acceptable salt thereofõ is administered
alone (without an
additional therapeutic agent) for the treatment of one or more of the disease
and/or disorders
associated with TLR7 activity described herein.
In other embodiments, a compound of Formula (I), or a pharmaceutically
acceptable salt
thereof, or a pharmaceutical composition containing a compound of Formula (I),
Formula (la) or
Formula (lb), or pharmaceutically acceptable salt thereofõ is administered in
combination with
one or more additional therapeutic agents, for the treatment of one or more of
the disease
and/or disorders associated with TLR7 activity described herein.
In other embodiments, a compound of Formula (I), or a pharmaceutically
acceptable salt
thereof, or a pharmaceutical composition containing a compound of Formula (I),
Formula (la) or
Formula (lb), or pharmaceutically acceptable salt thereofõ is formulated in
combination with one
or more additional therapeutic agents and administered for the treatment of
one or more of the
disease and/or disorders associated with TLR7 activity described herein.
In other embodiments, a compound of Formula (I), or a pharmaceutically
acceptable salt
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
thereof, or a pharmaceutical composition containing a compound of Formula (I),
Formula (la) or
Formula (lb), or pharmaceutically acceptable salt thereofõ is administered
sequentially with one
or more additional therapeutic agents, for the treatment of one or more of the
disease and/or
disorders associated with TLR7 activity described herein.
5 In other embodiments, the combination treatments include administration
of a compound of
Formula (I), or a pharmaceutically acceptable salt thereof, or a
pharmaceutical composition
containing a compound of Formula (I), prior to administration of one or more
additional
therapeutic agents, for the treatment of one or more of the disease and/or
disorders associated
with TLR7 activity described herein.
10 In other embodiments, the combination treatments include administration
of a compound of
Formula (I), or a pharmaceutically acceptable salt thereof, or a
pharmaceutical composition
containing a compound of Formula (I), subsequent to administration of one or
more additional
therapeutic agents, for the treatment of one or more of the disease and/or
disorders associated
with TLR7 activity described herein.
15 In certain embodiments, the combination treatments include
administration of a compound
of Formula (I), or a pharmaceutically acceptable salt thereof, or a
pharmaceutical composition
containing a compound of Formula (I), concurrently with one or more additional
therapeutic
agents, for the treatment of one or more of the disease and/or disorders
associated with TLR7
activity described herein.
20 In certain embodiments, the combination treatments include
administration of a compound
of Formula (I), or a pharmaceutically acceptable salt thereof, or a
pharmaceutical composition
containing a compound of Formula (I) formulated with one or more additional
therapeutic
agents, for the treatment of one or more of the disease and/or disorders
associated with TLR7
activity described herein.
25 In certain embodiments of the combination therapies described herein,
the compounds of
Formula (I), Formula (la) or Formula (lb), or a pharmaceutically acceptable
salt thereof, and the
additional therapeutics agent(s) act additively. In certain embodiments of the
combination
therapies described herein, the compounds of Formula (I), or pharmaceutically
acceptable salts
thereof, and the additional therapeutics agent(s) act synergistically.
30 In other embodiments, a compound of Formula (I), Formula (la) or Formula
(lb), or a
pharmaceutically acceptable salt thereof, or a pharmaceutical composition
containing a
compound of Formula (I), Formula (la) or Formula (lb), or pharmaceutically
acceptable salt
thereof, is administered to a patient who has not previously undergone or is
not currently
undergoing treatment with another therapeutic agent.
35 The additional therapeutic agents used in combination with a compound of
Formula (I),
Formula (la) or Formula (lb), or a pharmaceutically acceptable salt thereof,
include, but are not
limited to antibiotics or antibacterial agents, antiemetic agents, antifungal
agents, anti-
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
36
inflammatory agents, antiviral agents, viral enzyme inhibitors or anticancer
agents.
In certain embodiments, the pharmaceutical composition containing a compound
of Formula
(I), Formula (la) or Formula (lb), or a pharmaceutically acceptable salt
thereof, are
immunogenic compositions.
In other embodiments, the compound(s) of Formula (I), Formula (la) or Formula
(lb), or a
pharmaceutically acceptable salt thereof, are immune potentiators and impart
an
immunostimulatory effect upon administration when compared to formulations
that do not
contain a compound of Formula (I), Formula (la) or Formula (lb), or
pharmaceutically
acceptable salt thereof..
The immunostimulatory effect referred to herein is often an enhancement of the
immunogenic composition's effect. In certain embodiments the enhancement of
the efficacy of
the immunogenic composition is by at least 10% relative to the effect of the
immunogenic
composition in the absence of the immune potentiator. In certain embodiments
the
enhancement of the efficacy of the immunogenic composition is by at least 20%
relative to the
effect of the immunogenic composition in the absence of the immune
potentiator. In certain
embodiments the enhancement of the efficacy of the immunogenic composition is
by at least
30% relative to the effect of the immunogenic composition in the absence of
the immune
potentiator. In certain embodiments the enhancement of the efficacy of the
immunogenic
composition is by at least 40% relative to the effect of the immunogenic
composition in the
absence of the immune potentiator. In certain embodiments the enhancement of
the efficacy of
the immunogenic composition is by at least 50% relative to the effect of the
immunogenic
composition in the absence of the immune potentiator. In certain embodiments
the
enhancement of the efficacy of the immunogenic composition is by at least 60%
relative to the
effect of the immunogenic composition in the absence of the immune
potentiator. In certain
embodiments the enhancement of the efficacy of the immunogenic composition is
by at least
70% relative to the effect of the immunogenic composition in the absence of
the immune
potentiator. In certain embodiments the enhancement of the efficacy of the
immunogenic
composition is by at least 80% relative to the effect of the immunogenic
composition in the
absence of the immune potentiator. In certain embodiments the enhancement of
the efficacy of
the immunogenic composition is by at least 90% relative to the effect of the
immunogenic
composition in the absence of the immune potentiator. In certain embodiments
the
enhancement of the efficacy of the immunogenic composition is by at least 100%
relative to the
effect of the immunogenic composition in the absence of the immune
potentiator.
The immunogenic compositions as disclosed herein may be used in a method for
raising or
enhancing an immune response in a mammal comprising the step of administering
an effective
amount of an immunogenic composition as disclosed herein. The immune response
is
preferably protective and preferably involves antibodies and/or cell-mediated
immunity.
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
37
In certain embodimentsembodiments, the immunogenic compositions disclosed
herein may
be used as a medicament, e.g., for use in raising or enhancing an immune
response in a
mammal.
In certain embodiments, the immunogenic compositions disclosed herein may be
used in
the manufacture of a medicament for raising an immune response in a mammal.
The mammal
is preferably a human, but may be, e.g., a cow, a pig, a chicken, a cat or a
dog.
One way of checking efficacy of therapeutic treatment involves monitoring
pathogen
infection after administration of the immunogenic compositions disclosed
herein. One way of
checking efficacy of prophylactic treatment involves monitoring immune
responses, systemically
(such as monitoring the level of IgG1 and IgG2a production) and/or mucosally
(such as
monitoring the level of IgA production), against the antigens included in or
administered in
conjunction with the immunogenic compositions disclosed herein after
administration of the
immunogenic composition (and the antigen if administered separately).
Typically, antigen-
specific serum antibody responses are determined post-immunisation but pre-
challenge
whereas antigen-specific mucosal antibody responses are determined post-
immunisation and
post-challenge.
Another way of assessing the immunogenicity of the immunogenic compositions
disclosed
herein where the antigen is a protein is to express the proteins
reconribinantly for screening
patient sera or mucosal secretions by immunoblot and/or microarrays. A
positive reaction
between the protein and the patient sample indicates that the patient has
mounted an immune
response to the protein in question. This method may also be used to identify
immunodominant
antigens and/or epitopes within protein antigens.
The efficacy of the immunogenic compositions can also be determined in vivo by
challenging appropriate animal models of the pathogen of interest infection.
95 The immunogenic compositions disclosed herein will generally be
administered directly to a
subject. Direct delivery may be accomplished by parenteral injection (e.g.,
subcutaneously,
intraperitoneally, intravenously, intramuscularly, or to the interstitial
space of a tissue), or
mucosally, such as by rectal, oral (e.g., tablet, spray), vaginal, topical,
transdermal or
transcutaneous, intranasal, ocular, aural, pulmonary or other mucosal
administration.
The immunogenic compositions may be used to elicit systemic and/or mucosal
immunity,
preferably to elicit an enhanced systemic and/or mucosal immunity.
Preferably the enhanced systemic and/or mucosal immunity is reflected in an
enhanced
TH1 and/or TH2 immune response. Preferably, the enhanced immune response
includes an
increase in the production of IgG1 and/or IgG2a and/or IgA.
Certain aspects and examples of the invention are provided in the following
listing of
enumerated embodiments. It will be recognized that features specified in each
embodiment may
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
38
be combined with other specified features to provide further embodiments of
the present
invention.
1. Compounds, and the pharmaceutically acceptable salts, individual isomers
and mixture of
isomers thereof, have a structure according to Formula (I):
-0
R I R3
N
/ R4
H2N N
Formula (I)
R1 is -NHR6 or -NHCHR6R9;
R3 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H
or tetrazolyl;
R4 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H
or tetrazolyl;
where, when R3 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -0F2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
C(=0)0H, -L2C(=0)0H, -L7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
or when R4 is H, then R3 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H or tetrazolyl;
L1 is -(CF12)m-;
1_2 15 -(CH2)m-;
= -(CH2)m(CHR11CH2)m(CH2),-,-;
R6 is -C3-C6alkyl;
R7 is -C1-C3alkyl;
R9 is LiOH;
each R11 is independently selected from -OH;
R12 is
a) an unsubstituted tetrazolyl;
b) a tetrazolyl substituted with -1_1C(=0)0H;
or
c) a 5-6 membered heterocycloalkyl having 1 to 2 heteroatoms independently
selected from N and 0 substituted with -C(=0)0H;
each m is independently selected from 1, 2, 3, and 4,
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
39
and
each n is independently selected from 0, 1, 2, 3, and 4.
2. In certain embodiments, the compound of Formula (I) is a compound of
Formula (la) or
Formula (lb):
-0 -0
RI II R3 RI
Ni\E
R4
H2N N 112N N
Formula (la) Formula (lb).
3. In certain embodiments of the compounds of Formula (I),
R1 is -NHR6 or -NHCHR6R9;
R3 is H, -0L2C(=0)0H or tetrazolyl;
1714 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H or tetrazolyl;
where, when R3 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H or tetrazolyl;
or when R4 is H, then R3 is -0L2C(=0)0H or tetrazolyl;
L1 is -(CF12)m-;
1_2 is -(CF12)m-;
L7 = -(CH2)m(CHR11CH2)m(Ch12)ri-;
R6 is -C3-C6alkyl;
R7 is -C1-C3alkyl; methyl
R9 is LiOH;
each R11 is independently selected from -OH;
R12 is
a) an unsubstituted tetrazolyl;
95 b) a tetrazolyl substituted with -1_1C(=0)0H;
or
C) a 5-6 membered heterocycloalkyl having 1 to 2 heteroatoms independently
selected from N and 0 substituted with -C(=0)0H;
each m is independently selected from 1, 2, 3, and 4,
and
each n is independently selected from 0, 1, 2, 3, and 4.
4. In certain embodiments of the compounds of Formula (I), Formula (la) or
Formula (lb),
R1 is -NHR6 or -NHCHR6R9;
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
A3 is H, -L2R12, -0L2R12, -C(CH3)21:112, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
CF2C(=0)0H, -CH=CHC(=0)0H, -C(=0)0H, -L2C(=0)0H, -L7C(=0)0H, -
NN-1`,1,.. N.-
\'&T.
OL2C(=0)0H, µ32,,N'N or -4'N
R4 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
5 CF2C(=0)OH, -CH=CHC(=0)OH, -C(=0)0H, -L2C(=0)0H, -L7C(=0)0H,
N-NH
4)1\is:"
OL2C(=0)0H, or
where, when R3 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0F17), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
HN-N
C(=0)0H, -L2C(=0)0H, -1_7C(=0)0H, -0L2C(=0)0H, or
N-NH
\414:Ni
10 =
or when R4 is H, then R3 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(01:17), -CF2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
HN-1\,1
C(=0)0H, -L2C(=0)0H, -L7C(=0)0H, -0L2C(=0)0Hõ
N-NH
NiciN
=
15 Li is -(CF12)rn-;
1_2 is -(CF12)m-;
1_7 = -(CH2)rn(CHR11C1-12)m(CF12)n-;
R6 is -C3-00alkyl;
A7 is -C1-C3alkyl;
20 R9 isLi0H;
each R11 is independently selected from -OH;
0
HN-N N-NH N-N
I
N 1/4.A./21-1 N N \-AJ2F1 ssN II 'N
R12 is or I
each m is independently selected from 1, 2, 3, and 4,
and
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
41
each n is independently selected from 0, 1, 2, 3, and 4.
5. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb),
R1 is -NHR6 or -NHCHR6R9;
HN-1\,1 N-NH
NA.N:N k:11'NiN
R3 iS H, -0L2C(=0)0H, or
R4 is H, -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -P(=0)0H(0R7), -CF2R12, -
HN-N
CF2C(=0)OH, -CH=CHC(=0)OH, -C(=0)0H, -L2C(=0)0H, -L7C(=0)0H, N or
N-NH
4 2,N
N
where, when R3 is H, then R4 is -L2R12, -0L2R12, -C(CH3)2R12, -P(=0)(OH)2, -
P(=0)0H(0R7), -0F2R12, -CF2C(=0)0H, -CH=CHC(=0)0H, -
HN-N N-NH
4NANs:N NiN
1_ = 10 C(=0)0H, -L2C(=0)0H, -7C(=0)0H, or
HN-Ns N-NH
µ,N
or when R4 is H, then R3 is -0L2C(=0)0Hõ N N
L1 is -(CF12)m-;
1_2 15 -(CF12)m-;
= -(CH2)m(CHRIICH2)m(CH2),-,-;
R6 is -C3-C6alkyl;
R7 is -C1-C3alkyl;
R9 is LiOH;
each R11 is independently selected from -OH;
0
HN-N N-NH N-N
,21\1 n y LA-121-1 LAJ211
N N N =
R12 is , I or I
each m is independently selected from 1, 2, 3, and 4,
and
each n is independently selected from 0, 1, 2, 3, and 4.
6. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), Li
is -CH2-; L2 is -CH2- or -CH2CH2-; L7 = 7(CH2)2(CHR11CH2)2-; R6 is -C4alkyl or
-05alkyl; R7 is
methyl, ethyl or propyl, and R11 is OH.
7. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), R1
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
42
is -NHR6; R3 is H; R4 is -L2R12; L2 is -(CH2)m-; R6 is -C3-05alkyl;
F112 is
a) an unsubstituted tetrazolyl;
b) a tetrazolyl substituted with -1_1C(=0)0H;
or
C) a 5-6 membered heterocycloalkyl having 1 to 2 heteroatoms independently
selected from N and 0 substituted with -C(=0)0H,
and
each m is independently selected from 1, 2, 3, and 4.
8. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), R1
is -NHR6; R3 is H; R4 is -L2R12; L2 is -CH2- or -CH2CH2-; R6 is -05alkyl; and
0
HN-N N-NH N-N N CO2H N CO2H ),,õ,CO2H
µ:11
R12 is -1; N AN AN -nr
, I or I
9. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), R1
is -NHR6; R3 is H; R4 is -L2R12; L2 is -CH2- or -CH2CH2-; R6 is -05alkyl; and
NCO2H N Nõ(..µõ I
:N
Al2 is A N A N 'yr , I or I
10. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), R1
is -NHR6; R3 is H; R4 is -L2R12; L2 is -CH2- or -CH2CH2-; R6 is -05alkyl; and
HN-N, N-NH
JN
2,1\1
A N -172, N
R12 is or
11. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), R1
is -NHR6; R3 is H; R4 is -L2R12; L2 is -CH2- or -CH2CH2-; R6 is -05alkyl; and
CO2H CO2H .'"CO2H
R12 is I , I or I
12. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb), R1
is -NHR6; R3 is H; R4 is -L2C(=0)0H; L2 is -CH2- or -CH2CH2-; and R6 is -
05alkyl.
13. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb),
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
43
HN-Nõ N-NH
µ4N:,N
R12 is' or
14. In certain embodiments of the compounds of Formula (1), Formula (la) and
Formula (lb),
NCO2H N CO2H N CO2H
R12 is I , I or I
15. In certain embodiments of the compounds of Formula (1), Formula (la) and
Formula (lb) the
compound is selected from:
5-(5-((1H-tetrazol-5-yl)methoxy)-2-methoxybenzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-2,4-diamine;
5-(5-((1H-tetrazol-5-yhmethyl)-2-methoxybenzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrim dine-
2,4-diamine;
(S)-2-((5-(5-((1H-tetrazol-5-yhmethyl)-2-methoxybenzyl)-2-amino-5H-pyrrolo[3,2-
d]pyrimidin-4-y1)amino)hexan-1-01;
(S)-2-((5-(5-((1H-tetrazol-5-yhmethyl)-2-rnethoxybenzyl)-2-amino-5H-
pyrrolo[3,2-
d]pyrimidin-4-Aamino)pentan-1-ol;
5-(5-(2-(1H-tetrazol-5-ypethyl)-2-methoxybenzy1)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrim idine-
2,4-diamine;
N4-hexy1-5-(2-methoxy-5-(1H-tetrazol-5-y1)benzyl)-5H-pyrrolo[3,2-d]pyrimidine-
2,4-
diamine;
(S)-2-((2-am ino-5-(2-methoxy-5-(1H-tetrazol-5-yhbenzy1)-5H-pyrrolo[3,2-
d]pyrim idin-4-
yl)amino)hexan-1-01;
2-(5-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxybenzyl)-2H-tetrazol-2-yhacetic acid;
5-(2-methoxy-4-(1H-tetrazol-5-yl)benzyl)-N4-pentyl-5H-pyrrolo[3,2-d]pyrimidine-
2,4-
diam ine;
5-(2-methoxy-5-(1 H-tetrazol-5-yl)benzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-2,4-
diamine;
5-(5-(2-(1H-tetrazol-5-yl)propan-2-y1)-2-methoxybenzy1)-N4-pentyl-5H-
pyrrolo[3,2-
d]pyrim idine-2,4-diamine;
(S)-2-((5-(5-(2-(1H-tetrazol-5-yl)propan-2-y1)-2-methoxybenzy1)-2-amino-5H-
pyrrolo[3,2-
d]pyrimidin-4-y1)amino)hexan-1-01;
5-(5-(difluoro(1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-N4-pentyl-5H-
pyrrolo[3,2-
d]pyrimidine-2,4-diamine;
2-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxypheny1)-2,2-difluoroacetic acid;
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
44
(3R,5R)-7-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-
4-
methoxypheny1)-3,5-dihydroxyheptanoic acid;
2-(34(2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenypacetic acid;
(S)-2-(3-((2-amino-4-((1-hydroxyhexan-2-yl)amino)-5H-pyrrolo[3,2-d]pyrimidin-5-
y1)methyl)-4-methoxyphenyl)acetic acid;
3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxybenzoic
acid;
(3-((2-amino-4-(pentylam ino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)m ethyl)-4-
methoxyphenyl)phosphonic acid;
methyl hydrogen (3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yOmethyl)-4-
methoxyphenyl)phosphonate;
(E)-3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenypacrylic acid;
3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenyl)propanoic acid;
(S)-3-(3-((2-amino-4-((1-hydroxyhexan-2-yl)amino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)rnethyl)-4-methoxyphenyl)propanoic acid;
(S)-1-(4-((2-am ino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrim idin-5-yl)methyl)-3-
acid ;
(R)-1-(3-((2-amino-4-(pentylannino)-5H-pyrrolo[3,2-d]pyrinnidin-5-yl)methyl)-4-
methoxybenzyl)pyrrolidine-2-carboxylic acid, and
2-(4-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-3-
methoxyphenoxy)acetic acid.
16. In certain embodiments of the compounds of Formula (I), Formula (la) and
Formula (lb) the
compound is selected from:
2-(34(2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenypacetic acid ;
3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxybenzoic
acid;
5-(5-((1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-
2,4-diamine;
5-(5-(2-(1H-tetrazol-5-ypethyl)-2-methoxybenzy1)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-
2,4-diamine;
(S)-1-(4-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-3-
methoxybenzyppyrrolidine-2-carboxylic acid, and
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
(R)-1-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxybenzyppyrrolidine-2-carboxylic acid.
17. Another embodiment are methods of using compounds of Formula (I), Formula
(la) and
Formula (lb), and pharmaceutical compositions comprising such compounds of
Formula (I),
5 Formula (la) and Formula (lb).
18. Another embodiment are pharmaceutical compositions that include a
therapeutically
effective amount of a compound of Formula (I), Formula (la) or Formula (lb),
and a
pharmaceutically acceptable carrier. In certain embodiments of such
pharmaceutical
compositions, the pharmaceutical composition is formulated for intravenous
administration,
10 intravitrial administration, intramuscular administration, oral
administration, rectal
administration inhalation, nasal administration, topical administration,
ophthalmic
administration or otic administration. In other embodiments, the
pharmaceutical
compositions are in the form of a tablet, a pill, a capsule, a liquid, an
inhalant, a nasal spray
solution, a suppository, a solution, an emulsion, an ointment, eye drop or ear
drop. In other
15 embodiments, such pharmaceutical compositions further include one or
more additional
therapeutic agents.
19. Another embodiment are medicaments for treating a patient with a disease
or disorder
associated with TLR7 receptor activity, and such medicaments include a
therapeutically
effective amount of a compound of Formula (I), Formula (la) or Formula (lb).
20 20. Another embodiment is the use of a compound of Formula (I), Formula
(la) or Formula (lb)
in the manufacture of a medicament for treating a disease or disorder
associated with TLR7
activity.
21. In certain embodiments of the use of a compound of Formula (I), Formula
(la) or Formula
(lb) in the manufacture of a medicament the disease is an infectious disease,
a viral
95 infectious disease, an inflammatory disease, a respiratory disease, a
dermatological
disease, an autoimmune disease, a cell-proliferative disease or cancer.
22. In certain embodiments of such the use of a compound of Formula (I),
Formula (la) or
Formula (lb) in the manufacture of a medicament the disease is asthma, chronic
obstructive
pulmonary disease (COPD), adult respiratory distress syndrome (ARDS),
ulcerative colitis,
30 Crohn's disease, bronchitis, dermatitis, actinic keratosis, basal cell
carcinoma, bladder
cancer, allergic rhinitis, psoriasis, scleroderma, urticaria, rheumatoid
arthritis, multiple
sclerosis, cancer, breast cancer, HIV, hepatitis, hepatitis C or lupus.
23. In certain embodiments of such the use of a compound of Formula (I),
Formula (la) or
Formula (lb) in the manufacture of a medicament the disease is hepatitis B,
hepatitis C,
35 colorectal cancer or hepatocellular carcinoma.
24. Another embodiment is methods for activating a TLR7 receptor, wherein the
method
includes administering to a system or a subject in need thereof, a
therapeutically effective
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
46
amount of a compound of Formula (I), Formula (la) or Formula (lb), or
pharmaceutically
acceptable salts or pharmaceutical compositions thereof, thereby activating
the TLR
receptor.
25. In certain embodiments of such methods for activating a TLR7 receptor, the
methods
include administering the compound to a cell or tissue system or to a human or
animal
subject.
26. Another embodiment is methods for treating a disease or disorder
associated with TLR7
activity, wherein the method includes administering to a system or subject in
need of such
treatment an effective amount of a compound of Formula (I), Formula (la) or
Formula (lb), or
pharmaceutically acceptable salts thereof, thereby treating the disease or
disorder.
27. In certain embodiments of such methods for treating a disease or disorder
associated with
TLR7 activity, the methods include administering the compound to a cell or
tissue system or
to a human or animal subject.
28. In certain embodiments of such methods for treating a disease or disorder
associated with
TLR7 activity, the disease or condition is an infectious disease, a viral
infectious disease, an
inflammatory disease, a respiratory disease, a dermatological disease, an
autoimmune
disease, a cell-proliferative disease or cancer.
29. In certain embodiments of such methods for treating a disease or disorder
associated with
TLR7 activity, the disease or condition is asthma, chronic obstructive
pulmonary disease
90 (CORD), adult respiratory distress syndrome (ARDS), ulcerative colitis,
Crohn's disease,
bronchitis, dermatitis, actinic keratosis, basal cell carcinoma, bladder
cancer, allergic
rhinitis, psoriasis, scleroderma, urticaria, rheumatoid arthritis, multiple
sclerosis, cancer,
breast cancer, HIV, hepatitis, hepatitis C or lupus.
30. In certain embodiments of such methods for treating a disease or disorder
associated with
95 TLR7 activity, the disease or condition is hepatitis B, hepatitis C,
colorectal cancer or
hepatocellular carcinoma.
