Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
133~06
TITLE OF THE I~VENTION
Medical Instrument
BACKGROUND OF THE INVENTION
Field of the Invention
This invention relates to a blood reservoir or a hollow fibre
type oxygenator in which the blood flow channel section of the
blood reservoir or at least a fraction of the outer wall surfaces of
the hollow fibre of the oxygenator is subjected to a chemical
treatment such as with acid or albumin solutions or
polyhydroxyethyl methacrylate, hereafter referred to as PHEMA,
discharge treatment such as by corona or plasma treatment or with
ozone and thereby hydrophilized to improve wettability thereof
with the blood.
This invention also relates to medical implements or
instruments in which the blood flow channel sections are
hydrophilizingly treated using a polymer such as hydroxyethyl
methacrylate (HEMA), methyl methacrylate (MMA) or using
poly(oxyethylene)-poly(oxypropylene) block polymer after treated
using above polymer.
Prior Art
Various polymers employed in the materials of medical
implements are generally of hydrophobic properties. However,
depending on usage and applications, not few of these materials
require a hydrophilizing surface treatment.
In an oxygenator, for example, the housing is divided by a
gas exchange membrane into two sections and a gas exchange is
effected between the blood flowing in one of these sections and the
gas, mostly oxygen, flowing in the other section. As a gas exchange
membrane, polypropylene, polystylene or silicone membranes,
exhibiting hydrophobic properties, are frequently employed.
It is noted that, before using the oxygenator, a priming
operation is usually carried out to remove the air contained in the
oxygenator. The membrane is hydrophobic and exhibits only poor
affinity with water so that the air cannot be eliminated completely
in the course of the priming operation. Above all, in the case of a
'~' r~
1332906
-2-
hollow fibre type oxygenator employing porous hollow fibre as the
gas exchange membrane, with the blood being caused to flow
outside of the hollow fibre, there is demonstrated a strong
tendency for the air to become trapped between the adjacent fibers
of the hollow fibre. The result is that these adjacent fibers become
flocculated by the air to form so-called cavities to lower the gas
exchange performance.
The majority of the hollow fibre type oxygenators hitherto
evolved were of the type in which the blood is caused to flow
within the inside of the hollow fibre. However, because of large
presseure losses encountered, it is felt that this type of the
oxygenator can be applied to pulsed flow excorporeal circulation,
separate excorporeal circulation or blood cardioplegia, only with
considerable difficulties.
When the blood and the gas are caused to flow outside and
inside of the hollow fibre, respectively, pressure losses may be
lowered, so that the blood can be supplied to the oxygenator and
thence to the blood reservoir by blood movement caused only by
the pressure head from the patient's body without the necessity of
providing a blood delivery pump ahead of the oxygenator in the
circulating circuit. In this manner, the oxygenator can be adapted
to blood cardioplegia or to separate excorporeal circulation.
With the hollow fibre-formed by a hydrophobic resin, the
fibre surface exhibits only low wettability by the blood, so that the
blood cannot be distributed satisfactorily between the adjacent
hollow fibre. Hence an effective gas exchange via the hollow fibre
is obstructed and a sufficient gas exchange performance is not
achieved. For wetting the outer wall surfaces of the hollow fibre,
the air bubbles remaining between the hollow fibre need be
removed by imparting a physical shock, such as by a laborious
operation of striking the oxygenator.
In open heart surgery, a blood circuit with a built-in
oxygenator is used in place of the living lung to eliminate carbon
dioxide in the blood and to replenish oxygen in excorporeal
circulation.
~3~ 133290~
In the excorporeal blood circulation circuit with the built-in
oxygenator, a blood reservoir is provided to eliminate air bubbles
occasionally flowing int the circuit or to store and replenish the
blood in the event of possible decrease in the blood circulation
caused by, for example, tube rupture in the circuit.
In view of relative ease with which the stored blood quantity
can be ascertained and the blood of a large volume can be stored, a
hard shell type blood reservoir, formed of a hard material, is
generally employed. Since the blood reservoir can then be
incorporated easily into the oxygenator, there is proposed an
oxygenator with a built-in blood reservoir.
However, when the blood reservoir exhibits hydrophobic
properties, the blood or the priming liquid is not allowed to flow
uniformly on the overall surface of the blood flow channel, but
flows as a partialized flow into the blood reservoir to produce air
bubbles .
The hard shell type blood reservoir 1 integrated to the
oxgenator is shown diagrammatically in Fig. 1 and formed by a
housing 7 formed of a hard material and including a blood inlet
port 2, a blood influent section 5 communicating with the inlet port
and presenting a bottom surface having substantially no drop from
the inlet port 2, a blood reservoir section 6 communicating with the
blood influent section and presenting a bottom surface gradually
descending from the section 5 and a blood outlet 3 formed at the
bottom of the reservoir section 6. It is noted that the bottom
surfaces of the blood influent section 5 and the blood resevoir
section 6 and the lateral sides of the housing 7 represent a blood
flow channel surface.
The blood introduced via the blood inlet port 2 is caused to
flow on the blood flow channel surface 4 so as to descend to and be
stored in the blood storage section 6.
Heretofore, the housing 7 of the blood reservoir 1 was formed
by a member of a material exhibiting hydrophobic properties, such
as, for example, hard vinyl chloride resin, styrene resin or
carbonate resin. As a result, the the blood flow channel surface 4
. --. i
; .
~4~ 1332~6
also exhibited hydrophobic properties, so that, on performing
bloodless priming, the priming liquid will flow as a partialized flow
without flowing uniformly on the overall blood flow channel
surface 4. In this manner, the liquid flow will be disturbed and the
priming liquid flows into the blood storage section 6 just like a fall
flows down into a pond to produce the air bubbles or foam in the
blood storage section 6. In addition, the above materials are not
said to be satisfactory in compatibility to the blood.
It will be noted that the filter medium for removal of foreign
matter of a blood filter or arterial filter provided downstream of
the excorporeal circulating circuit is in the form of pleats, for
elevating the properties of removal of foreign matter, so that air
bubbles cannot be removed easily. Also, since the materials are
hydrophobic, they are low in wettability, so that a certain pressure
head is necessitated in the priming operation, which renders the
priming operation difficult.
For combatting the above deficiency and improving
wettability of the blood flow channel surface by the blood or the
priming liquid, attempts have been made to hydrolyze the blood
flow channei surface by a suitble surface treatment. However,
these attempts have not met with success because of difficulties in
simultaneously achieving uniform hydrophilic properties and
improved compatibility to the blood without changing the
properties of the material.
It is therefore an object of the present invention to provide
an oxygenator in which the outer wall surface of the hollow fibre is
hydrophilized to improve wettability thereof by the blood and the
priming liquid to suppress formation of air bubbles without
lowering the gas exchange properties.
It is another object of the present invention to provide a
blood reservoir in which the blood flow channel surface is
hydrophilized in part or in its entirety to improve wettability
thereof by the blood and the priming liquid to suppress generation
of air bubbles or foam by the blood flowing into the blood storage
section.
