Note: Descriptions are shown in the official language in which they were submitted.
-2- 2019959
It is well known that mixtures of minerals, and in particular raw
potassium salts, can be broken down into their component fractions by
electrostatic means. To accomplish this, the raw potassium salts are ground
until the intergrowths are destroyed, then the fraction having a grain size of up
5 to 2 mm is separated out of the ground material. This fraction of the raw
potassium salts is then mixed with small amounts of chemical conditioners
which facilitate the transfer of charge between the particles of the mixture
during the subsequent step when the mixture is triboelectrically charged under
defined conditions of relative humidity and temperature in the ambient air. The
10 triboelectrically charged mixture is then fed to electrostatic free fall separators
where it is separated into a product fraction, middlings and residue. This
method of processing potassium salts is described in detail by Singewald et al.
in the journal "Chem.-lng.-Techn." 55 (1983), pp. 30 to 45.
However, it is also a known fact that extremely fine fractions having
15 grain sizes of less than 0.1 mm, which may be present in substantial quantities
in the ground mixtures of potassium salts, exert a highly disruptive influence
on the electrostatic processing of these mixtures. Therefore, German Patents
12 59 803 and 12 79 572 reveal processes by means of which these
disrupting influences can be largely avoided. According to these processes,
20 the ground-up mixture of raw potassium salts can be broken down in a first
electrostatic stage into a "fines" fraction, comprising the extremely fine-sizedfractions, a residue, a pre-concentrated product fraction and possibly a
middlings fraction. In both cases, it si necessary by means of a wet process
to further treat the separated fines fraction before a saleable product can be
25 produced from it. The considerably amounts (up to 20%) of fine-sized materialpresent in ground-up raw potassium salts can cause serious disruptions when
the material is then further processes by wet means.
This gave rise to the need to find ways by which even the fine-sized
fractions of ground mixtures of minerals, and in particular raw potassium salts,30 can be processed electrostatically, i.e. using dry methods.
In general terms, the present invention provides, in one aspect thereof,
a process for processing ground-up mixtures of minerals, especially raw
potassium slats, having substantial amounts of fines, by means of electrostatic
separation in free fall separators following conditioning and triboelectric
r
D
_ ~3~ 2019~59
charging wherein, following conditioning and triboelectric charging, the mineralmixture is separated in a defined separation size range into a coarse fraction
and a fine fraction, and the coarse fraction is then electrostatically processedin free fall separators, while the fine fraction is separately fed to an
electrostatic free fall separator in which the fine fraction is exposed to an
electric filed on which is superimposed an air flow oriented vertically from theinlet to the foot of the separator.
In another aspect, the invention provides apparatus for carrying out the
above method, wherein tubes or annular pipes made of electrically non-
conducting materials are arranged in the discharge ducts of the free fall
separator, said pipes or tubes being provided with a number of nozzle openings
directed towards the foot of the separator and being connected with a device
which produces a negative pressure.
The separation size range in question here is between 0.04 and 0.315
mm. The preferred size range for this process is between 0.063 and 0.2 mm.
The flow rate of the vertical air flow is advantageously less than 3 m/sec
and preferentially less than 1 m/sec.
The invention is described below on the basis of the processing of raw
potassium salt. In order to implement the process according to the invention,
the raw potassium slat is ground to sizes below 2 mm and conditioned using
known chemical conditions; it is then triboelectrically charged by setting the
individual salt particles in vigorous motion, for example in a fluidized bed, in a
suitably warmed carrier air flow having a predetermined relative humidity. The
fine fractions of the salt mixture are carried out of the fluidized bed in the
carrier air flow and then separated from the carrier air, for example in a cyclone
or another suitable fines separating device.
The separated fine fraction containing particle sizes of less than 0.1 mm
is then fed into an electrostatic tube, belt or roll separator, preferably a tube
or belt separator.
Particularly good separating results can be achieved in separators n
which an air flow is generated and maintained from the feed chute to the
bottom end of the separator. The flow rate of this air flow should be less than
3 m/sec, and preferentially less than 1 m/sec.