31. Another embodiment is methods for treating a cell-proliferative disease,
comprising
administering to a system or subject in need of such treatment an effective
amount of a
compound of Formula (I), Formula (la) or Formula (lb), or pharmaceutically
acceptable salts
30 thereof; wherein the cell-proliferative disease is bladder cancer,
lymphoma, osteosarcoma,
melanoma, or a tumor of breast, renal, prostate, colorectal, thyroid, ovarian,
pancreatic,
neuronal, lung, uterine or gastrointestinal tumor.
32. In certain embodiments of such methods for treating a cell-proliferative
disease, the the cell-
proliferative disease is, colorectal cancer or hepatocellular carcinoma.
35 33. Another embodiment is compounds for use in a method of medical
treatment, wherein the
method of medical treatment is for treating a disease associated with TLR7
receptor activity,
wherein the disease is selected from an infectious disease, a viral infectious
disease, an
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
47
inflammatory disease, a respiratory disease, a dermatological disease, an
autoimmune
disease, a cell-proliferative disease or cancer, and wherein the compound is a
compound of
Formula (I), Formula (la) or Formula (lb).
34. In certain embodiments of such use in a method of medical treatment, the
disease or
condition is asthma, chronic obstructive pulmonary disease (COPD), adult
respiratory
distress syndrome (ARDS), ulcerative colitis, Crohn's disease, bronchitis,
dermatitis, actinic
keratosis, basal cell carcinoma, bladder cancer, allergic rhinitis, psoriasis,
scleroderma,
urticaria, rheumatoid arthritis, multiple sclerosis, cancer, breast cancer,
HIV, hepatitis,
hepatitis C or lupus.
35. In certain embodiments of such use in a method of medical treatment, the
disease is
hepatitis B, hepatitis C, colorectal cancer or hepatocellular carcinoma.
Examples
The following examples illustrate the preparation of certain exemplary
compounds of
Formula (I). Table 1 gives the Human TLR7 ECK, (nM) values obtained using
these exemplary
compounds.
Synthesis of Exemplary Compounds
Example 1
5-(5-((1H-tetrazol-5-Mmethoxv)-2-methoxybenzv1)-N4-Dentyl-5H-ovrrolof3,2-
clOvrimidine-Z4-
diamine
Me0 Me0 Br CN Me0 NBS
mCPBA, Cs2CO3
CHO NaOH OH 0 ON (PhCO2)2
2 3
CI H
Me0
Me0 op /
5 H2N N CI OCN-N H2
Br OCN
/
Cs2CO3
4 H2N N Cs2CO3
Me0 6 Me0
TMSN3,
WNH Bu2SnO WNH
CDCN _________________________________
N,51 I\IT2)1
/ /
H2N N H2N N
8
7
Step 1: Preparation of 4-methoxy-3-methylphenol (2)
To a round bottom flask containing 4-methoxy-3-methylbenzaldehyde (1 equiv) in
CH2Cl2
(0.3M) was added m-CPBA (1.2 equiv) in CH2C12 (0.2M). The reaction was stirred
at room
temperature for 16 hours, quenched with excess saturated sodium thiosulfate
(aq) and treated
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
48
with excess 2N NaOH (aq). After stirring at room temperature for 16 hours, the
pH was
adjusted to 7 with NaHCO3 and aqueous layer was extracted with CH2Cl2. The
organics were
dried over Na2SO4 and concentrated under vacuum. The title compound (2) was
used crude in
the next step.
Step 2: Preparation of 2-(4-methoxy-3-methylphenoxy)acetonitrile (3)
To a round bottom flask containing the 4-methoxy-3-methylphenol (2, 1 equiv)
from the
previous step, Cs2003(2.5 equiv) and DMF (0.25M) was added bromoacetonitrile
(2 equiv).
The reaction was stirred at room temperature for 16 hours, quenched with water
and extracted
with ethyl acetate. The organics were dried over Na2SO4 and concentrated under
vacuum. The
residual was purified by silica gel chromatography to afford nitrile product
3.
Step 3: Preparation of 2-(3-(bromomethvI)-4-methoxvphenoxv)acetonitrile (4)
To a round bottom flask containing 2-(4-methoxy-3-methylphenoxy)acetonitrile
(3, 1 equiv)
from the previous step, (PhCO2)2 (0.01 equiv) and CCI4 (0.25M) was added NBS
(1 equiv). The
reaction was stirred at 75 C for 1 hour, concentrated under vacuum, and
purified by silica gel
chromatography to afford bromide 4.
Step 4: Preparation of 2-(3-((2-amino-4-chloro-5H-pyrrolo[3,2-d]pyrimidin-5-
Amethyl)-4-
methoxvphenoxvlacetonitrile (6)
To a round bottom flask containing commercially available 4-chloro-5H-
pyrrolo[3,2-
d]pyrimidin-2-amine 5 (1 equiv), DMF (0.25M), and Cs2CO3(1.2 equiv) was added
2-(3-
(bromomethyl)-4-methoxyphenoxy)acetonitrile (4, 1 equiv) from the previous
step. The reaction
was stirred at room temperature for 16 hours, quenched with water and
extracted with ethyl
acetate. The organics were dried over Na2SO4 and concentrated under vacuum.
The title
compound (6) was used crude in the next step.
Step 5: Preparation of 2-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-yl)methyl)-4-
methoxvphenoxvlacetonitrile (7)
To a round bottom flask containing 2-(3-((2-amino-4-chloro-5H-pyrrolo[3,2-
d]pyrimidin-5-
yl)methyl)-4-methoxyphenoxy)acetonitrile (6, 1 equiv) from the previous step,
Cs2CO3(2 equiv)
and DMF (0.25M) was added pentyl amine (2 equiv). The reaction was stirred at
90 C for 16
hours, quenched with water and extracted with ethyl acetate. The organics were
dried over
Na2SO4 and concentrated under vacuum. The residual was purified by silica gel
chromatography to afford the title product 7.
Step 6: Preparation of 5-(5-((1H-tetrazol-5-yl)methoxy)-2-methoxybenzyl)-N4-
pentyl-5H-
pyrrolo[3,2-d]pyrimidine-2,4-diamine (8)
To a round bottom flask containing 2-(3-((2-amino-4-(pentylamino)-5H-
pyrrolo[3,2-
d]pyrimidin-5-yl)methyl)-4-methoxyphenoxy)acetonitrile (7, 1 equiv) from the
previous step,
Bu2SnO (2 equiv) and NMP (0.25M) was added TMSN3 (10 equiv). The reaction was
stirred at
125 C for 16 hours, quenched with water and extracted with ethyl acetate. The
organics were
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
49
dried over Na2SO4 and concentrated under vacuum. The residual was purified by
reverse
phase HPLC to afford tetrazole product 8. 1H NMR (d-DMS0): 67.39 (d, 1H), 7.05
(d, 1H),
6.97 (d, 1H), 6.22 (s, 1H), 6.17 (d, 1H), 5.48 (s, 2H), 4.95 (s, 2H), 3.77 (s,
3H), 3.47-3.42 (m,
2H), 1.51-1.43 (m, 2H), 1.26 - 1.21 (m, 2H), 1.15-1.09 (m, 2H), 0.82 (t, 3H).
LRMS [M+H] =
438.2.
Example 2
5-(5-((1H-tetrazol-5-Amethyl)-2-methoxybenzyl)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-2,4-
diamine
CI
N'"
,
LJJ
Me0 Me0 H2N Ncab
NBS, A1BN 11
CN ________________________________ Br LW ON ___________
9 10 1) Cs2003,
2) pentylamine
Me0 Me0
HN-N,
TMSN3,
ON Bu2Sn3 WNH
H2N N H2N N
12 13
Step 1: Preparation of 2-(3-(bromomethyl)-4-methoxyphenyl)acetonitrile (10)
To a round bottom flask containing 2-(4-methoxy-3-methylphenyl)acetonitrile 9
(1 equiv),
AIBN (0.07 equiv) and CC14. (0.25M) was added NBS (1.1 equiv). The reaction
was stirred at 75
C for 1 hour, concentrated under vacuum, and purified by silica gel
chromatography to afford
product 10.
Step 2: Preparation of 2-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-yl)methyl)-4-
methoxyphenyl)acetonitrile (12)
To a solution of commercially available 2,4-dichloro-5H-pyrrolo[3,2-
d]pyrimidine 11 (1
equiv), Cs2003 (4 equiv) in DMF (0.3M) was added 2-(3-(bromomethyl)-4-
methoxyphenypacetonitrile (10) from the previous step (1.1 equiv). The
reaction mixture was
stirred at room temperature for 5 hours, then treated with pentylamine (3
equiv). The reaction
mixture was heated to 100 C for 16 hours, quenched with water and extracted
with ethyl
acetate. The organic layer was washed with brine, dried over Na2SO4 and
concentrated under
vacuum. The residual was purified via silica gel column chromatography to
afford product 12.
Step 3: Preparation of 5-(5-((1H-tetrazol-5-vpmethvI)-2-methoxvbenzvl)-N4-
pentyl-5H-
pyrrolo[3,2-d]pyrimidine-2,4-diamine (13)
To a round bottom flask containing the 2-(3-((2-amino-4-(pentylamino)-5H-
pyrrolo[3,2-
d]pyrimidin-5-yl)methyl)-4-methoxyphenypacetonitrile (12) from the previous
step (1 equiv),
Bu2SnO (2 equiv) and NMP (0.25M) was added TMSN3 (10 equiv). The reaction was
stirred at
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
125 C for 16 hours, quenched with water and extracted with ethyl acetate. The
organics were
dried over Na2SO4 and concentrated under vacuum. The residual was purified by
reverse
phase HPLC to afford tetrazole product 13. 1H NMR (400 MHz, DMSO-d6): 6 7.25
(d, J= 3.2
Hz, 1H), 7.16 (dd, J = 2.0, 8.4 Hz, 1H), 6.97 (d, J = 8.8 Hz, 1H), 6.73 (d, J
= 2.0 Hz, 1H), 6.70
5 (br s, 1H), 6.62 (br s, 2H), 6.04 (d, J = 3.2 Hz, 1H), 5.42 (s, 2H), 3.91
(s, 2H), 3.78 (s, 3H), 3.42-
3.36 (m, 2H), 1.45-1.41 (m, 2H), 1.29-1.20 (m, 2H), 1.17-1.09 (m, 2H), 0.83
(t, J= 7.2 Hz, 3H);
LRMS [M+H]=422.3.
Example 3
2-(5-(34(2-amino-4-(pentylamino)-5H-pyrrolo[3,2-dipyrimidin-5-yOmethyl)-4-
methoxybenzyl)-
10 2H-tetrazol-2-yl)acetic acid
Me0
/¨COOH
W NH N¨N
/
N
-LNX51/
H2N N
Preparation of 2-(5-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yOmethyl)-4-
methoxybenzy1)-2H-tetrazol-2-ypacetic acid
To a solution of 5-(5-((2H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-N4-pentyl-5H-
pyrrolo[3,2-
15 .. d]pyrimidine-2,4-diamine (13) (Example 2) in DMSO (0.15 M) was added
ethyl 2-bromoacetate
(0.95 equiv) and K2CO3(4.0 equiv). The suspension was stirred at room
temperature for 90
minutes and then purified with reverse phase HPLC to afford a white solid.
To the solution of the above white solid in Et0H/THF (1:1, v/v 0.02 M) was
added 2.0 N
NaOH (2.0 equiv). The solution was stirred at room temperature for 1 hour and
then purified
20 with pre-HPLC to afford the title compound. TFA salt = 1H NMR (400 MHz,
DMSO-d6): 57.38-
7.37 (m, 3H), 7.27(t, J= 5.6 Hz, 1H), 7.22 (dd, J= 2.4, 8.4 Hz, 1H), 7.02 (d,
J= 8.4 Hz, 1H),
6.49 (d, J= 1.6 Hz, 1H), 6.19 (d, J= 3.2 Hz, 1H), 5.53 (s, 2H), 5.50 (s, 2H),
4.06 (s, 2H), 3.80
(s, 3H), 3.44-3.39 (m, 2H), 1.46-1.38 (m, 2H), 1.26-1.16 (m, 2H), 1.09-1.02
(m, 2H), 0.80 (t, J=
7.2 Hz, 3H); LRMS [M+1-1] = 480.2.
25 Example 4
2-(34(2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yOmethyl)-4-
methoxyphenyl)acetic acid
COOH
W NH 40
N N OMe
H2N -
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
51
Preparation of 2-(34(2-amino-4-(pentvlamino)-5H-pyrrolo[3,2-dipvrimidin-5-
v1)methvI)-4-
methoxyphenyl)acetic acid
2-(34(2-Amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenyl)acetonitrile (12) (Example 2, Step 2) was suspended in Et0H: 4N
NaOH
(v/v=1:2, 0.1 M). The reaction mixture was stirred at 80 C overnight. After
neutralization with 1
N HCI, the title compound was collected by filtration as a white solid. 1H NMR
(400 MHz,
DMSO-d): 6 (d-DMS0): E7.38 (d, 1H), 7.25 (br s, 2H), 7.19 (d, 1H), 7.01 (d,
1H), 6.64 (s, 1H),
6.56 (s, 1H), 6.17 (d, 1H), 5.50 (s, 2H), 3.81 (s, 3H), 3.52-3.42 (m, 2H),
3.38 (s, 2H), 1.50-1.43
(m, 2H), 1.29-1.20 (m, 2H), 1.16-1.07 (m, 2H), 0.82 (t, 3H). LRMS [M-F1-1] =
398.2.