;
-;
13~0~
The present invention has been fulfilled as a result of our
perseverant researches for combatting the above deficiencies
mainly caused by the hydrophobic properties of the blood flow
channel sections of the medical istruments. It is an object of the
present invention to provide a medical instrument in which at least
the blood flow channel section thereof is subjected to a
hydrophilizing treatment to improve wettability thereof by the
blood or by the priming liquid to prevent affixture of air bubbles
while simultaneously improving compatibility to the blood of the
medical instrument.
The present invention has been fulfilled as a result of our
perseverant researches for eliminating the above deficiencies
mainly caused by the hydrophobic properties of the blood flow
channel sections of the medical instrument. It is another object of
the present invention to provide a medical instrument in which at
least the blood flow channel section thereof is subjected to a
hydrophilizing treatment to improve wettability thereof by the
blood or by the priming liquid while simultaneously improving
compatibility to the blood of the medical ~instrument.
DISCI OSURE OF THE INVENIION
According to a first aspect of the present invention, there is
provided a hollow fibre type oxygenator in which the outer sides of
the fibre are used as the blood flow channel, wherein at least some
of said hollow fibre have the outer wall sides thereof
hydrophilizingly treated so that air bubbles can be affixed to the
outer wall sides of the hollow fibre only difficultly.
Preferably, the hollow fibre is a polyolefinic resin and the
hydrophilizingly treated hollow fibre has such properties that the
liquid surface is raised along the outer walls of the hollow fibre
when one of the hollow fibre is introduced into water from a
direction normal to the water surface.
According to a second aspect of the present invention, there
is provided a blood reservoir comprising a blood flow channel
surface inclined at least partially, a blood influent section located
)~
13~290S
upstream of said blood flow channel surface, a blood storage
section located below said blood flow channel surface, and a blood
effluent section located downstream of said blood storage section,
wherein said blood flow channel surface exhibits hydrophilic
properties so that the blood introduced into the reservoir via the
blood influent section and flowing down on the blood flow channel
surface will flow substantially uniformly on the surface in its
entirety without exhibiting the tendency to form a partialized flow
on said surface.
The contact angle of the blood flow channel surface exhibiting
the hydrophilic properties with respect to water is preferably less
than 90 and more preferably not more than 80.
According to a third aspect of the present invention, there is
provided a medical instrument wherein at least the blood flow
channel section thereof is coated in part or in its entirety by a
polymer containing hydroxyethyl methacrylate (HEMA) and methyl
methacrylate (MMA).
Preferably, hydroxyethyl methacrylate (HEMA) and methyl
methacrylate (MMA) exist separately as respective separate
segments. Prefearbly, the weight ratio of the segments containing
hydroxyethyl methacrylate (HEMA) to the segments containing
methyl methacrylate (MMA) is 50: 50 to 95: 5. More perferably,
the contents of hydroxyethyl methacrylate (HEMA) in said segment
containing hydroxyethyl methacrylate (HEMA) are not less than 50
wt. % and the contents of methyl methacrylate (MMA) in said
segment containing methyl methacrylate (MMA) are not less than
70 wt.%. .
Preferably, the medical instrument is an oxygenator and the
blood flow channel section is the outer wall of porous hollow fibre
housed within the oxygenator or a blood reservoir.
According to a fourth aspect of the present invention,there is
provided a medical instrument wherein at least the blood flow
channel section is coated in part or in its entirety by a polymer
containing hydroxyethyl methacrylate (HEMA) and methyl
methacrylate (MMA).
7 133290~
Preferably, hydroxyethyl methacrylate (HEMA) and methyl
methacrylate (MMA) exist separately as respective separate
segments .
Prefearbly, the weight ratio of the segments containing
hydroxyethyl methacrylate (HEMA) to the segments containing
methyl methacrylate (MMA) is 50: 50 to 95: 5. More perferably,
the contents of hydroxyethyl methacrylate (HEMA) in said segment
containing hydroxyethyl methacrylate (HEMA) are not less than 50
wt. % and wherein the contents of methyl methacrylate (MMA) in
said segment containing methyl methacrylate (MMA) are not less
than 70 wt. %.
Additionally, after the medical instrument is treated by the
polymer containing HEMA and MMA, it is treated by a
poly(oxyethylene)-poly(oxypropylene) block polymer having the
general formula
HO(CH2CH20)a-(CH-CH20)b-(CH2CH20)CH
CH3
Preferably, a + c and b in the above formula are 2 to 2000 and 10
.
to 150, respectively.
Preferably, the medical instrument is an oxygenator and the
blood flow channel section is the outer wall of porous hollow fibre
housed within the oxygenator or a blood reservoir.
The medical instruments also include a blood reservoir and a
blood filter.
The medical instruments further include components of a
pump oxygenator such as the aforementioned pump oxygenator,
blood reservoir or blood filter and a pump oxygenating circuit
system formed by these components.
BREF DESCRIPTION OF THE DRAWINGS
Fig. 1 is a longitudinal cross-sectional view showing an
embodiment of a blood reservoir according to the present invention
and a prior art device.
.~' p
1332~0~
Fig.2 is a front view, partially broken away and showing a
pump oxygenator having hollow fibre according to the present
invention.
Fig.3 is a cross-sectional view of a blood filter.
Figs.4 and 5 are diagrammatic views showing a pump
oxygenating circuit system.
9 1~32~0S
DETAILED DESCRIPTION OF THE INVENTION
~ hollow fibre type oxygenator according to a first aspect and
a blood reservoir according to a second aspect of the present
invention will be described in detail with reference to a blood
reservoir of the type to be built into the oxygenator acording to a
preferred embodiment of the present invention.
The hard shell type blood reservoir 1 incorporated to the
oxygenator is shown diagrammatically in Fig. 1 and formed by a
housing 7 formed of a hard material and including a blood inlet
port 2, a blood influent section 5 communicating with the inlet port
and presenting a bottom surface having substantially no drop from
the inlet port 2, a blood reservoir or storage section 6
communicating with the blood influent setion and presenting a
bottom surface gradually descending from the section 5 and a
blood outlet port 3 formed at the bottom of the reservoir section 6.
It is noted that the bottom surfaces of the blood influent section 5
and the blood resevoir section 6 and the lateral sides of the housing
7 represent a blood flow channel surface 4.
The above described blood reservoir is provided within an
excorporeal circulating circuit of the oxygenator. It is however
more preferred that the blood reservoir be used as an oxygenating
unit in which it is combined as one with the oxygenator and a heat
exchanger, as shown for example in Fig.2.
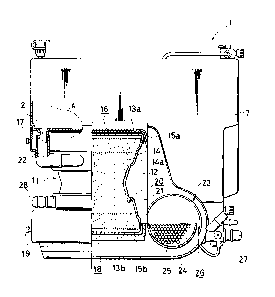
In the preferred embodiment shown in Fig.2, an oxygenator
11 includes a housing formed by a main body of the housing 12
and attachment covers 13a, 13b closing both open ends of the main
body of the housing 12. There are provided within the housing a
multiplicity of hollow fibres 14 parallel to and spaced apart from
one another along the longitudinal direction of the housing.