D
~ -4 2019gS9
The air flow in the direction of fall through the free fall shaft is almost
totally free of swirling and this considerably improves the deflection of the
charged salt particles, particularly the finest low-weight sizes, towards the
oppositely charged electrode.
An air flow of this type can advantageously be generated by means of
tubes made of electrically non-conducting material whose walls contain a large
number of nozzle-like openings pointing towards the foot of the separator and
connected via a control device to a system for generating negative pressure.
These tubes are arranged in the particles discharge ducts of the separator; theyrun parallel to its horizontal axis or lower edge and they extend over almost the
entire length of the separating ducts.
The present invention will be described by way of examples, with
reference to the accompanying drawings, wherein:
Figure 1 is a diagrammatic representation of an apparatus according to
1 5 the present invention;
Figure 2 is a diagrammatic representation of the separator of Figure 1,
showing a detail thereof;
Figure 3 is a diagrammatic representation of a method according to
German Patent 12 59 803; and
Figure 4 is a diagrammatic representation similar to that of Figure 3 but
showing the method of the present invention.
Turning now to Figures 1 and 2, a device is depicted by way of example
in diagrammatic form in Figures 1 and 2.
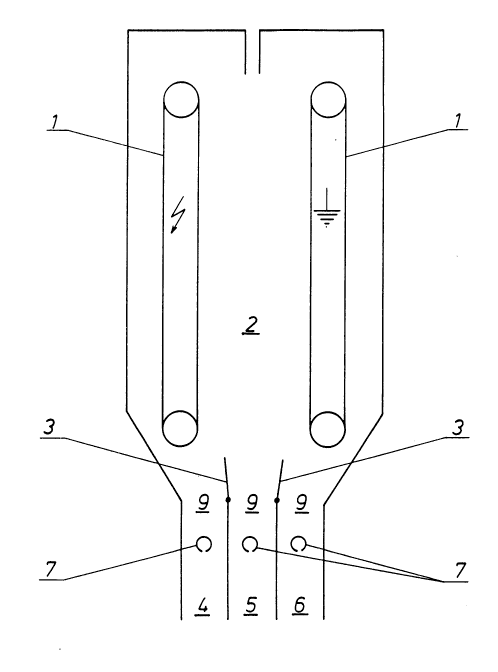
The apparatus is shown as comprising an upright rectangular chamber
or free fall shaft 2. The free fall shaft 2 displays two side walls which are
generally upright and each of which merges with a downwardly and inwards
inclined bottom wall, so that the bottom walls form a hopper-like downwardly
narrowing generally wedge shaped configuration which extends parallel with
obtuse angled corners between the side walls and the bottom walls.
Below the free fall shaft 2, which is bounded by the belt electrodes 1,
one at each side wall of the chamber 2, generally horizontal tubes 7 are
arranged each in one of particle discharge ducts 4 to 6. As shown, the tubes
7 only partly obstruct the passage through the respective discharge duct 4 -
~5~ 2019959
6. The tubes 7 are connected to the device for generating negative pressure,
for instance a vacuum pump.
Fig. 2 depicts, in diagrammatic form, a part section through such a tube
7, showing the openings 8 oriented towards the bottom of the discharge duct.
5 It is, of course, possible to provide a large number of such tubes 7 in a free fall
shaft.
It is also, however, possible to fit in the discharge ducts of the separator
on or more horizontally arranged annular pipes (not shown in the diagrammatic
drawings) made of electrically non-conducting material which would define
10 horizontally elongated annular chamber having in their outer walls downwardlyoriented nozzles or nozzle openings. With these annular pipes it is also possible
to achieve the desired air flow if the annular chambers are connected via
suitable control devices to a device for generating negative pressure.
The air flowing through the nozzle-like openings 8 into the tubes 7 or the
15 annular chamber creates a slight negative pressure in the upper part 9 of theparticle discharge ducts, and in turn this negative pressure creates, in the free
fall shaft 2, a vertically directed downward air flow running from the inlet to
the bottom of the separator.