Example 5
(S)-2434(2-amino-4-((1-hydroxvhexan-2-v1)amino)-5H-orrolo[3,2-dloyrimidin-5-
MmethvI)-
4-methoxyphenyl)acetic acid
COON
H0,1
'ANH
NLN OMe
A
H2N N
Preparation of (S)-2-(3-((2-amino-4-((1-hydroxyhexan-2-yl)amino)-5H-
pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-methoxyphenyl)acetic acid
(S)-2-(3-((2-amino-4-((1-hydroxyhexan-2-yl)amino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-
4-methoxyphenyl)acetic acid was prepared using the same protocol as Example 4.
(S)-2-
aminohexan-1-ol was used instead of pentylamine. 1H NMR (d-DMS0): 612.23 (s,
1H), 7.51
(d, 1H), 7.36 (br, 2H), 7.21 (d, 1H), 7.04 (d, 1H), 6.57 (s, 1H), 6.23 (d,
1H), 6.21 (d, 1H), 5.61 (d,
1H), 5.45 (d, 1H), 4.32-4.24 (m, 1H), 3.83 (s, 3H), 3.38 (s, 2H), 3.38-3.33
(m, 2H), 1.52-1.43 (m,
1H), 1.24-1.08(m, 3H), 1.00-0.85 (m, 2H), 0.76 (t, 3H). LRMS [M+1-1] = 428.2.
Example 6
(S)-2-((5-(5-((1H-tetrazol-5-yOmethyl)-2-methoxybenzyl)-2-amino-5H-pyrrolo[3,2-
d]pyrimidin-
4-Mamino)hexan-1-0l
Me0
HN¨N,
NH
N
H2N N
Preparation of (S)-2-((5-(5-((1H-tetrazol-5-yOmethyl)-2-methoxybenzyl)-2-amino-
5H-pyrrolo[3,2-
d]pyrimidin-4-Aamino)hexan-1-ol
(S)-2-((5-(5-((1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-2-amino-5H-
pyrrolo[3,2-
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
52
d]pyrimidin-4-yl)amino)hexan-1-ol was prepared following the similar protocols
as Example 2.
(S)-2-aminohexan-1-ol was used instead of N-pentylamine. 1H NMR (400 MHz, DMSO-
d6): 6
7.36 (d, J= 3.2 Hz, 1H), 7.14 (dd, J= 2.0, 8.4 Hz, 1H), 6.97 (d, J= 8.8 Hz,
1H), 6.69 (d, J= 1.6
Hz, 1H), 6.34 (br s, 2H), 6.07 (d, J= 2.8 Hz, 1H), 5.63 (d, J= 8.4 Hz, 1H),
5.46 (d, J= 16.8 Hz,
1H), 5.34(d, J= 16.8 Hz, 1H), 4.28-4.20 (m, 1H), 3.88 (d, J= 15.2 Hz, 1H),
3.83 (d, J= 15.2
Hz, 1H), 3.80 (s, 3H), 3.42-3.31 (m, 2H), 1.52-1.41 (m, 1H), 1.27-1.12 (m,
3H), 1.05-0.95 (m,
2H), 0.78 (t, J= 7.6 Hz, 3H); LRMS [M+H]=452.3.
Example 7
(S)-2-((5-(5-((1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-2-amino-5H-
pyrrolo[3,2-d]pyrim1din-
4-yl)amino)pentan-1-ol
Me0
HN¨N,
NH
N
H2N N
Preparation of (S)-2-((5-(5-((1H-tetrazol-5-yOrnethyl)-2-methoxybenzy1)-2-
amino-5H-pyrrolo[3,2-
d]pyrimidin-411)amino)pentan-1-ol
(S)-2-((5-(5-((1H-tetrazol-5-yl)methyl)-2-methoxybenzyl)-2-am ino-5H-
pyrrolo[3,2-
d]pyrimidin-4-yl)amino)pentan-1-ol was prepared following the similar
protocols as Example 2.
(S)-2-aminopentan-1-ol was used instead of pentylamine. 1H NMR (400 MHz, DMSO-
d6): 6
7.45 (d, J = 3.2 Hz, 1H), 7.21 (dd, J = 2.0, 8.4 Hz, 1H), 6.34 (br s, 2H),
7.03 (d, J = 8.4 Hz, 1H),
6.62 (d, J= 2.0 Hz, 1H), 6.18 (d, J= 3.2 Hz, 1H), 6.06 (d, J= 8.4 Hz, 1H),
5.55 (d, J= 16.8 Hz,
1H), 5.41 (d, J= 16.8 Hz, 1H), 4.32-4.24 (m, 1H), 3.99 (s, 2H), 3.81 (s, 3H),
3.41-3.34 (m, 2H),
1.44-1.36 (m, 1H), 1.23-1.13 (m, 1H), 1.01-0.89 (m, 2H), 0.73(t, J= 7.2 Hz,
3H); LRMS
[M+H]=438.2.
Example 8
5-(2-methoxy-5-(1H-tetrazol-5-yl)benzyl)-N4-pentyl-5H-pyrrolo[3,2-d]pyrim
idine-2,4-diam me
Me0
W NH N.
I N
N N-K1
H2N N
Preparation of 5-(2-methoxy-5-(1H-tetrazol-5-yl)benzyl)-N4-pentyl-5H-
pyrrolo[3,2-d]pyrimidine-
2,4-diamine
5-(2-methoxy-5-(1H-tetrazol-5-yl)benzyl)-N4-pentyl-5H-pyrrolo[3,2-d]pyrimidine-
2,4-diamine
was prepared following the similar protocols as Example 2, Steps 2-3. 3-
(chloromethyl)-4-
methoxybenzonitrile (McKillop, A. et at. Tetrahedron Lett., 1983, 24, 1933-36)
was used
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
53
instead of 2-(3-(bromomethyl)-4-methoxyphenyl)acetonitrile (10) in Step 2. 1H
NMR (400 MHz,
DMSO-d6): 67.87 (dd, J= 2.0, 8.4 Hz, 1H), 7.35 (d, J= 2.0 Hz, 1H), 7.28 (d, J=
3.2 Hz, 1H),
7.09 (d, J = 8.8 Hz, 1H), 5.98 (d, J = 2.8 Hz, 1H), 5.61 (br s, 1H), 5.48 (br
s, 2H), 5.42 (s, 2H),
3.88 (s, 3H), 3.30-3.26 (m, 2H), 1.42-1.34 (m, 2H), 1.24-1.15 (m, 2H), 1.11-
1.04 (m, 2H), 0.79
(t, J= 7.6 Hz, 3H); LRMS [M+H]=408.2.
Example 9
N4-hexy1-5-(2-methoxy-5-(1H-tetrazol-5-y1)benzyl)-5H-pyrrolo[3,2-d]pyrimidine-
2,4-diamine
Me0
N
H2N N z
Preparation of N4-hexy1-5-(2-methoxy-5-(1H-tetrazol-5-y1)benzyl)-5H-
pyrrolo[3,2-d]pyrim idine-
2,4-diamine
N4- hexy1-5-(2-methoxy-5-(1H-tetrazol-5-y1)benzyl)-5 H-pyrrolo[3,2-d]pyrim
idine-2,4-diam me
was prepared following the similar protocols as Example 2, Steps 2-3. 3-
(chloromethyl)-4-
methoxybenzonitrile (McKillop, A. et at. Tetrahedron Lett., 1983, 24, 1933-36)
was used
instead of 2-(3-(bromomethyl)-4-methoxyphenypacetonitrile (10), and N-
hexylamine was used
instead of N-pentylamine in Step 2. TFA Salt = 1H NMR (400 MHz, DMSO-d6): 6
12.3 (br s, 1H),
7.98 (d, 1H), 7.51 (d, 1H), 7.40 (br s, 2H), 7.35 (s, 1H), 7.3 (dd, 2H), 6.27
(d, 1H), 5.65 (s, 2H),
3.93 (s, 3H), 3.51-3.40 (m, 2H), 1.42-1.31 (m, 2H), 1.10-0.94 (m, 6H), 0.74
(t, 3H); LRMS
[M+H]=422.3.
Example 10
(S)-2-((2-amino-5-(2-methoxy-5-(1H-tetrazol-5-yl)benzyl)-5H-pyrrolo[3,2-
d]pyrimidin-4-
y1)amino)hexan-1-ol
Me0
N
N¨N
H2N N
Preparation of (S)-2-((2-amino-5-(2-methoxy-5-(1H-tetrazol-5-yObenzy1)-5H-
pyrrolo[3,2-
dlpyrimidin-4-4amino)hexan-1-ol
(S)-2-((2-amino-5-(2-methoxy-5-(1H-tetrazol-5-yl)benzyl)-5H-pyrrolo[3,2-
d]pyrimidin-4-
y0amino)hexan-1-ol was prepared following the similar protocols as Example 2,
Step 2-3. 3-
(chloromethyl)-4-methoxybenzonitrile (McKillop, A. et al. Tetrahedron Lett.,
1983, 24, 1933-36)
was used instead of 2-(3-(brommethyl)-4-methoxyphenypacetonitrile (10), and
(S)-2-
aminohexan-1-ol was used instead of N-pentylamine in step 2. LRMS [M+H]=438.2.
Example 11
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
54
5-(5-(2-(1H-tetrazol-5-vI)propan-2-v1)-2-methoxvbenzv1)-N4-pentv1-5H-
pwrolo[3,2-
d]pyrinnidine-2,4-diannine
1)
N "
H2N Nj,õ) Me0
Cs2CO3; HN¨Nõ
Me0 Me0 then W NH
Msa. pentylamine
CN CN __________
N
Et3N OH OMs 2) TMSN3, Bu2SnO I /
14 15 H2N N16
Step1: Preparation of 5-(2-cyanopropan-2-yI)-2-methoxybenzyl methanesulfonate
(16)
To a 0 C solution of 2-(3-(hydroxymethyl)-4-methoxypheny1)-2-
methylpropanenitrile (14,
1equiv) (Shetty, R.; Moffett, K. K. Tetrahedron Lett. 2006, 47,8021) in CH2Cl2
was added Et3N
(3 equiv) and MsCI (1.2 equiv). The reaction mixture was stirred for 2 hours,
quenched with
water, and extracted with CH2Cl2. The organics were dried over Na2SO4 and
concentrated
under vacuum. The residual was used crude in the next reaction.
.. Step 2-3: Preparation of 5-(5-(2-(1H-tetrazol-5-y0propan-2-y1)-2-
methoxybenzyl)-N4-pentyl-5H-
pyrrolo[3,2-d]pyrimidine-2,4-diamine (16)
The title compound was prepared using similar protocols as Example 2, Steps 2-
3. 5-(2-
cyanopropan-2-y1)-2-methoxybenzyl methanesulfonate (15) from the previous step
was used
instead of 2-(3-(bromomethyl)-4-methoxyphenyl)acetonitrile (10). 1H NMR (400
MHz, DMS0-
d6): 67.22 (d, J= 2.8 Hz, 1H), 7.10 (dd, J= 2.4, 8.8 Hz, 1H), 7.01 (br s, 1H),
6.96 (br s, 2H),
6.95 (d, J= 8.8 Hz, 1H), 6.53 (d, J= 2.4 Hz, 1H), 6.03 (d, J= 3.2 Hz, 1H),
5.43 (5, 2H), 3.77 (s,
3H), 3.43-3.36 (m, 2H), 1.56 (s, 6H), 1.48-1.41 (m, 2H), 1.28-1.18 (m, 2H),
1.13-1.06 (m, 2H),
0.82 (t, J= 7.2 Hz, 3H); LRMS [M+H]=450.3.