These hollow fibres 14 are maintained liquid-tightly with
respect to the main body of the housing 12 by partition walls 15a,
15b at the both ends with the opening ends of the fibres remaining
open.
, ~
;
-10- ~L3~0~
A gas inlet space 16 formed by the attachment cover 13a,
main body of the housing 12 and the partition wall 15 so as to be
in communication with the inner space of the hollow fibres
communicates in turn with a gas inlet port 17, and a gas discharge
space 18 formed by the attachment cover 13b, main body of the
housing 12 and the partition wall 15b so as to be in communication
with the inner space of the hollow fibres communicates in turn
with a gas discharge port 19.
A blood chamber 20 defined by the inner wall of the main
body of the housing 12, partition walls 15a, 15b and the outer
walls of the hollow fibres 14 communicates with a blood inlet port
21 and a blood outlet port 22.
The oxygenator 11 shown herein is of the type in which gas
exchange is performed with the oxygen-containing gas such as air
being blown in the inner space of the hollow fibres 14 and with the
blood being caused to flow on the outer sides of the hollow fibres
14.
The hollow fibres 14 may be formed of any suitable
hydrophobic materials customarily used for oxygenators. For
example, polytetrafluoroethylene, polypropylene or silicone is
preferred .
To the blood outlet 22 of the oxygenator 11, there is
connected liquid-tightly the blood inlet port 2 of the blood
reservoir 1 described with reference to Fig.1.
A heat exchanger 23 is connected to the blood outlet 21 of
the oxygenator 11. The heat-exchanger 23 includes a casing 24 in
which a multiplicity of heat..excahnger tubes 25 are arranged
parallel to and at a spacing from one another along the length of
the casing 24. The both ends of the heat exchanger tubes 25 are
held liquid-tightly with respect to the side walls of the casing 24
by partition walls, not shown, with the opening ends of the tubes
remalning open.
A spacing 26 defined by these partition walls, side walls of
the casing 24 and the outer walls of the heat exchanger tubes 25 is
kept in communication with a blood inlet port 27 and the blood
1332~0~
inlet 21 to the oxygenator 11. The inner spacing of the heat
exchanger tubes 25 liquid-tightly separated from the spacing 26 is
kept in communication with a water inlet port 28 communicating
with the outer side of one of the prtition walls of the casing 24 and
a water outlet port, not shown, communicating with the outer side
of the other partition wall of the casding 24.
In the above described heat exchanger 23, the blood flows
into the heat exchanger 23 via blood inlet port 27 to flow along the
outer sides of the heat exchanger tubes 25, while warm or cold
water flows in the inside of the heat exchanger tubes 25 to warm
or chill the blood contacting with the heat exchanger tubes 15.
However, it is also possible to use a heat exchanger of the type in
which the blood flows in the inside of the heat exchanger tubes and
the cooling or heating medium is caused to flow in the outer sides
of the heat exchanger tubes.
It is an important feature of the present invention that part
or all of the blood flow channel surface from the blood inlet 21 to
the blood outlet 22 in the above exemplified oxygenator on which
the blood is likely to contact with oxygenator components,
especially the hollow fibres, is subjected to- a hydrophilizing
treatment. It is not always necessary that the totality of the hollow
fibres making up the hollow fibre bundle be hydrophilized. For
example, more than half the hollow fibres may be hydrophilized
and arrayed uniformly alternately with the remaining
unhydrophilized fibres. In the event that the central portion of the
hollow fibre bundle is constricted and throttled axially by the
housing of the oxygenator, it suffices that at least this constricted
portion be hydrophlized.
According to the present invention, as already described with
reference to the blood resevoir exemplified in Fig.l, all or part of
the blood flow channel surface of the blood reservoir exhibits
hydrophilic properties. It may be the material constituting the
blood flow channel surface itself that exhibits these properties.
There are a number of methods of hydrophilizing the blood
flow channel surface. The following summarizes some of these
.,
-12- 1 3~2~D~
methods that may be employed in connection with the present
nvention.
(1) Acid Treatment
The acids that may be employed include KMnO4/H2SO4 and
K2Cr204/H2SO4 solutions. Above all, the KMnO4/H2SO4 solution is
preferred. For hydrophilizing treatment, the concentrations of 0.05
to 1 wt. % of KMnO4 and 90 to 100 wt. % of H2SO4 are preferred as
the concentrations of the components of the above solution.
In addition to the above acid mixtures, acids such as H2SO4
may be used alone.
(2) Treatment with an Albumin Aqueous Solution
An albumin aqueous solution of 0.5 to 8 w/v % may be
conveniently employed for hydrophilizing treatment.
(3) Treatment with PHEMA
This is the treatment with PHEMA or polyhydroxyethyl
methacrylate. For the intended hydrophilizing treatment, the
concentration of 0.05 to 4 wt. % is preferred.
(4) Corona Discharge Treatment
In the corona discharge treatment, the corona discharge is
caused to occur on a material to introduce hydrophilic groups on
the material surface. The duration of processing is set in
dependence upon the degree of the required hydrophilization.
(5) Plasma Treatment
In plasma processing, active species produced by the glow
discharge are used to treat the polymer surface. The duration of
processing is again set in dependence upon the degree of
hydrophilization required.
(6) Ozone Treatment
In the ozone treatment, Ozone is caused to occur on a material
to introduce hydrophilic groups on the material surface. The
duration of processing is set in dependence upon the degree of
hydrophilization required.
As described above, various methods may be employed for
hydrophilizing the oxygenator. For easiness of understanding, the
-13- 13~2~
degree of hydrophilization may be evaluated in terms of
wettability by water.
A hollow fibre may be said to be endowed with hydrophilic
properties when the water surface rises along the outer wall of the
fibre introduced into water from the direction normal to the water
surface.
When the fibre is not hydrophilized, that is, not subjected to a
hydrophilizing treatment, the water surface becomes concave
towards below along the outer wall of the hollow fibre.
In a material by which the blood reservoir of the present
invention is formed, a contact angle with water of not more than 90
degrees, preferably up to 80 degrees may endow satisfactory
wettability with both priming solution and blood.
Operation
In the oxygenator unit shown in Fig.2, in which the blood
reservoir 1 is integrated with the oxygenator 11 and the heat
exchanger, the blood flowing into heat exchanger 23 via blood inlet
port 27 is heated or cooled until it arrives at the blood inlet port 21
of the oxygenator 11. The blood flowing into the oxygenator 11 via
blood inlet 21 undergoes gas exchange with the oxygen-containing
gas via hollow fibres 14 with the oxygen-containing gas circulated
through the inside of the hollow fibres 14, as the blood flows
through the blood chamber 20, such that carbon dioxide in the
blood is removed while the consumed oxygen is replenished.