As already mentioned, when they are separated from the coarse
20 fractions and further processed by wet means, the fines fraction (up to 20%)
contained in the ground-up raw potassium salts can considerably disrupt the
dissolving process and drastically increase the amounts of process liquor used.
These disadvantage are avoided by the process according tot he invention and
are replaced by the advantages of being able to fully process the raw
25 potassium salts by the dry electrostatic separating procedure.
In the following examples, a ground-up raw potassium salt is used as the
mineral mixture.
This raw potassium salt has a K20 content of 18.2%. Apart from very
small amounts of kieserite and anhydrite, the remainder consists of rock salt.
30 The raw potassium salt is conditioned using 150 g/t salicylic acid and then fed
to the fluidized bed device where it is treated and triboelectrically charged byair having 10% relative humidity and a temperature of 60C.
The coarse and fine fractions separated in the fluidized bed device have
the following screen analyses:
D
i
-6- ` 2019959
Coarse Fraction Fines Fraction
Grain Size (mm) Content wt.% Grain Size (mm) Content wt.%
>0.5 29.2 >0.1 11.1
0.2 to 0.5 47.5 0.063 to 0.1 33.3
0.16 to 0.2 8.7 0.04 to 0.063 31.7
0.1 to 0.16 12.1 >0.04 23.9
>0.1 2.5
In each case 10 t of the raw potassium salt pretreated in the manner
described are used in the following examples.
10 ExamPle 1 (comparison, as per German Patent 12 59 803)
This example follows the process flow chart illustrated in Fig.3. The following
separating results are obtained:
Amount K20 Content K20 Yield
(t) (%) (%)
Product pre-concentrate 2.98 44.1 72.2
Fines fraction 0.76 38.7 16.1
Residue 6.26 3.4 11.7
Mixing the product pre-concentratethe fines fraction gives a concentrate
having 43.0 wt.% K20 in a yield of 88.3 wt.% K20. The residue contains a
mean K20 content of 3.4 wt%, namely 2.3 wt.% in the >0.1 mm screen
fraction and 8.2 wt.% in the <0.1 mm screen fraction.
20 Example 2 (comparison, as per diagram in Figure 4, but without air flow)
Following the triboelectric charging process, the fine and coarse fractions are
fed to separate electrostatic free fall separators. The following separating
results are obtained:
D
7 2019959
K20
Quantity Content K2O Yield
Relative to Relative to
Separator Overall Process
(t) (%) (%) (%)
Coarse
Concentrate 2.88 46.7 92.7 73.9
Coarse Residue 5.32 2.0 7.3 5.9
Fine Concentrate0.83 35.5 80.2 16.2
Fine Residue 0.97 7.5 19.8 4.0
Mixing the coarse and fine concentrates gives a concentrate having a
44.2 wt.% K2O content in a yield of 90.1 wt.% K2O. This corresponds to an
increase of 1.8% in the K2O yield compared with the results obtained in
Example 1.
10 ExamPle 3 (according to the invention, separate processing of the fines fraction
by means of an air flow in the free fall shaft, as per the diagram in Fig. 4)
The following separating results are obtained:
K20
QuantityContentK2O Yield
Relative to Relative to
Separator Overall Process
(t) (%) (%) (%)
Coarse
Concentrate 2.88 46.7 92.7 74.0
Coarse Residue 5.32 2.0 7.3 5.9
Fine Concentrate0.86 39.6 92.8 18.7
Fine Residue 0.94 2.8 7.2 1.4
'l
D
-8- 201~S9
Mixing the coarse and fine concentrates give a concentrate having a
45.1 wt.% K20 content in a yield of 92.7 wt.% K20. Compared with the
results according to Examples 1 and 2, this is where the highest values are
obtained. A comparison of the separating results further shows that the fines
5 separating efficiency of the separator is considerably improved by the vertical
air flow.
D