Example 12
(S)-2-((5-(5-(2-(1H-tetrazol-5-yl)propan-2-y1)-2-methoxybenzy1)-2-amino-5H-
pyrrolo[3,2-
d]pyrimidin-4-y1)amino)hexan-1-ol
Me0
HN-Nõ
,
NH
N
I /
H2N N
Preparation of (S)-2-((5-(5-(2-(1H-tetrazol-5-Apropan-2-y1)-2-methoxybenzy1)-2-
am ino-5H-
pyrrolo1.3,2-d1ovrimidin-4-vpamino)hexan-1-ol
(S)-2-((5-(5-(2-(1H-tetrazol-5-yl)propan-2-y1)-2-methoxybenzy1)-2-amino-5H-
pyrrolo[3,2-
cl]pyrimidin-4-y1)amino)hexan-1-ol was prepared following the similar
protocols as Example 11.
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
(S)-2-aminohexan-1-01was used instead of N-pentylamine. 1H NMR (400 MHz, DMSO-
d6): 6
7.37 (d, 1H), 7.16 (dd, 1H), 7.12 (br s, 2H), 7.01 (d, 1H), 6.25 (d, 1H), 6.15
(d, 1H), 6.07 (d, 1H),
5.56 (d, 1H), 5.44 (d, 1H), 4.26-4.16 (m, 1H), 3.81 (s, 3H), 3.35 (d, 2H),
1.58 (s, 3H), 1.53 (s,
3H), 1.39-1.24 (m, 1H), 1.24-1.08 (m, 3H), 1.00-0.84 (m, 2H), 0.75 (t, 3H);
LRMS [M+H]=480.3.
5 Example 13
34(2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yOmethyl)-4-
methoxybenzoic acid
Me0 Me0
W NH CO "P Me NaOH wNH ggP CO2H I 2
N
H2N N H2N N
17 18
Step 1: Preparation of methyl 3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-
yl)methyl)-4-methoxybenzoate (17)
10 Methyl 3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-
methoxybenzoate (17) was prepared following the same protocol as Example 2,
Steps 2-3.
Methyl 3-(chloromethyl)-4-methoxybenzoate was used in place of 2-(3-
(bromomethyl)-4-
methoxyphenypacetonitrile (10).
Step 2: Preparation of 3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-
5-yl)methyl)-4-
15 methoxybenzoic acid (18)
To a solution of Methyl 3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-
5-yl)methyl)-
4-methoxybenzoate (17 from the previous step, 1 equiv) in EtOH (0.1M) was
added 2N NaOH
(2equiv). The reaction mixture was stirred at 80 C overnight and the entire
reaction was
purified via reverse phase HPLC to afford product. TEA Salt = 1H NMR (400 MHz,
DMSO-d6): 6
20 7.90 (d, 1H), 7.50 (d, 1H), 7.41 (s, 2H), 7.37 (t, 1H), 7.15 (d, 1H),
7.13 (d, 1H), 6.24 (d, 1H),
5.60 (s, 2H), 3.91 (s, 3H), 3.44 (q, 2H), 1.46-1.38 (m, 2H), 1.20-1.10 (m,
2H), 1.03-0.95 (m, 2H),
0.76 (t, 3H). LRMS [M+H] = 384.2.
Example 14
5-(2-methoxv-4-(1H-tetrazol-5-Mbenzyn-N4-Dentv1-5H-pyrrolo13,2-dlpyrimidine-Z4-
diamine
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
56
Me0 CN
Me0
Br
CI 1) WNH 4k, CN
Cs2CO3 1 n. NaN3, ZnCl2
Cr -N 2)WNH2 Cr -N NMP
19
Me0 HN¨N Me0 HN¨N
µN-N W NH ft \N-N
N kX.51 Zn
1\13' -N acetic acid H2N N
20 21
Step 1: Preparation of 4-((2-chloro-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-
5-Arnethyl)-3-
methoxybenzonitrile (19)
To a solution of commercially available 2,4-dichloro-5H-pyrrolo[3,2-
d]pyrimidine (1 equiv),
Cs2CO3 (4 equiv) in DMF (0.3M) was added commercially available 4-
(bromomethyl)-3-
methoxybenzonitrile (1.1 equiv). The reaction mixture was stirred at room
temperature for 5
hours, then treated with pentylamine (3 equiv). The reaction mixture was
heated to 100 C for
16 hours, quenched with water and extracted with ethyl acetate. The organic
layer was washed
with brine, dried over Na2SO4 and concentrated under vacuum. The residual was
purified via
silica gel column chromatography to afford product 19.
Step 2: Preparation of 2-azido-5-(2-methoxy-4-(1H-tetrazol-5-yl)benzyl)-N-
pentyl-5H-
pyrrolo[3,2-d]pyrimidin-4-amine (20)
A slurry of 4-((2-chloro-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-3-
methoxybenzonitrile (19) from the previous step (1 equiv.), ZnCl2 (10 equiv.)
and NaN3 (15
equiv.) in NMP (0.3M) was heated to 150 C for 4 hours then cooled to room
temperature and
diluted with water upon which product 20 precipitates. Product 20 was isolated
by vacuum
filtration and used crude in the next step.
Step 3: Preparation of 5-(2-methoxy-4-(1H-tetrazol-5-yObenzy1)-N4-pentyl-5H-
pyrrolo[3,2-
d]pyrimidine-2,4-diamine (21)
A slurry of crude 2-azido-5-(2-methoxy-4-(1H-tetrazol-5-yl)benzyl)-N-pentyl-5H-
pyrrolo[3,2-
d]pyrimidin-4-amine (20, from previous step, 1 equiv.) and Zn dust (30 equiv.)
in AcOH (0.3M)
was heated to 80 C for 15 hours then cooled to room temperature, diluted with
10%
Me0H/CH2012 and filtered through a pad of celite. The filtrate was
concentrated under vacuum
and purified by reverse phase HPLC to afford product 21. TFA Salt = 1H NMR
(400 MHz,
DMSO-de): 612.4 (br s, 1H), 7.71 (d, J= 1.6 Hz, 1H), 7.55 (dd, J= 1.2, 7.6 Hz,
1H), 7.51 (d, J=
3.2 Hz, 1H), 7.45 (br s, 2H), 7.35 (t, J = 6.0 Hz, 1H), 6.62 (d, J = 5.6 Hz,
1H), 6.26 (d, J = 2.8
Hz, 1H), 5.66 (s, 2H), 3.96 (s, 3H), 3.43 (q, J = 6.4 Hz, 2H), 1.45-1.37 (m,
2H), 1.17-1.08 (m,
2H), 1.02-0.95 (m, 2H), 0.67 (t, J= 7.2 Hz, 3H); LRMS [M+H]=408.2.
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
57
Example 15
5-(5-(2-(1H-tetrazol-5-vUethyl)-2-methoxvbenzvl)-N4-Dentvl-5H-Dyrrolof3,2-
d1Dvrimidine-24-
diamine
Me0 Me0
Me0 rai 1) DIBAL WI) 1) Pd(0A02,
2) MsCI, Et3N W NH iAcrylonitrile WNH
Me02C 3) CI 1\1X..,N) 2) Pd/C, H2 N I N
N- 11
22 I / 3) NaN3, ZnCl2
N H2N N H2N N
I / 23 24
H2N N
Cs2CO3
4) pentyl amine
Steps 1-3: Preparation of 5-(5-iodo-2-methoxybenzy1)-N4-penty1-5H-pyrrolo[3,2-
d]pyrimidine-
2,4-diamine (23)
To a solution of methyl 5-iodo-2-methoxybenzoate 22 (1 equiv) in THE (0.3M) at
0 C was
added DIBAL (2.1 equiv). The reaction mixture was allowed to warm to room
temperature for 1
hour, then cooled to 0 C and quenched with saturated Na2SO4 (aq) until a fine
white solid was
formed. Excess solid Na2SO4 was added and the reaction mixture was filtered
through a pad of
Celite and washed with ether. The filtrate was concentrated under vacuum to
afford a white
solid. The white solid was dissolved in CH2Cl2 (0.3M), cooled to 0 C and
treated with Et3N (2
equiv) and MsCI (1.2 equiv). The reaction mixture was stirred for 3 hours,
quenched with water
and extracted with CH2Cl2. The organics were dried over Na2SO4 and
concentrated under
vacuum. The residual was used crude in the next reaction.
To a solution of commercially available 2,4-dichloro-5H-pyrrolo[3,2-
d]pyrimidine (1 equiv),
Cs2CO3 (4 equiv) in DMF (0.3M) was added the residual from above (1.1 equiv).
The reaction
mixture was stirred at room temperature for 5 hours, then treated with
pentylamine (3 equiv).
The reaction mixture was heated to 100 C for 16 hours, quenched with water and
extracted
with ethyl acetate. The organic layer was washed with brine, dried over Na2SO4
and
concentrated under vacuum. The residual was purified via silica gel column
chromatography to
afford product 23.
Steps 4-6: Preparation of 5-(5-(2-(2H-tetrazol-5-yOethyl)-2-methoxybenzyl)-N4-
pentyl-5H-
pyrrolo[3,2-d]pyrimidine-2,4-diamine (24)
95 To a solution of 5-(5-iodo-2-methoxybenzy1)-N4-penty1-5H-pyrrolo[3,2-
d]pyrimidine-2,4-
diamine (23) from the previous step in DMF (1.0 M) was added palladium acetate
(0.3 equiv),
triethylamine (1.3 equiv) and acrylonitrile (1.0 equiv). The reaction micture
was heated at 100
C for 40 minutes with microwave. It was cooled down to room temperature and
purified with
reverse phase HPLC to afford the product for next reaction.
To a solution of the product above in Me0H (0.1 M) was added palladium on
carbon (10%
wt., 0.3 equiv) and the suspension was stirred under hydrogen for 1 hour at
room temperature.
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
58
The catalyst was filtered out and the filtrate was concentrated for silica gel
column
chromatography to afford the product. To a solution of the product above in
NMP (0.1 M) was
added zinc chloride (7.0 equiv) and sodium azide (7.0 equiv). The suspension
was heated to
150 C for 20 hours and cooled down for reverse phase HPLC purification to
afford 5-(5-(2-(2H-
tetrazol-5-ypethyl)-2-methoxybenzy1)-N4-pentyl-5H-pyrrolo[3,2-d]pyrimidine-2,4-
diamine (24).
TFA salt = 1H NMR (400 MHz, DMSO-d6): 512.16, (s, 1H), 7.36 (s, 2H), 7.31 (d,
J = 2.8, 1H),
7.28 (t, J= 5.6 Hz, 1H), 7.17 (dd, J= 2.0, 8.4 Hz, 1H), 6.99 (d, J = 8.4 Hz,
1H), 6.56 (d, J= 1.6
Hz, 1H), 6.19 (d, J = 2.8 Hz, 1H), 5.47 (s, 2H), 3.78 (s, 3H), 3.48-3.43 (m,
2H), 3.04 (t, J = 7.2
Hz, 2H), 2.88 (t, J = 7.2 Hz, 2H), 1.50-1.43 (m, 2H), 1.26-1.19 (m, 2H), 1.13-
1.07 (m, 2H), 0.81
(t, J= 7.2 Hz, 3H); LRMS [M+H] = 436.3.
Example 16
5-(5-(difluoro(1H-tetrazol-5-Amethyl)-2-methoxybenzyl)-N4-pentyl-5H-pyrrolop,2-
0yrimidine-2,4-diamine
Me0 Me0 io i) 7N NH3/Me0H
1$ Cu powder CO2Et 2) AIBN, NBS
____________________________ =
Br CO2Et
F F 3) CI H
25 F F 26
H2N N
CS2CO3
4) pentyl amine
Me0
Me0 HN¨N,
0 µ,
WNH 1) (CF3C0)20, W NH
N
NH2 pyridine
F F
H2N
F
ZnCl2, NaN3
/
H2NN / 28
N 27
Step 1: Preparation of ethyl 2,2-difluoro-2-(4-methoxy-3-methylphenyl)acetate
(26)
To a solution 4-iodo-1-methoxy-2-methylbenzene (1 equiv) and ethyl 2-bromo-2,2-
difluoroacetate (2 equiv) in DMF (0.3M) was added Cu powder (3 equiv). The
reaction slurry
was heated to 80 C for 1.5 days, quenched with saturated NaH2PO4 (aq) and
extracted with
ethyl acetate. The organics were dried over Na2SO4 and concentrated under
vacuum. The
residual was purified via silica gel column chromatography to afford product
26.
Steps 2-4: 2-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-
methoxypheny1)-2,2-difluoroacetamide (27)
Ethyl 2,2-difluoro-2-(4-methoxy-3-methylphenyl)acetate (26) from the previous
step was
treated with 7N ammonia in methanol (5 equiv). The reaction mixture was
stirred at room
temperature for 2 hours, then concentrated under vacuum to afford a white
solid. The white
solid was dissolved in CCI4 (0.3M) and treated with AIBN (0.08 equiv) and NBS
(1.2 equiv).