It is noted that, since the blood flow channel surface of
oxygenator components, above all, preferably the overall surface of
the outer walls 14a of the blood flow channel surface of the hollow
fibre 14, is hydrophilized by the above described hydrophilizing
treatment, for improving wettability by the blood, there is no risk
that the fibers be flocculated by the air to form cavities, as in the
above described prior art. On the other hand, the blood may flow
smoothly into the blood chamber 20 without stagnation so that the
gas exchange may take place efficiently.
The blood thus replenished with oxygen exits at the blood
outlet 22 of the oxygenator 11 to flow into the blood reservoir 1
`' 'Ç~
-14- 13329~6
via blood inlet port 2 of the blood reservoir 1. The blood thus
introduced via blood inlet port 2 then reaches the blood influent
section 5 communicating with the port 2 to flow smoothly on the
blood flow channel surface 4 to flow down quietly into the blood
reservoir section 6 for storage therein.
Heretofore, the housing 7 of the blood reservoir 1 was formed
of a hydrophobic material, such as hard vinyl chloride resin,
styrene resin or carbonate resin. For this reason, the blood flow
channel surface presents hydrophobic properties. Thus, in the
event of bloodless priming, the priming liquid does not flow on the
overall surface of the blood flow channel surface 4, but flows
unevenly, causing disturbances in the liquid stream. Thus an
inconvenience is caused that the priming liquid flows into the blood
reservoir section 6 as a waterfall drops into a pond so that the
priming liquid forms bubbles in the blood reservoir section.
According to the present invention, the blood flow channel
surface 4 of the blood reservoir 1 described with reference to Fig. 1
is hydrophilized by the above described methods of hydrophilizng
treatment whereby the wettability of the channel surface by the
blood and the priming liquid is improved. The blood and the
priming liquid do not flow unevenly or at one time on the blood
flow channel surface 4, but forms a uniform flow as a whole to
prevent foaming.
The present invention will be explained further in connection
with a medical instrument according to third and fourth
embodinents of the invention.
According to the present invention, the material coating at
least the blood flow channel section of the medical instrument in
part or as a whole by way of a hydrophilizing treatment is
preferably hydroxyethyl methacrylate (abbreviated hereafter to
HEMA ) and methyl methacrylate (abbreviated hereafter to MMA ).
It is because these polymers have a high degree of safety,
adaptability to blood, bioadaptabilty and polymer synthesis and
coating can be made easily.
. ,
-15- 13~2~0~
It is preferred that the polymer containing HEMA and MMA
be in the form of a block copolymer in which HEMA and MMA are
bonded together as separate segments. It is also preferred that the
segments exist separately in the polymer, since then the segments
containing MMA exhibiting high water-proofness can be intimately
bonded to the base material while the segments containing HEMA
exhibiting hydrophilic properties can be disposed on the surface to
provide for a surface hydrophilizing treatment having high degree
of stability.
It is also preferred that the weight ratio of the HEMA
containing segment, referred to hereafter as A segment, to the
MMA containing segment, referred to hereafter as B segment, be
50: 50 to 95: 5. By the A segment and the B segment herein are
meant those fragments or portions mainly containing HEMA and
MMA, respectively. With the weight ratio of the A segment to B
segment less than 50: 50, the hydrophilic properties on the surface
are lowered. With the weight ratio be in excess of 95: S, the
coating polymer is likely to be eluted or peeled during usage.
It is also preferred that the contents of HEMA in the A
segment be not lower than 50 wt. % and that the contents of MMA
in the B segment be not lower than 70 wt. %. With the contents of
HEMA in the segment A less than 50 wt. %, the hydrophilic
properties are lowered. With the contents of MMA in the polymer
less than 70 wt. %, tightness of bonding of the segment to the base
material is lowered, depending on the properties of polymer
components other than MMA.
It is noted that, according to the present invention, there is
no specific limitation to the method of coating the polymer
containing HEMA and MMA on the medical instrument and
practically any method of coating may be employed.
Although a number of methods may be contemplated for
preparing the polymer employed in the present invention, one may
use a method including producing an acrylic polymer having
peroxy bonds in the main chain (HEMA containing polymer) and
effecting dispersion polymerization, using this polymer as the
i .
-16- 13~29~ ~
polymerization initiator, to produce a block copolymer with a MMA
containing polymer.
According to a third aspect of the present invention, the
medical instrument is preferably a porous hollow fibre housed
within an oxygenator or a blood reservoir. In any case, at least the
blood flow channel section thereof is coated in part or as a whole
by the above described polymer to improve wettability by blood
and adaptbility or compatibility to blood
Thus, in the blood reservoir, the polymer is coated mainly on
the blood flow channel surface of the housing to render the flow of
the priming liquid and the blood to the blood reservoir section
more smooth. In the porous hollow fibre, one of the inner and
outer walls of the hollow fibre is used in general as the blood flow
channel surface. However, the present invention is applied to the
hollow fibre, the outer wall of which, above all, is used as the blood
flow channel surface. Thus the outer wall of the hollow fibre is
coated with the above polymer to improve wettability by blood
and to render the blood stream more smooth.
In this manner, adaptability to the blood of the hollow fibre
may be improved simultaneously.
It is to be noted that portions other than the portions forming
the blood flow channel surface may also be subjected to the above
described hydrophilizing treatment.
According to a fourth aspect of the present invention, for
improving wettability, after the blood flow channel section of the
medical instrument is processed and coated with the above
polymer containing HEMA and MMA, it is processed and treated
with a poly(oxyethylene)-poly(oxypropylene) block polymer
represented by the following general formula (I)
HO(CH2CH20)a-( 1CH-CH20)b-(CH2CH20)cH
CH3
It is noted that there is no specific limitation to means of coating
the medical instrument with the block copolymer and any known
methods for coating may be employed.
13~2~5~
- 1 7 -
In this manner, wettability and the properties of preventing
foam deposition or affixture may be improved simultaneously. For
obtaining the above results for the medical instrument of the
present invention, a+c in the above is 2 to 2000, preferably 2 to
500 and more preferably 3 to 300, and b is 10 to 150, preferably
10 to 100 and more preferably 15 to 70. Outside of these ranges,
hydrophilic properties of the block copolymer itself are lowered or
the affinity to the hydrophobic section is lowered, so that
wettability on the surface of the material following the processing
is correspondingly lowered.
The medical instruments according to a fourth aspect of the
present invention include those instruments and components
thereof likely to contact with blood, such as circuit for
cardiopulmonary by-pass, artificial dialyzating system, blood
plasma separating system and a variety of catheters. In the case of
the circuit for cardiopulmonary by-pass, the medical instruments
mean both the respective components making up the system and
the system in its entirety. The components making up the system
include an oxygenator, above all, a film or membrane type
oxygenator, a blood reservoir, such as arterial reservoir, vena
reservoir or cardiotomic reservoir, bubble trap, centrifugal pump
and tubes connecting these components. In any of the above case,
at least the blood flow channel sections of these components are
coated in part or as a whole by the above polymer to improve
wettability by blood, adaptability or compatibility to blood and the
properties of preventing affixture of air bubbles.
That is to say, in the blood reservoir, the above polymer is
coated mainly on the blood flow channel surface of the housing to
render the flow of the blood and the priming liquid to the blood
reservoir section more smooth. In the case of the porous hollow
fibre within the oxygenator, one of the inner and outer walls of the
hollow fibre is used as the blood flow channel surface. Especially,
in the case of the hollow fibre the outer wall side of which is used
as the blood flow channel surface, the outer wall of the hollow fibre
,~
'.' '~
1 8 1 3 3 2 ~ ~ ~
can be coated with the above polymer to improve wettability by
blood to render the blood flow more smooth.
In this manner, the adaptability to blood of the hollow fibre
is also improved simultaneously. In the case of the hollow fibre
the inner wall of which is used as the blood flow channel surface,
the inner wall of the fibre may also be coated with the above
polymer to improve the properties of preventing formation of air
bubbles and adaptability to blood.
It is to be noted that the portions of the hollow fibre other
than those forming the blood flow channel surface may also be
subjected to the above described hydrophilizing treatment.
The medical instrument according to the present invention
includes a blood flow filter 30 shown in cross-section in Fig. 3. This
filter is incorporated into, for example, a pump oxygenating circuit
to effect ultimate defoaming at the time the blood is returned into
the patient's body.
The blood flow filter 30 has a housing 31 having an upper air
discharge port 32, a central blood inlet port 33 and a lower blood
outlet port 34.
A filter medium 35 formed by a polyester net is provided
between the blood inlet port 33 and the blood outlet port 34.
The blood introduced into the blood inlet port 32 flows down
the wall of the housing 31 as it gyrates to reach the filter medium
35. During this time interval, the blood defoamed by the filter
material 35 is returned to the patient's body via outlet port 34,
while tha air removed from the blood is discharged via air outlet
port 34. .
Such blood filter is also preferably processed by a polymer
containing HEMA and MMA or in addition thereto with the block
copolymer represented by the above general formula (I), similarly
to the blood reservoir or the pump oxygenator described above. In
this manner, the defoaming properties are improved, while the
priming time is reduced as a result of improved wettability. In a
hard shell type blood reservoir, one or more components 36 in the
form of a sponge and/or a net may be provided halfway in the
1332~
blood flow channel for improving the defoaming efficiency and
removal of impurities. In this case, only the aforementioned
components or the blood reservoir including these conmponents in
its entirety may be processed by the above polymer to defoam the
trapped air bubblres quickly.
Fig.4 shows an example of a pump oxygenating circuit system
40 including the aforementioned blood reservoir, pump oxygenator
and blood filter. In this system ,the blood reservoir 42, pump 43,
oxygenator 45 with a heat exchanger 44 and a blood filter 30 are
interconnected with a human body 41 by a tube 46 to form an
excorporeal circulating circuit.
Such circuit as a whole is preferably processed with the
aforementioned polymers containing HEMA and MMA or with the
block polymer represented by the general formula (I) to provide
for improved wettability and the properties of preventing affixture
of air bubbles.
OPERATION
The operation of an oxygenator including a blood reservoir
and porous hollow fibres as the medical instrument of the present
invention will be explained in more detail.
It is a feature of the present invention that the blood flow
channel surface 4 of the blood reservoir 1 of Fig.l is coated in part
or as a whole by the polymer containing HEMA and MMA or in
addition thereto by the block polymer represented by the above
general formula (I) to prevent wettability by blood and the
properties of preventing affixture of air bubles.
With the blood flow channel surface 4 being thus coated with
the polymer containing HEMA and MMA or further with the block
copolymer represented by the above general formula (I) by way of
a hydrophilizng treatment, the blood and the priming liquid
introduced at the blood inlet port 2 is caused to flow smoothly on
the blood flow channel surface 4 without becoming stagnant so that
it flows smoothly on the gradually descending surface until it
reaches the blood reservoir section 6. In this manner, the blood
.
,
-20- 13329~6
and the priming liquid are caused to flow quietly within the blood
reservoir section 6 without any disturbances in the liquid flow or
resultant foaming as in the prior art system.
The above described blood reservoir is provided within the
excorporeal circulating blood circuit. However, it is preferably
combined with an oxygenator and a heat exchanger to form an
oxygenating device or system, as shown for example in Fig.2.
-21- 1~32~0~
In the embodiment shown in Fig.2, the oxygenator 11 has a
housing formed by a cylindrical main body of the housing 12 and
attachment covers 13a, 13b closing both open ends of the main
body of the housing 12. Within the entire region of the housing, a
bundle of a multiplicity of hollow fibres 14 is arranged along the
length of the housing so that the fibres are juxtaposed to and
spaced apart from one another.
The both ends of the hollow fibres 14 are supported liquid-
tightly against the main body of the housing 12 by partition walls
15a, 15b, with the opening ends of the fibres being not closed. A
gas inflow space 16 formed by the attachment cover 13a, main
body of the housing 12 and the partition wall 15 so as to be in
communication with the inner space of the hollow fibres
communicates in turn with a gas inflow port 17, and a gas outflow
space 18 formed by the attachment cover 13b, main body of the
housing 12 and the partition wall lSb so as to be in communication
with the inner space of the hollow fibres communicates in turn
with a gas outflow port 19b.
A blood chamber 20 defined by the inner wall of the main
body of the housing 12, partition walls 15a, 15b and the outer
walls of the hollow fibres 14 communicates with a blood inlet port
21 and a blood outlet port 22.
The oxygenator 11 shown herein is of the type in which gas
exchange is performed with the oxygen-containing gas such as air
being blown in the inner space of the hollow fibres 14 and with the
blood being caused to flow on the outer sides of the hollow fibres
14.
The hollow fibres 14 may be formed of any suitable
hydrophobic materials customarily used for oxygenators. For
example, polytetrafluoroethylene, polypropylene or silicone is
preferred .
According to the present invention, the outer wall 14a
forming the blood flow channel surface of the hollow fibres 14 is
preferably coated as a whole by the polymer containing HEMA and
MMA or further with the block copolymer represented by the
-22- 133~9~
above general formula (I) and thereby hydrophilized to improve
wettability thereof by blood.
The hollow fibre type oxygenators developed heretofore were
generaly of the type in which the blood is caused to flow within the
interior of the hollow fibre. However, because of larger pressure
losses encountered, the hollow fibre type-oxygenators can be
adapted only difficultly to pulsapile perfusion, separate excorporeal
circulation or to blood cardioplegia.
The hollow fibre formed by a hydrophobic resin exhibits poor
surface wettability by blood so that the blood is not permeated to a
space between the adjacent hollow fibres and an efficient gas
exchange via the hollow fibres is obstructed with the result that
sufficient gas exchange properties are not obtained. For wetting
the outer wall surface of the hollow fibre, air bubbles remaining
within the space between the hollow fibres need be removed by a
laborious operation of imparting a physical shock to the
oxygenator, such as by striking.