The reaction slurry was stirred at 70 C for 15 hours. The solids were removed
by vacuum
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
59
filtration. The filtrated was concentrated under vacuum and the residual was
used crude in the
next reaction.
To a solution of commercially available 2,4-dichloro-5H-pyrrolo[3,2-
d]pyrimidine (1 equiv),
Cs2003 (4 equiv) in DMF (0.3M) was added the residual from above (1.1 equiv).
The reaction
mixture was stirred at room temperature for 5 hours, then treated with
pentylamine (3 equiv).
The reaction mixture was heated to 100 C for 16 hours, quenched with water and
extracted
with ethyl acetate. The organic layer was washed with brine, dried over Na2SO4
and
concentrated under vacuum. The residual was purified via silica gel column
chromatography to
afford product 27.
Steps 5-6: Preparation of 5-(5-(difluoro(1H-tetrazol-5-yOmethyl)-2-
methoxybenzyl)-N4-pentyl-
5H-pyrrolo[3,2-dlovr1midine-2,4-diamine (28)
To 2-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxypheny1)-2,2-difluoroacetamide (27) from the previous step in CH2Cl2
(0.1M) at 0 C was
added pyridine (10 equiv), followed by trifluoroacetic anhydride (1.3 equiv).
After 3 hours,
additional trifluoroacetic anhydride (1.3 equiv) was added. The reaction
mixture was stirred for
an additional 3 hours, quenched with saturated NaHCO3 (aq) and extracted with
CH2Cl2. The
organic layer was washed with brine, dried over Na2SO4 and concentrated under
vacuum. The
residual was dissolved in NMP (0.3M) and treated with NaN3 (7 equiv) and ZnCl2
(7 equiv). The
reaction mixture was heated to 100 C for 16 hours. The reaction mixture was
purified by
reverse phase HPLC to afford 5-(5-(difluoro(1H-tetrazol-5-yl)methyl)-2-
methoxybenzyl)-N4-
pentyl-5H-pyrrolo[3,2-d]pyrimidine-2,4-diamine (28). TFA salt = 1H NMR (400
MHz, DMSO-d6):
6 12.22 (s, 1H), 7.53 (dd, 1H), 7.44 (d, 1H), 7.42-7.35 (m, 3H), 7.20 (d, 1H),
6.76 (d, 1H), 6.22
(d, 1H), 5.59 (s, 2H), 3.88 (s, 3H), 3.45-3.35 (m, 2H), 1.44-1.31 (m, 2H),
1.24-1.11 (m, 2H),
1.08-0.93 (m, 2H), 0.77 (t, 3H); LRMS [M+H]=458.2.
Example 17
2-(3-((2-amino-4-(pentylamino)-5H-pyrrolof3,2-dlpyrimidin-5-vOmethyl)-4-
methoxypheny1)-Z2-
difluoroacetic acid
Me0
Me0 0 1) NBS, AIBN CI 0 1) LION
2) CI N ) F 2)
F
N '--LX1r1 F
A 26
H2N N> 29
Cs2CO3 Me0
W NH 0
NLN F OH
H2N N
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
Steps 1-2: Preparation of ethyl 2-(34(2-amino-4-chloro-5H-pyrrolo[3,2-
dlpyrimidin-5-yl)methyl)-
4-nriethoxypheny1)-2,2-difluoroacetate (29)
Ethyl 2,2-difluoro-2-(4-methoxy-3-methylphenyl)acetate (26) (from Example 16,
Step 1) was
dissolved in CCI4(0.3 M). NBS (1.0 equiv.) and AIBN (0.05 equiv.) were added,
and the reaction
5 micture was stirred at 80 C for 2 hours. It was cooled down to room
temperature and
concentrated. The residue was purified with silica gel column chromatography
to afford a
colorless oil.
To a solution of comercially available 4-chloro-5H-pyrrolo[3,2-d]pyrimidin-2-
amine (1.0
equiv) and Cs2CO3 (1.3 equiv) in DMF was added the oil from above (1.0 equiv).
The reaction
10 mixture was stirred at room temperature for 5 hours. It was quenched
with water and extracted
with 10% Me0H/DCM (v/v). The combined organic extracts were concentrated and
purified by
silica gel column chromatography to afford the compound (29) as a white solid.
Steps 3-4: Preparation of 2-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-
yl)methyl)-4-methoxypheny1)-2,2-difluoroacetic acid (30)
15 To a solution of ethyl 2-(3-((2-amino-4-chloro-5H-pyrrolo[3,2-
d]pyrimidin-5-yl)methyl)-4-
methoxypheny1)-2,2-difluoroacetate (29) in water/Me0H (1:1 v/v, 0.3 M) from
the previous step
was added LiOH (1.4 equiv) and it was stirred for 10 minutes at room
temperature. It was
quenched with 1N HCI (1.0 equiv). The solution was concentrated and the
residue was used
crude for the next reaction.
90 To a solution of the residue above in NMP (0.1 M) was added pentyl amine
(3.0 equiv). The
solution was heated at 100 C for 2 hours and cooled down to room temperature
for reverse
phase HPLC purification to afford 2-(3-((2-amino-4-(pentylamino)-5H-
pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-methoxypheny1)-2,2-difluoroacetic acid (30) as a white solid. 1H
NMR (400 MHz,
DMSO-d6): 6 7.47-7.44 (m, 3H), 7.41 (d, J = 2.8 Hz, 1H), 7.23 (s, 1H), 7.09
(d, J= 8.4 Hz, 1H),
25 7.00 (d, J= 1.6 Hz, 1H), 6.10 (d, J= 3.2 Hz, 1H), 5.54 (s, 2H), 3.85 (s,
3H), 3.47 -3.41 (m, 2H),
1.51-1.44 (m, 2H), 1.27-1.20 (m, 2H), 1.15-1.10 (m, 2H), 0.82(t, J= 7.2 Hz,
3H); LRMS [M+H]
= 434.1.
Example 18
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
61
(3R.5R)-743-((2-amino-4-(oentylamino)-5H-ovrrolo13,2-dlovrimidin-5-v1)methyl)-
4-
methoxypheny1)-3,5-dihydroxyheptanoic
1) TBSCI, Et3N
Me0 io 2) LAH Me0
3) Mn02
40 LiHMDS
CO2Me CHO
Ph
OH OTBS
31 32
N ,sr tBu
00 66 0
33 /
o
1) TBAF
2) MsCI, Et3N WNH OtBu
OtBu _________________________________
OTBS 0,,,0 0 3) CI 0
34 H2N
H2N N
Cs2CO3
4) pentyl amine
1) Pd/C, H2
2) HCI
3) NaOH W NH OH
OH OH 0
36
acid H2N N
Steps 1-3: Preparation of 3-(((tert-butyldimethylsilypoxy)methyl)-4-
methoxybenzaldehyde (32)
5 To a DMF (0.3M) solution of commercially available methyl 3-
(hydroxymethybI)-4-
methoxybenzoate (31) (1 equiv) was added Et3N (3 equiv) and TBSCI (1.4 equiv).
The
reaction mixture was stirred at room temperature for 15 hours, quenched with
water and
extracted with ethyl acetate. The organic layer was washed with brine, dried
over Na2SO4 and
concentrated under vacuum. The residual was dissolved in THE (0.3M) and added
to a 0 C
10 THF (0.3M) slurry of LAH (1 equiv). The reaction was allowed to warm to
room temperature for
1 hour, cooled to 0 C and quenched with saturated Na2SO4 (aq) until a fine
white solid was
formed. Excess solid Na2SO4 was added and the reaction mixture was filtered
through a pad of
Celite and washed with ether. The filtrate was concentrated under vacuum to
afford a clear oil.
The clear oil was dissolved in 1,4-dioxane (0.6M) and treated with Mn02 (2.1
equiv). The
15 reaction mixture was ref luxed for lh, diluted with ethyl acetate and
filtered through a pad of
celite. The filtrate was concentrated and the residual was purified via silica
gel column
chromatography to afford aldehyde product 32.
Step 4: Preparation of tert-butyl 2-((4R,65)-6-(3-(((tert-
butyldimethylsilyl)oxy)methyl)-4-
methoxystyry1)-2,2-dimethyl-1,3-dioxan-4-y1)acetate (34)
20 To a -78 C THF (0.3M) solution of aldehyde 32 from the previous step (1
equiv) and
commercially available sulfone 33 (1.3 equiv) was added LHMDS (2.6 equiv). The
reaction
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
62
mixture was stirred at -78 C for 1 hour, quenched with saturated NaHCO3 (aq)
and extract with
ethyl acetate. The organic layer was washed with brine, dried over Na2SO4 and
concentrated
under vacuum. The residual was purified via silica gel column chromatography
to afford alkene
product 34.
Steps 5-7: Preparation of tert-butyl 2-((4R,65)-6-(3-((2-amino-4-(pentylamino)-
5H-pyrrolo[3,2-
d]pyrimidin-5-yl)methyl)-4-methoxystyry1)-2,2-dimethyl-1,3-dioxan-4-yOacetate
(35)
To a THF (0.3M) solution of alkene 34 from the previous step (1 equiv) was
added 1M
TBAF/THF (1.5 equiv). The reaction mixture was stirred at room temperature for
1 hour,
quenched with water and extracted with ethyl acetate. The organic layer was
washed with
brine, dried over Na2SO4 and concentrated under vacuum. The residual was
dissolved in
CH2Cl2 (0.3M), cooled to 0 C and treated with Et3N (2 equiv) and MsCI (1.2
equiv). The
reaction mixture was stirred for 3 hours, quenched with water and extracted
with CH2Cl2. The
organics were dried over Na2SO4 and concentrated under vacuum. The residual
was used
crude in the next reaction.
To a solution of commercially available 2,4-dichloro-5H-pyrrolo[3,2-
d]pyrimidine (1 equiv),
Cs2CO3 (4 equiv) in DMF (0.3M) was added the residual from above (1.1 equiv).
The reaction
mixture was stirred at room temperature for 5 hours, then treated with
pentylamine (3 equiv).
The reaction mixture was heated to 100 C for 16 hours, quenched with water and
extracted
with ethyl acetate. The organic layer was washed with brine, dried over Na2SO4
and
concentrated under vacuum. The residual was purified via silica gel column
chromatography to
afford product 35.
Steps 8-10: Preparation of (3R,5R)-7-(34(2-amino-4-(pentylamino)-5H-
pyrrolo[3,2-d]pyrimidin-
5-yl)methyl)-4-methoxypheny1)-3,5-dihydroxyheptanoic acid (36)
An ethyl acetate (0.3M) slurry of tert-butyl 2-((4R,65)-6-(3-((2-amino-4-
(pentylamino)-5H-
pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-methoxystyry1)-2,2-dimethyl-1,3-dioxan-
4-y1)acetate (35)
from the previous step (1 equiv) and 10% Pd on carbon (1 equiv) was purged via
bubbling with
a hydrogen balloon for 15 minutes. The reaction mixture was stirred under a
hydrogen
atmosphere for 3 hours, dilute with ethyl acetate and filtered over a pad of
celite. The filtrate
was concentrated and residual was dissolved in MeCN (0.03M) and treated with
0.1M HCI (3
equiv). The reaction mixture was stirred at room temperature for 15 hours and
treated with 1M
NaOH (11 equiv). The reaction mixture was stirred for 15 hours, the pH was
adjusted to 7-8
and the entire reaction was purified via reverse phase HPLC to afford product
36. 1H NMR (400
MHz, CD30D): 6 7.41 (d, 1H), 7.17 (dd, 1H), 6.99 (d, 1H), 6.45 (d, 1H), 6.24
(d, 1H), 5.54-5.45
(m, 2H), 4.05-3.96 (m, 1H), 3.90 (s, 3H), 3.66-3.58 (m, 1H), 3.55-3.42 (m,
2H), 2.64-2.54 (m,
1H), 2.54-2.45 (m, 1H), 2.33-2.22 (m, 2H), 1.62-1.40(m, 6H), 1.33-1.21 (m,
2H), 1.15-1.05 (m,
2H), 0.87 (t, 3H); LRMS [M+H]=500.3.
Example 19
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
63
(34(2-amino-4-(pentylamino)-5H-0yrrolo13,2-dipyrimidin-5-v11methyl)-4-
methoxyphenyl)phosphonic acid
and
Example 20
methyl hydrogen (3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-cl]pyrimidin-5-
yl)methyl)-4-
methoxyphenyl)phosphonate
Me0 Pd(OAc)2, Me 1) NBS, AIBN
= ____________________________________________________ OMe
W I P(OMe)3 P1-0Me 2) ci
8
37 38
H2N /
N
Cs2CO3
3) pentylamine
Me0
OH
W NHNN
12-0
8 H
I /
Me0 H2N N
OMe P'-0Me TMSI 40a
WNH
N 8 Me0
OM
H2N N
W P-0
39 8 "
I /
H2N N
40b
Step 1: Preparation of dimethvl (4-methoxy-3-methylphenvl)0hos1honate (38)
4-lodo-1-methoxy-2-methylbenzene 37 (1 equiv., commercially available),
trimethyl
phosphite (1.1 equiv.), and palladium acetate (0.1 equiv.) were added to
acetonitrile (0.4 M) in a
microwave tube, and was irradiated at 160 C for 1 hour with stirring. ISCO
purification (silica
column) afforded the title product 38 as a colorless oil.