Thus, with the blood being caused to flow outside of the
hollow fibre and the gas within the inner space of the fibres,
pressure losses may be reduced, such that the blood can be
supplied to the oxygenator and thence to the blood reservoir by the
blood being taken out by the pressure head from the patient's body
without the necessity of providing a blood delivery pump ahead of
the oxygenator in the circulating circuit. Thus the oxygenator can
be adapted to blood cardioplegia or separate excorporeal
circulation .
To the blood outlet 22- of the oxygenatorll is liquid-tightly
connected the blood inlet port 2 of the blood reservoir 1 described
with reference to Fig. 1.
A heat exchanger 23 is connected to the blood outlet of the
oxygenator 11. The heat exchanger 23 includes a casing 24 in
which a multiplicity of heat excahnger tubes 25 are arranged
parallel to and at a spacing from one another along the length of
the casing 24 The both ends of the heat exchanger tubes 25 are
held liquid-tightly with respect to the side walls of the casing 24
- - 133~906
by partition walls, not shown, with the opening ends of the tubes
remalning open.
A spacing 26 defined by these partition walls, side walls of
the casing 24 and the outer walls of the heat exchanger tubes 25 is
kept in communication with a blood inlet port 27 and the blood
inlet 21 to the oxygenator 11. The inner spacing of the heat
exchanger tubes 25 liquid-tightly separated from the spacing 26 is
kept in communication with a water inlet port 28 communicating
with one of the prtition walls of the casing 24 and a water outlet
port, not shown, communicating with the outer sides of the other
partition wall of the casing 24.
In the above described heat exchanger 23, the blood flows
into the heat exchanger 23 via blood inlet port 27 to flow along the
outer sides of the heat exchanger tubes 25, while warm or cold
water flows in the inside of the heat exchanger tubes 25 from the
water inlet port 28 to warm or chill the blood contacting with the
heat exchanger tubes 15. However, it is also possiblé to use a heat
exchanger of the type in which the blood flows in the inside of the
heat exchanger tubes and the cooling or heating medium is caused
to flow in the outer sides of the heat exchanger tubes.
In the oxygenating unit in which the blood reservoir 1 is
integrated to the oxygenator 11 and the heat exchanger 23, the
blood flowing into the inside of the heat exchanger 23 via the blood
inlet port 27 is heated or chilled until it reaches the blood inlet 21
of the oxygenator 11. The blood flowing from the blood inlet 21 of
the oxygenator 11 undergoes a gas exchange with the oxygen
containing gas flowing in the-inside space of the hollow fibre 14, as
the blood flows through the blood chamber 20, so that excess
carbon dioxide in the blood is removed, while oxygen is
replenished to supplement consumed oxygen.
Preferably the overall surface of the outer wall 14a of the
hollow fibres 14 acting as the blood flow channel surface is covered
by the HEMA ot MMA containing polymer or further thereon the
block polymer represented by the above general formula (I) and
-24- 1332~
thereby hydrophilized to improve the wettability by the blood and
the properties of preventing affixture of air bubbles, so that there
is no risk for the fibers to be flocculated by the air to form so-
called cavities as in the above described prior art system. In
addition, the blood may flow smoothly into the blood chamber 20
without residing there so that the gas exchange may be performed
efficiently.
The blood thus replenished with oxygen flows out at a blood
outlet 22 of the oxygenator 11 to flow then into the blood reservoir
1 via the blood inlet 2 of the blood reservoir 1 communicating with
the outlet 22. The blood introduced via the blood inlet 2 then
reaches the blood influent section 25 contiguous to the blood inlet 2
to pass by the defoaming member 36 to flow smoothly on the blood
flow channel surface 4 coated by the polymer containing HEMA and
MMA or in addition thereto the block polymer of the formula (I) to
flow down quietly and stored in the blood reservoir section 6
without forming air bubbles in the blood reservoir section 6. These
effects in the blood reservoir 1 are most outstanding at the time of
bloodless priming.
The blood thus stored in the blood reservoir section 6 without
forming air bubbles is led out at the blood outlet 3 at the lower
portion of the blood reservoir section 6 for blood delivery.
And due to improvement of adaptability to blood, it is
expected that decrease of platelets is restrained.
It is noted that the description of the above embodiment has
been made with reference to hollow fibres in an oxygenator.
However, the present inventivn is not limited thereto but may also
be applied to hollow fibres employed for example in
hemoconcentrators .
In the blood filter shown in Fig.3, the properties of
preventing affixture of air bubbles and adaptability to blood are
improved, as in the preceding embodiment. In addition, when the
blood filter is processed with the block copolymer represented by
the general formula (I), among the aforementioned polymers,
e~
-25- 133290~
wettability of the net-like filter material is improved to facilitate
the priming operation.
The same applies for the pump oxygenating circuit system
shown in Figs. 4 and 5.
EXAMPLES
The description with reference to certain Examples will be
given hereinbelow for illustration of the present invention.
Examples for First Aspect of the Inventoin
Comparative Example
Using 34000 microporous polypropylene hollow fibres, each
300 microns in outside diameter, an oxygenator having a film or
membrane area of 3.1 m2 was prepared.
Example 1
A 0.4 % solution of KMnO4/conc. H2SO4 was charged into an
oxygenator prepared similarly to the Comparative Example. While
the narrow portion of the oxygenator was struck, the oxygenator
was allowed to stand for five minutes, and the liquid inside the
oxygenator was discharged. The oxygenator was washed with
water and dried in air.
Example 2
2 liters of a 4 % albumin solution was circulated in the
oxygenator prepared similarly to Comparative Example at a rate of
2 liters per minute as the constricted portion of the oxygenator was
struck. The oxygenator was then dried in air.
Example 3
A methanol solution containing 1 w/v % of PHEMA was
charged into an oxygenator prepared in the same manner as in
Comparative Example. While the constricted portion of the
oxygenator was struck, the oxygenator was allowed to stand for
one minute. After the liquid inside the oxygenator was discharged,
the oxygenator was dried in air.
Example 4
Using a corona discharge unit HFS 202 produced by Kasuga
Denki KK, the same hollow fibres of microporous polypropylene as
1332~
- 2 6 -
those employed in Comparative Example, were taken up on a
bobbin at a take-up speed of 50 m/min, while the current of 6.5 A
at 120 V was caused to flow in an electrode 10 mm wide and 20
mm long, wound by a Teflon sheet 80 microns thick. Then, using
these hollow fibres, an oxygenator similar to that used in the
Comparative Example was assembled.
Example S
The hollow fibres same as those used in the Comparative
Example were cut to lengths of 50 cm and arrayed within a tank so
as to avoid stacking. The fibres were then subjected to plasma
processing at 100 W for two minutes at 1o-l torr and 30 to 40C.
Using the thus produced plasma processed hollow fibres, an
oxygenator similar to that used in Comparative Example was
prep ared .
Example 6
After a gas port of an oxygenator prepared in the similar
manner as in Comparative Example, ozone was introduced for ten
minutes via a blood inlet port. Then, after ozone was similarly
introduced via blood outlet port for ten minutes, the outer wall of
the hollow fibre was subjected to a hydrophilizing treatment.
Test Example 1
The ability of oxygen addition of the oxygenators prepared in
Comparative Example and Examples 1 to 6 was measured in
accordance with AAMI standards. The results are shown in Table
1. With regard to the properties of the oxygenators shown in Table
1, the indications given below have the following meanings:
Before striking: The oxygen addition ability of the blood was
measured immediately after priming of the oxygenator with the
cow's blood.
After striking: The oxygen addition ability of the blood was
measured after the constricted mid portion of the oxygenator was
struck strongly with forceps as the blood was circulated after
termination of the above priming.
, ~--
-27- 13~06
Table 1
Ability of Oxygen Addition
Before Striking (/O) After Striking (%)
Comparative Example 13 100
Example 1 96 100
Example 2 100 100
Example 3 95 100
Example 4 90 100
Example 5 93 100
Example 6 96 100
Test Example 2
Tests were conducted for measuring hydrophilic properties of
the hollow fibres employed in oxygenators produced in
Comparative Example and Examples 1 to 6. A beaker was filled
with water and each one of the above hollow fibres was inserted
into water from above orthogonally to the water surface. As a
result, a meniscus was formed at the boundary between the outer
wall of the hollow fibre and the liquid surface. In the case of fibres
of the Comparative Example,-the liquid surface was seen to be
concave towards below along the outer wall of the hollow fibre,
whereas, in the fibres of the Examples 1 to 6, the liquid surface was
seen to rise along the outer wall of the fibre, thus indicating that
hydrophilic properties were endowed to these fibres.
, ~
-28- 133290~
Examples For Second Aspect of the Invention
Comparative Example
A blood reservoir as shown in Fig. 1 was prepared by injection
molding of a polycarbonate resin.
Example 1
After the blood flow channel surface of a blood reservoir
prepared similarly to the Comparative Example was dipped for five
minutes in a 0.4% KMnO4/conc. H2SO4, the liquid inside the
reservoir was discharged and the reservoir was then washed with
water and dried in air.
Example 2
A blood reservoir prepared in the similar manner to
Comparative Example was filled with a 4 % albumin solution and
allowed to stand stationarily for one minute. The liquid inside the
blood reservoir was discharged and the blood reservoir was dried
in air in a clean bench.
Example 3
A blood reservoir prepared in the similar manner to
Comparative Example was filled with a methanol solution
containing 0.2 w/v % of PHEMA and allowed to stand stationarily.
The liquid inside the blood reservoir was discharged and the blood
reservoir was dried in air in a clean bench.
Example 4
Using a corona discharge unit HFS 202, produced by Kasuga
Denki KK, the blood flow channel surface of a blood reservoir
prepared in Comparative Example was subjected to a corona
discharge treatment with the current of 5 A at 120 V being caused
to flow in an electrode which is 10 mm wide and 20 mm long and
which is wound by a Teflon sheet 80 microns thick.
Example 5
A blood reservoir produced in Comparative Example was
subjected to plasma processing in a tank at 100 W for two minutes
at a pressure of 10-1 torr and at a temperature of 30 to 40 C.
Example 6
-29- 133~ 0~
A blood reservoir produced in Comparative Example was
placed in a glass desiccator. After the temperature within the
desiccator was set to 50 C, 2 was caused to flow into an ozone
generator at a flow rate of 0.8 liter per minute to produce ozone at
100 V, with the so-produced ozone being caused to flow into the
desiccator. After this processing was carried out for 20 minutes,
the inside space of the desiccator was replaced by 2 The blood
reservoir was then taken out of the desiccator to terminate the
ozone processlng.
Test Example 1
The blood flow channel surfaces of the blood reservoirs
produced in accordance with the Comparative Example and
Examples 1 to 6 were partially cut off to measure the contact angle
with respect to water. The results are as shown in Table 2.
Table 2
Contact Angle (degrees)
~omparative Example 92
Example 1 40
Example 2 33
Example 3 39
Example 4 49
Example 5 65
Example 6 62
Test Example 2
With 300 ml of physiological saline water being stored in
blood reservoir sections of the blood reservoirs produced in
Comparative Example and Examples 1 to 6, physiological saline
water was caused to flow on the blood flow channel section at a
- r~
13~29Q~
flow rate of 4 liters per minute and the manner in which the
physiological saline water flows on the blood channel surface and
the generation of air bubbles in the blood reservoir sections were
observed. The results are shown in Table 3.
Table 3
Blood Flow State Foaming State at
Blood Storage Section
Comparative Example Two to three steak-like marked foaming
blood flows were observed
Example 1 Blood flowed on the overall flow no foaming
channel since it started to flow
Example 2 same as above same as above
Example 3 same as above same as above
Example 4 same as above same as above
Example 5 same as above same as above
Example 6 same as above same as above
,~
1332~0~
EMBODIMENTS FOR THlRD AND FOURTH ASPECTS OF THE
INVENTION
Example 1 and Comparative Examples 1 and 2
By way of Example 1, a 2 % (methanol/methyl cellosolve =
92/8) block copolymer of MMA ( B segment )/HEMA ( A segment )
was coated in the state of a solution on a microporous
polypropylene flat film. Also, by way of Comparative Example 1,
the above microporous polypropylene flat film was not subjected to
any treatment such as hydrophilizing treatment. Finally, by way of
Comparative Example 2, a 2 % PHEMA solution in methanol was
coated on a flat microporous polypropylene film to hydrophilize its
surface. Using these films, the following tests on the properties
were conducted.
i) Contact Angle
The contact angle of the surface of the polymer of Example 1
with respect to water was mesured and found to be equal to 62.
After the polymer of Example 1 was allowed to stand in the
polymer-coated state at room temperature for five days, the
contact angle was again measured, and found to be substantially
not changed.
The contact angle of the flat film of Comparative Example 1
with respect to water was about 109, while that of the flat film of
Comparative Example 2 was 64. It has thus been shown that the
above coating by the polymer results in the hydrophilized film.
ii) Tests on Ability to Expand Platelet
By way of tests on platelet spread ability test, the human
blood was treated by a 1/10 vol. part of a 3.8 % sodium citrate
solution against coagulation and centrifuged for 15 minutes at 800
r.p.m. The supernatant was sampled and adjusted to a
concentration of 60,000 per microliter by a diluent (physiological
saline water/3.8% sodium citrate = 9/1). The supernatant thus
treated was dropped on film samples and allowed to stand for 30
minutes, after which the form and the number of the affixed
platelets were checked.
",-~
-32- 133~aS
In Example 1, the rates of affixture were 85%, 15% and 0% for
types I,II and III, respectively, with the sum of the affixed
platelets being 467/0.5 mm2. In Comparative Example 1, the rates
were 49%, 23% and 28% for the types I, II and III, respetively,
with the sum of the affixted platelets being 1386/0.5 mm2 and, in
Comparative Example 2, the same rates were 30%, 25% and 45% for
types I, II and III, respectively, with the sum of the affixd platelets
being 1771 /0.5 mm2.