Steps 2-3: Preparation of dimethyl (34(2-amino-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-
yl)methyl)-4-methoxyphenyl)phosphonate (39)
Dimethyl (3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-
4-
methoxyphenyl)phosphonate (39) was prepared following the same protocol as
Example 2,
Steps 1-2, using dimethyl (4-nnethoxy-3-methylphenyl)phosphonate (38) instead
of 2-(4-
methoxy-3-methylphenyl)acetonitrile (9) in Step 1.
Preparation of (3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrim idin-5-
yOmethyl)-4-
nnethoxyphenyl)phosphonic acid (40a) and methyl hydrogen (3-((2-amino-4-
(pentylannino)-5H-
pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-methoxyphenyl)phosphonate (40b)
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
64
Dimethyl (3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yOmethyl)-4-
methoxy-
phenyl)phosphonate (39) from the previous step was treated with TMSI to afford
(3-((2-amino-4-
(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenyl)phosphonic acid (40a)
LRMS [M+ H] = 420.2, and methyl hydrogen (3-((2-amino-4-(pentylamino)-5H-
pyrrolo[3,2-
d]pyrimidin-5-yl)methyl)-4-methoxyphenyl)phosphonate (40b) 1H NMR (DMSO-d6): 8
12.38 (s,
1H), 7.62 (dd, 1H), 7.46 (d, 1H), 7.44 (s, 2H), 7.39 (t, 1H), 7.18 (dd, 1H),
6.83 (d, 1H), 6.23 (d,
1H), 5.60 (s, 2H), 3.87 (s, 3H), 3.43 (q, 2H), 3.35 (d, 3H), 1.48-1.38 (m,
2H), 1.24-1.16 (m, 2H),
1.10-1.01 (m, 2H), 0.80 (t, 3H). LRMS [M+H] = 434.2
Example 21
3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-Amethyl)-4-
methoxvphenvi)propanoic acid
CI H
Br
I /
NBS, A1BN H2N N
= _____________________________ Br __________________ 0 = Br
0 2)
41
H2N N N'BO H2NINXH
/ 0 \
H2, Pd/C
0 0
Br
Pd2dba3
42 4
3
H2NyN N NaOH H2N
0 0
0 0
44 45
Step 1: Preparation of 4-bronno-2-(bronnomethyI)-1-methoxybenzene (41)
4-bromo-1-methoxy-2-methylbenzene (commercially available, 1.0 equiv.) was
dissolved in
chloroform (0.1 M), and was treated with N-bromosuccinimide (1.1 equiv.) and
azobisisobutyronitrile (AIBN, catalytic amount). The reaction mixture was
stirred at ref lux
overnight. After cooling down to room temperature, the reaction mixture was
purified by ISCO
(silica gel column, Et0Ac/hexanes) to afford 4-Bromo-2-(brornomethyl)-1-
methoxybenzene as a
white solid (41).
Step 2: Preparation of 5-(5-bromo-2-methoxybenzy1)-N4-penty1-5H-pyrrolo[3,2-
d]pyrimidine-2,4-
diamine (42)
4-Chloro-5H-pyrrolo[3,2-d]pyrimidin-2-amine (cornmercially available, 1
equiv.) was
dissolved in NMP (0.1 M) and stirred at room temperature under N2. Potassium
carbonate (1
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
equiv.) was added, followed by addition of 4-Bromo-2-(bromomethyl)-1-
methoxybenzene 41
(from the previous step, 1 equiv.) to give a suspension. The reaction was
stirred at room
temperature for 18 hours and LCMS shows complete conversion. N-pentyl amine (2
equiv.) was
added, followed by potassium carbonate (1 equiv.). The reaction mixture was
stirred at 60 C
5 overnight. After cooling down to room temperature, the reaction mixture
was concentrated en
vacuuo, and was purified by ISCO chromatography (0 ¨ 100% Et0Ac in hexanes) to
afford
product 5-(5-Bromo-2-methoxybenzy1)-N4-penty1-5H-pyrrolo[3,2-d]pyrimidine-2,4-
diamine 42 as
a white solid.
Step 3: Preparation of (E)-ethvl 3-(34(2-am ino-4-(pentvlamino)-5H-pyrrolo1-
3,2-d1ovrim idin-5-
10 yOmethyl)-4-methoxyphenyl)acrylate (43)
5-(5-Bromo-2-methoxybenzy1)-N4-penty1-5H-pyrrolo[3,2-d]pyrimidine-2,4-diamine
42 (from
the previous step, 1 equiv.), (E)-ethyl 3-(4,4,5,5-tetramethy1-1,3,2-
dioxaborolan-2-yl)acrylate
(1.5 equiv.), tris(dibenzylideneacetone)dipalladium(0) (0.1 equiv.), 2-
dicyclohexylphosphino-
2',6'-dimethoxybiphenyl (Sphos, 0.2 equiv.) and K3PO4 (2 equiv.) were
dissolved in 4:1 n-
15 butanol:water (0.1 M). After degassing with N2, the vessel was sealed
and heated at 100 C
overnight. After cooling down to room temperature, the reaction mixture was
quenched with
equal volume of saturated sodium bicarbonate aqueous solution, and was
extracted with DCM.
The organic layers were combined and dried over anhydrous sodium sulfate, and
concentrated
in vacuuo. The crude was purified by reverse phase prep-HPLC to afford title
compound (43) as
20 a white solid.
Step 4: Preparation of ethyl 3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-
yOmethyl)-4-methoxyphenyl)propanoate (44)
(E)-Ethyl 3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrim idin-5-
yl)methyl)-4-
methoxyphenyl)acrylate (43) (from the previous step, 1 equiv.) and 10%
palladium on carbon
25 (0.1 equiv.) were suspended in ethanol (0.1 M). The mixture was stirred
under H2 atmosphere
overnight with stirring. Solvents were removed under vacuum, and the crude was
purified by
ISCO (DCM-Et0Ac, silica gel) to afford product (44) as white solid.
Step 5: Preparation of 3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3.2-
d]pyrimidin-5-y1)methyl)-4-
methoxyphenyl)propanoic acid (45)
30 Ethyl 3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-
methoxypheny1)-propanoate (44) (from the previous step, 1 equiv) was dissolved
in Et0H and
2N NaOH (2equiv) was added. The reaction mixture was stirred at 80 C
overnight, the entire
reaction was purified via reverse phase HPLC to afford product 45. 1H NMR
(Me0D) 67.37 (d,
1H), 7.22 (1H), 7.01 (d, 1H), 6.67 (s, 1H), 6.22 (d, 1H), 5.49 (s, 2H), 3.89
(s, 3H), 3.53 (t, 2H),
35 2.76 (t, 2H), 2.46 (t, 2H), 1.52-1.45 (m, 2H), 1.36 - 1.24 (m, 2H), 1.18
- 1.10 (m, 2H), 0.88 (t, J
= 7.3, 3H). LRMS [M-F1-1] = 412.2
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
66
Example 22
(E)-3-(3-((2-amino-44Pentylam ino)-5H-pyrrolo[3,2-dlpyrimidin-5-yl)methyl)-4-
methoxyphenyl)acrylic acid
H2N N
0
0
OH
Preparation of (E)-3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-dlpyrim idin-
5-yl)methyl)-4-
methoxyphenyl)acrylic acid
(E)-3-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenypacrylic acid was prepared using similar protocols as Example 21,
Step 5. (E)-
ethyl 3-(3-((2-amino-4-(pentylam ino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)nn
ethyl)-4-
methoxyphenyl)acrylate (43, from Example 20, Step 3) was used instead of ethyl
3-(3-42-
amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-4-
methoxyphenyl)propanoate
(44). 1H NMR (Me0D): ö7.61 (d, 1H), 7.51 (d, 1H), 7.40 (d, 1H), 7.13 (d, 1H),
6.95 (s, 1H),
6.26 (d, 1H), 6.18 (d, 1H), 5.56 (s, 2H), 3.94 (s, 3H), 3.55 (t, 2H), 1.52-
1.44 (m, 2H), 1.30-1.21
(m, 2H), 1.14 - 1.07 (m, 2H), 0.84 (t, J = 7.3, 3H). LRMS [M+1-1] = 410.2
Example 23
(S)-3-(34(2-amino-4-((1-hydroxyhexan-2-yl)amino)-5H-pyrrolo[3,2-d]pyrimidin-5-
y1)methyl)-4-
methoxyphenyl)propanoic acid
Me0
NH CO2H
N Jx51
H2NN /
Preparation of (S)-3-(3-((2-amino-4-((1-hydroxyhexan-2-yl)amino)-5H-
pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-methoxyphenyl)propanoic acid
(S)-3-(3-((2-amino-4-((1-hydroxyhexan-2-yl)amino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-
4-methoxyphenyl)propanoic acid was prepared using similar protocols as Example
21. (S)-2-
aminohexan-1-ol was used instead of N-pentylamine in Step 2. 1H NMR (d-DMS0):
57.31 (d,
1H), 7.14 (d, 1H), 6.99 (d, 1H), 6.45 (s, 1H), 6.01 (d, 1H), 5.70 (br, 2H),
5.47 (d, 1H), 5.29 (d,
1H), 5.15 (d, 1H), 4.18-4.12 (m, 1H), 3.82 (s, 3H), 3.36-3.25 (m, 2H), 2.60
(t, 2H), 2.32 (t, 2H),
1.49-1.40 (m, 1H), 1.17-1.08(m, 3H), 0.95-0.88 (m, 2H), 0.75 (t, 3H). LRMS
[M+H] = 442.2.
Example 24
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
67
(R)-1-(34(2-amino-4-(ventvlamino)-5H-pyrrolo13,2-dlovrimidin-5-v1)methvI)-4-
methoxybenzyl)pyrrolidine-2-carboxylic acid
Me0 Me0
WNH DIBAL
41
SOCl2
-NH Me k
CI
N5--LX) CO2Me
KI5LX5 H 1 I /
H2N N / H2N N
H2N N
46 47
Me0
HN0 Me0 TEA -'NH
e02tBe''NH WI NO N-5LX.NI) CO2H
N 'CO2tBu /
/ FI2N N
H2N N 49
48
Step1: Preparation of (3-q2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-
methoxyphenyl)methanol (46)
To a solution of methyl 3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-
5-yl)methyl)-
4-methoxybenzoate (from Example 13, Step 1) in THE (0.1M) at 0 C was added
DIBAL (4
equiv). The reaction mixture was allowed to warm to room temperature for 1
hour, cool to 0 C
and quenched with saturated Na2SO4 (aq) until a fine white solid was formed.
Excess solid
Na2SO4 was added and the reaction mixture was filtered through a pad of
Celite. The filtrate
was concentrated under vacuum and purified by ISCO (silica gel column,
Me0H/DCM) to afford
a white solid.
Step 2: Preparation of 5-(5-(chloromethyl)-2-methoxybenzy1)-N4-pentyl-5H-
pyrrolo[3,2-
d]pyrimidine-2,4-diamine (47)
To a solution of (3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-4-
rnethoxyphenyOrriethanol (46) (from previous step, 1 equiv) in chloroform
(0.1M) at 0 C, was
added SOCl2 (6 equiv). The reaction mixture was allowed to warm to room
temperature and
stirred at room temperature overnight. Then cooled to 0 C, quenched with
saturated NaHCO3
(aq), and extracted with DCM. The organic layer was washed with brine, dried
over Na2SO4 and
concentrated under vacuum. The residual was used by next step without
purification.
Step 3: Preparation of (R)-tert-butyl 1-(3-((2-amino-4-(pentylamino)-5H-
pyrrolo[3,2-d]pyrimidin-
5-yl)methyl)-4-methoxybenzyl)pyrrolidine-2-carboxylate (48)
To a solution of 5-(5-(chloromethyl)-2-methoxybenzy1)-N4-pentyl-5H-pyrrolo[3,2-
d]pyrimidine-2,4-diamine (47, from previous step, 1 equiv) in DCM (0.1M) at
room temperature
was added (R)-tert-butyl pyrrolidine-2-carboxylate (4 equiv). The reaction
mixture was stirred at
room temperature overnight. The reaction mixture was concentrated under
vacuum. The crude
was purified by reverse phase prep-H PLC to afford title compound as a white
solid.
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
68
Step 4: Preparation of (R)-1-(3-((2-amino-4-(Dentylamino)-5H-qyrrolo[3,2-
diqvrimidin-5-
yOmethyl)-4-nriethoxybenzyppyrrolidine-2-carboxylic acid (49)
To a solution of (R)-tert-butyl 1-(3-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-
yl)methyl)-4-methoxybenzyppyrrolidine-2-carboxylate (48, from the previous
step, 1 equiv) in
DCM (0.5M) at room temperature was added TFA (10 equiv). The reaction mixture
was stirred
at room temperature for 6 hours. After the reaction mixture was concentrated
under vacuum,
the crude was purified by reverse phase prep-HPLC to afford title compound 49
as a white
solid. 1H NMR (d-DMS0): 8 12.58 (s, 1H), 7.48 (br, 2H), 7.47 (d, 1H), 7.36 (t,
1H), 7.32 (d, 1H),
7.16 (d, 1H), 6.77 (s, 1H), 6.21 (d, 1H), 5.54 (s, 2H), 4.19-4.13 (m, 2H),
3.84 (s, 3H), 3.46-3.41
(m, 2H), 3.31-3.25 (m, 1H), 3.10-3.04 (m, 1H), 2.41-2.32 (m, 2H), 2.02-1.93
(m, 2H), 1.88-1.75
(m, 1H), 1.50-1.42 (m, 2H), 1.29-1.20 (m, 2H), 1.15-1.07 (m, 2H), 0.81 (t,
3H). LRMS [M+H] =
467.3
Example 25
(S)-1-(442-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yOmethyl)-3-
methoxybenzyhpyrrolidine-2-carboxylic acid
WMe NH
HOOC
N
A
HN N
Preparation of (5)-1-(4-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-
yl)methyl)-3-
methoxybenzyl)pyrrolidine-2-carboxylic acid
(S)-1-(4-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yl)methyl)-3-
methoxybenzyl)pyrrolidine-2-carboxylic acid was prepared using similar
protocols as Examples
13 and 24. Methyl 4-(chloromethyl)-3-methoxybenzoate was used instead of
methyl 3-
(chloromethyl)-4-methoxybenzoate in Example 13, Step 1. (S)-tert-butyl
pyrrolidine-2-
carboxylate was used instead of (R)-tert-butyl pyrrolidine-2-carboxylate in
Example 24, Step 3.
1H NMR (d-DMS0): 8 12.76 (s, 1H), 7.54 (br, 2H), 7.40-7.36 (m, 2H), 7.22 (s,
1H), 6.97 (d, 1H),
6.58 (d, 1H), 6.20 (d, 1H), 5.56 (s, 2H), 4.14-4.37 (m, 2H), 3.86 (s, 3H),
3.47-3.42 (m, 2H),
3.36-3.30 (m, 2H), 3.14-3.08 (m, 1H), 2.39-2.32 (m, 1H), 2.04-1.95 (m, 2H),
1.87-1.80 (m, 1H),
1.49-1.41 (m, 2H), 1.26-1.18 (m, 2H), 1.12-1.05 (m, 2H), 0.81 (t, 3H). LRMS
[M+H] = 467.3.
Example 26
2-(44(2-amino-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-yOmethyl)-3-
methoxyphenoxy)acetic acid
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
69
cati OMe OH TMSCHN2 a ms OMe
HO W HO Ms0
50 51
CI
H
N 0 0
0j=LOH
1) CI OMe
W NH W W NH
Cs2CO3, NH4OH
OMe NN OMe
then
Cu2O
A õ...1
W NH2 CI -N NMP H2N N
53
52
Step1: Preparation of methyl 2-(4-(hydroxymethyl)-3-nriethoxyphenoxy)acetate
(50)
2-(4-(hydroxymethyl)-3-methoxyphenoxy)acetic acid (commercially available, 1
equiv) was
stirred in methanol (0.5 M) at 0 C. Trimethylsilyldiazomethane (1.2 equiv.)
was added in a
dropwised fashion. The reaction mixture was allowed to warm to room
temperature and stirred
at room temperature for 1 hour, cool to 0 C, quenched with water, and
extracted with ethyl
acetate. The organic layer was washed with brine, dried over Na2SO4 and
concentrated under
vacuum. The residual was used by next step without purification.
Step2: Preparation of methyl 2-(3-methoxy-4-
(((methylsulfonyl)oxy)methyl)phenoxy)acetate (51)
Methyl 2-(4-(hydroxymethyl)-3-methoxyphenoxy)acetate (50, 1 equiv) from the
previous
step was stirred in dichloromethane (1 M) at 0 C. Triethylamine (2.4 equiv.)
and
methanesulfonyl chloride (1.2 equiv.) were added in a dropwised fashion. The
reaction mixture
was stirred for another 1 hour at 0 C, before it was quenched with water and
extracted with
ethyl acetate. The organic phases were combined, dried (Na2SO4), and
concentrated. The
crude product was carried on directly to the next step.
Step3: Preparation of methyl 2-(4-((2-chloro-4-(pentylamino)-5H-pyrrolo[3,2-
dipyrimidin-5-
yl)methyl)-3-methoxyphenoxy)acetate (52)
methyl 2-(4-((2-chloro-4-(pentylamino)-5H-pyrrolo[3,2-d]pyrimidin-5-ylynethyl)-
3-
methoxyphenoxy)acetate (52) was prepared following the same protocol as
Example 14, Step
.. 1, using methyl 2-(3-methoxy-4-(((methylsulfonyl)oxy)methyl)phenoxy)acetate
(51, from the
previous step) in place of 4-(bromomethyl)-3-methoxybenzonitrile.
Step4: Preparation of 2-(4-((2-amino-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-yl)methyl)-3-
methoxyphenoxy)acetic acid (53)
To a solution of methyl 2-(4-((2-chloro-4-(pentylamino)-5H-pyrrolo[3,2-
d]pyrimidin-5-
yl)methyl)-3-methoxyphenoxy)acetate (52, 1 equiv., from previous step) in NMP
(1 M) in
microwave tube was added NH4OH (equal volume to NMP) followed by copper(I)
oxide (0.1
equiv.). The tube was capped, and the suspension was heated at 110 C
overnight. After
cooling down to room temperature, the reaction mixture was purified by reverse
phase prep-
CA 02946408 2016-10-19
WO 2015/168269
PCT/US2015/028264
HPLC to afford title compound as a white solid. 1H NMR (CDC13-CD30D): 8 7.22
(s, 1H), 6.73
(d, 1H), 6.65 (d, 1H), 6.45 (d, 1H), 6.16 (d, 1H), 5.34 (s, 2H), 4.55 (s, 2H),
3.88 (s, 3H), 3.46 (t,
2H), 1.50-1.43 (m, 2H), 1.31-1.24 (m, 2H), 1.19-1.13(m, 2H), 0.86 (t, 3H).
LRMS [M+H] =
414.2.
5 Assays
Compounds of Formula (I) were assayed to measure their capacity as toll-like
receptor 7
agonists.
Human peripheral blood mononuclear cell assay
10 The
bioactivity of the compounds of Formula (I) were tested in the human
peripheral blood
assay (human PBMC) using a panel of independent normal human donors according
to
approved guidelines by the institutional review committee. Human PBMC were
isolated from
freshly peripheral blood using a Ficoll density gradient (GE healthcare 17-
1440-03). 30-35mLs
of peripheral human blood were layered onto 15mLs of Ficoll in 50 ml conical
tubes, followed by
15 centrifugation at 1800 rpm (Eppendorf Centrifuge 581OR with biohazard
caps over the tube
buckets) at room temperature for 30 minutes with no acceleration and no brake.
The buffy
layers were then collected and transferred onto new 50 ml conical tubes and
washed twice in
complete media consisting of RPM! 1640 (11875085 from Invitrogen Corporation,
Carlsbad,
California) supplemented with 10% heat inactivated fetal bovine serum (Gibco
10099-141), 1%
20 Pen-Strep (Gibco#15140-122), 1 mM non essential amino acids (Gibco#11140-
050), 1 mM
sodium pyruvate (Gibco#11360-070), 2 mM L-Glutamine (Gibco#25030-081) and 1 mM
HEPES (Gibco#15630-080). Viable cells were then counted using trypan blue
staining, plated
in 96 well flat bottom plates (Becton Dickinson #353070) at 2x105 cells per
well in 200 I total
volume of complete media. Compounds were then added in a 10 point dose
response format
25 starting at 100 p M, 3 fold dilution. Negative controls wells received
equal concentration of
DMSO. Culture supernatants were collected after 18-24 hours incubation at 37
C, 5% CO2,
stored at -20 C until further use.
IL-6 levels in the culture supernatants were measured using a Luminex kit
(Biorad). Data
analysis is performed using Prism software from GraphPad (San Diego, CA). Dose
response
30 curves are generated for each compound and E050 values were determined
as the
concentration that gives 50% of the maximal signal.
Reporter oene assay
Human embryonic kidney 293 (HEK 293) cells were stably transfected with human
TLR7
35 and an NF-kB-driven luciferase reporter vector (pNifty-Luciferase). As a
control assay, normal
Hek293 transfected with pNifty-Luc were used. Cells were cultured in DMEM
supplemented
with 2 mM L-glutamine, 10% heart inactivated FBS, 1% penicillin and
streptomycin, 2 pg/ml
puromycin (InvivoGen #ant-pr-5) and 5pg/mlof blasticidin (Invitrogen #46-
1120). Bright-GbTM
CA 02946408 2016-10-19
WO 2015/168269 PCT/US2015/028264
71
Luciferase assay buffer and substrate were supplied by Promega #E263B and
#E264B (assay
substrate and buffer respectively). 384 well clear-bottom plates were supplied
by Greiner bio-
one (#789163-G) and were custom bar-coded plates.
Cells were plated at 25,000 cells/well in 384-well plates in a final volume of
50 I of media.
Cells were allowed to adhere to the plates after overnight (18 hours) culture
at 372C and 5%
CO2. Serially diluted experimental and positive control compounds were then
dispensed to
each well and incubated for 7 hours at 372C and 5% CO2. Cells stimulated with
DMSO alone
also serve as negative controls. After the incubation, 30 pl of the pre-mix
assay buffer and
substrate buffer were added to each well according to manufacturer's
instructions. The
luminescence signal was read on a CLIPR machine with an integration time of 20
seconds per
plate.
Dose response curves are generated for each compound and EC50 values were
determined
as the concentration that gives 50% of the maximal signal.
Certain Assay Results
Various compounds of Formula (I) in free form or in pharmaceutically
acceptable salt form,
exhibit pharmacological properties, for example, as indicated by the in vitro
tests described in
this application. The EC50 value in those experiments is given as that
concentration of the test
compound in question that provoke a response halfway between the baseline and
maximum
responses. In other examples, compounds of Formula (I) have EC values in the
range from 1
nM to 2 pM. In other examples, compounds of Formula (I) have EC50 values in
the range from
1 nM to 1 pM. In other examples, compounds of Formula (I) have EC50 values in
the range from
1 nM to 500 nM. In other examples, compounds of Formula (I) have EC50 values
in the range
from 1 nM to 250 nM. In other examples, compounds of Formula (I) have EC50
values in the
range from 1 nM to 100 nM. In other examples, compounds of Formula (I) have
EC50 values in
the range from 1 nM to 50 nM. In other examples, compounds of Formula (I) have
EC50 values
in the range from 1 nM to 25 nM. In other examples, compounds of Formula (I)
have EC50
values in the range from 1 nM to 10 nM. Such ECK values are obtained relative
to the activity of
resiquimod set to 100%.
By way of example only, the EC50 for TLR-7 stimulation by certain compounds of
Formula (I)
are listed in Table 1.
Table 1
Human TLR7 Human TLR7
Example Example
Number
EC50 (PM) Number EC50 (PM)
HEK293 HEK293
1 0.1 14 1.4
2 0.05 15 0.06
. 81800293
72
Example Hujan,TLR7 Example Huon,TLR7
Number u (PM) Number WM)
HEK293 HEK293
3 0.2 16 0.2
4 0.05 17 0.5
0.4 18 0.6
6 0.1 19 0.08
7 0.8 20 0.3
8 0.3 21 0.4
9 0.8 22 0.7
0.2 23 0.4
11 0.1 24 0.08
12 0.2 25 0.008
13 0.1 26 0.4
It is understood that the examples and embodiments described herein are for
illustrative
purposes only and that various modifications or changes in light thereof will
be suggested to
5 persons skilled in the art and are to be included within the spirit and
purview of this application
and scope of the appended claims.
CA 2946408 2019-09-27