It may be seen from this that coating the polymer of the
Example in the above manner results not only in improved
hydrophilic properties, but in improved blood adaptability.
The above classification into the types I, II and III has been
made in accordance with the classification found in "Reaction of
Platelets on the Surface of High Polymer Material for Medical Use"
in "The Japanese Journal of Artificial Organs" 9(1), 228 to 231
(1980).
iii) Tests on Eluates
Tests on eluates were conducted in accordance with the
disposable set standard for cardiopulmonary by-pass by the
Ministry of Health and Welfare. Thus the circuit through which
flowed the blood in the cardiopulmonary by-pass was filled with
water which had been boiled and allowed to cool. Then, using a
suitable tool, the ends of degasifying tubes, oxygen blowing tubes
and connecting tubes were closed. The circuit was then heated at
70 + 1C for 30 minutes and cooled and the liquid contents were
taken out as the test liquid and subjected to tests to be described
later. The present oxygenator passed these tests.
The amount of consumtion of potassium permanganate was
within prescribed limits in Example 1, but exceeded the limits in
Comparative Example 2.
Example 2 and Comparative Example 3
By way of Example 2, a polycarbonate blood reservoir as
shown in Fig. l was prepared by injection molding and the blood
flow channel surface thereof was coated with the block polymer of
Example 1 by the same method as that of Example 1. The contact
,~
-33- 1332906
angle of the blood flow channel surface with respect to water was
measured and found to be 38.
On the other hand, a blood reservoir not coated with the
polymer of Example 2 was similarly prepared by way of
Comparative Example 3. The contact angle of the blood flow
channel surface with respect to water was measured and found to
be 91.
The blood storage sections of the blood reservoirs of the
Example 2 and the Comparative Example 3 were charged with 300
ml of physiological saline water and physiological saline water was
caused to flow on the blood flow channel surfaces of the blood
reservoirs to check for the manner in which the physiological saline
water flowed on the blood flow channel surfaces and the manner in
which the foam was generated in the blood storage sections.
It was now found that, in the Example 2, the physiological
saline water flowed uniformly and quietly on the blood flow
channel surfaces in their entirety and no foam was seen to be
generated in the blood storage sections. However, in Comparative
Example 3, two main thick flows were seen approximately at the
center of the blood flow channel surface with three fine
meandering flows about the cnetral flows, and air bubbles or foams
were seen to be floating to and fro in the blood storage section.
Example 3 and Comparative Example 4
By way of Example 3, an oxygenator was prepared with the
use of 34000 microporous polypropylene hollow fibres of 300 ,um
in outside diameter, and a 2 % (methanol/methylcellosolve = 92/8)
block copolymer solution of MMA (B segment)/HEMA (A segment)
was charged in the oxygenator, which was allowed to stand for one
minute while the constricted portion of the oxygenator was struck,
the liquid contents in the oxygenator being then discharged and
dried in air.
On the other hand, by way of Comparative Example 4, the
same oxygenator was charged with a methanol solution containing
2 w/v % of PHEMA and allowed to stand for one minute while the
~,
-34- 1332~0~
constricted portion of the oxygenator was struck, the liquid
contents in the oxygenator being then discharged and dried in air.
The oxygenators of Example 3 and Comparative Example 4
were then subjected to tests on eluates in accordance with the
disposable set standard for cardiopulmonary by-passes by the
Ministry of Health and Welfare. Thus the circuit through which
flowed the blood of the cardiopulmonary by-pass was filled with
water which had been boiled and allowed to cool. Then, using a
suitable tool, the ends of degasifying tubes, oxygen blowing tubes
and connecting tubes were closed. The circuit was then heated at
70 + 1 C for 30 minutes and cooled and the liquid contents were
taken out as the test liquid and subjected to the following tests
which the oxygenators under test must pass.
These tests include those on i) outward appearance and pH;
ii) foaming; iii) degree of cleanliness; iv) lead and cadmiun; v) zinc;
vi) a potassium permanganate reducing substance; and vii)
evaporation residues.
In Example 3, the tested oxygenators were found to be within
the reference standards for all of the tests. However, in
Comparative Example4, in the test for the potassium permanganate
reducing substance, the consumption of potassium permanganate
exceeded the reference standards.
Example 4
Using the blood filter shown in Fig.3, the following tests were
conducted. The housing of the blood filter was formed of
polycarbonate, the filter medium was a polypropylene net having a
mesh size of 380 meshes and the blood filtration area was
750 cm2.
The blood filter was charged with a 1 % methanol solution of
MMA (B segemnt)/HEMA (A segment)-20/80 and dried in air
immediately after the liquid contents were discharged.
The blood filter was then charged with an aqueous solution of
a block polymer having the above formula (I) wherein (a+c) =75
and b = 30 (Pluronic F-68 manufactured by BASF) and dried in air
immediately after the liquid contents were discharged.
133~906
A priming liquid (physiological saline solution) was charged
into the inside of the filter medium in the so-produced blood filter
from the blood outlet. At this time, the priming liquid was
accumulated in the inside of the filter msdium until the filter
medium was wetted. When the filter medium was wetted in this
manner, the priming liquid stored in the inside of the filter
medium was spurted out via the filter medium vigorously. The
time elapsed since the charging of the priming lliquid until ejection
of the priming liquid out of the filter medium was measured. With
the filter medium exhibiting poor wettability, the priming liquid
was not discharged until a certain pressure drop was established
with the progress of charging of the priming liquid.
The measured results are shown in the following Table 4.
Table 4
Processing Method remarks Efflux Time Duration (seconds)
1 10 20
none CX-AF *******$*****************
polymer containing HEMA and MMA 1.0 % *************
poly(oxyethylene)-poly(oxypropylene) 0.005 % **~***
block polymer
polymer containing HEMA and MMA (1 %) 0.005 % *****
+poly(oxyethylene)-poly(oxypropylene)
block polymer
133233~
- 3 6 -
E~CTS OF THE INVENTION
The oxygenator exhibits superior wettability by blood since
the hollow fibres are hydrophilized by, for example, acid-,
albumin-, PHEMA-, corona discharge- plasma- or ozone processing.
The result is that, since the air bubbles can be affixed only
difficultly to the outer wall surfaces of the hollow fibres, the space
between the fibers are not blocked by air bubbles and the blood
may flow smoothly so that the gas exchange may be performed
efficiently.
The blood reservoir of the present invention exhibits superior
wettability by the priming liquid and the blood since the blood
flow channel surface, above all, exhibits hydrophilic properties.
The result is that the blood flows down uniformly down the
blood flow channel surface without flowing partially, so that it
becomes possible to prevent air bubbles from being formed when
the blood flows from the blood flow channel surface into the blood
storage section.
The present invention provides a safe medical instrument
which is hydrophilizingly treated by that at least the blood flow
channel section of the instrument is coated by a polymer containing
HEMA and MMA or in addition thereto a block polymer
represented by the general formula (I) and thereby hydrophilized,
and which also is superior in adaptability, to blood, so that the
present invention may be applied extensively to hollow fibres or to
blood resevoirs.
. ~