Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
r~ 3~ 27
, . . .
.., ~`
APPARA~US AND M13THOD FOR RECOVERY OF 8PENT GLYCOI,
Introduction
This application relates to apparatus and a method for
recovering spent glycol for subsequent re-use. More
particularly, this application relates to a method for recovering
glycol based de-icing fluid and other glycol containing
induskrial fluids by removal of contaminants and waterl enabling
subsequent use of the fluid, as well as for purification of the
removed water, rendering the removed water safe for discharge
into the environment.
Back~round Of Thè Invention
The accumulation of ic2 on the wings of aircraft is a
recognized hazard. Ice distorts the shape of the airfoil, thus
reducing the lift needed for controlled ~light. Ice also
increases the weight of the aircraft, thereby impeding flight.
At present, ice is removed from the wings of aircraft prior
to ~light by spraying them with a de-icing fluid. Typically the
de-icing fluid is composed of ethylene glycol, propylene glycol
or a mixture of the two. At present, at most sites, deicing
fluid is discharyed as waste a~ter a single use. This practice
burdens the environment, and wastes the glycol. Accordingly, is
a need for recovering and recycling spent deicing fluid.
At present, commercial recycling of glycol involves
filtration, deionization or distillation. These processes
2 7
provide for recovery of glycol that is not, in general,
sufficiently pure to be used for aircraft deicing applications.
Ethylene and propylene glycols are also used as antifreeze
for automobile and truck engines. These antifreeze fluids must
be replaced after extended use. There is also a need to recover
glycols from these fluids for subse~uent use.
_mmary o~ he Invention
The present invention provid~s for the e.conomical recovery
of glycols from spent industrial fluids. The invention provides
for recovery of glycols of sufficient purity for use in aircraft
deicing. Water is stripped from glycol using a packed, heated
tower. The isolated glycols are purified using vacuum
distillation. Volatile organic compounds contaminaking e~fluent
produced in the process are removed, permitting discharge of
wa~te water into the environment. In a preferred embodiment,
grease, oil and sclids are separated from spent glycols by
mechanical means prior to stripping.
Brief Descrlp on Of The Drawinqs
Figure 1 is a schematic drawing of a settling tank for
cleaning spent glycol containing industrial fluid.
Figure 2 is a schematic drawing of apparatus for recovery of
spent glycol from i.ndustrial fluid.
Figure 3 is a schematic drawing of apparatus or removal of
volatile organic compounds ~rom effluent produced by the
~ 2 ~ .
apparatus for recovery of spent glycol from industrial fluid.
Detailed Description Of The Invention
It is preferred that grease, oil and sediment are removed
from spent industrial fluids prior to isolation and purification
of glycols from them. Preferred means for removal of grease, oil
and sediment is a settling tank with a steel m~sh partition
aeparating first and second portions. As shown in Figure 1,
spent industrial fluid ("in~luent") is added to a settling tank 1
by way of inlet pipe 2. First portion, 3 of settling tank 1 is
separated from second portion, 4, by partition 5. Partition 5 is
preferably a sheet of steel mesh.
In operation, spent glycol containing industrial fluid is
permitted to stand in settling tank 1 until sediment has fallen
to the bottom o~ tank 1, while organic contaminants, such as fuel
and hydraulic fluid have floated to the top. The organic
contaminants are removed Erom the surface of first portion 3 of
tank 1 by pump 6. Sediment is collected and removed from the
bottom of tank 1 as sludge. Partition 5 confines the organic
contaminants to first portion 3 of tank 1. Cleaned, spent glycol
containing industrial fluid is removed by way of line 7 from the
second portion 4 of tank 1.
Grease, oil and sediment may also be removed from spent
glycol containing industrial fluid prior to stripping and
purification with a cartridge type filter.
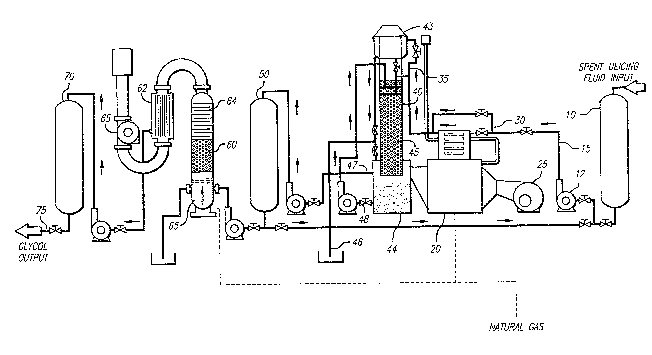
Referring to Figure 2, cleaned spent glycol containiny ~b~
.
industrial fluid is accumulated in raw product holding tank lo.
In operation, the spent fluid is pumped using a pneumatic
diaphragm pump 12 via stainless steel piping ~5 to heat exchanger
30, wherein it is warmed by 8 to 11 degrees centigrade using heat
from natural gas heater 20. Blower 25 is drives hot air from
heater 20 through packed tower 40. Preferably, blower 25 is an
adjustable low profile air blower powered by a continuous-run
high e~ficiency electric motor.
Water is stripped from the spent glycol containing
industrial fluid in packed tower ~0. Warm air from heater 20 is
introduced at the bottom of packed tower 40. The warmed fluid is
introduced at the top of packed tower 40 by way of steel piping
35. The warmed fluid percolates through stainless steel packing
45 where warm air produced by heater 20 drives oEf water by the
process of evaporation. In the presently preferred embodiment,
the interior of tower 40 is maintained at a temperature of 140 to
160 degrees centigrade bv hot air from heater 20. Presently
preferred packing is stainless steel packing obtained from Lan
Pac~.
Stripped ylycol accumulates in reservoir 44 ak the base of
packed tower 40. Water stripped from the spent glycol leaves
packed tower 40 as vapor through exhaust ceiling 43. Condensate
is removed via drain outlet 46.
The size of packed tower 40 is governed by the rate at which
spent glycol containing fluid is to be processed. In the
presently pre~erred embodimenk, the system accommodates a volume
2 ~
.
flow of about 10 gallons per minute, using tower dimensions of 20
inches (diameter~ and 25 feet (height). ~ifferent capacities may
be accommodated by adjusting dimensions using relationships well
known in chemical process engineering.
Progress of the skripping process is monitored by measuring
the specific gravity of the stripped glycol accumulated in
reservoir 44. Accumulated stripped glycol can be recirculated
through packed tower 40 by way of recirculating loop bypass valve
48 and associated pump until the glycol is sufficiently
dehydrated.
Dehydrated spent glycol is fed from packed tower outlet 47
to secondary holding tank 50. Therea~ter, the dehydrated glycol
is puriPied in vacuum distillation tower 60. By conducting the
distillation under vacuum, thermal decomposition of the glycol
and acid formation is avoided. Dehydrated glycol is introduced
into boiler 65 in distillation tower ~0 where it is vaporized by
a liquid propane or natural gas heater. Vacuum is provided by
vacuum pump 66. Glycol is cooled by condenser 64, continuing to
condense as further cooling is ef~ected by passive heat exchanger
62. Purified glycol is withdrawn at passive heat exchanger 62
and is stored in finished product hatch tank 70. Purified glycol
i5 wikhdrawn Prom the system at piping outlet 75.
The puri~ied glycol recovered is about 99.5% pure, and is
appropriate for use as deicing fluid for aircraft. I'he liquid
recovered ~rom distillation tower 60 includes bufPers, deicing
Pluid additives and volatile organic compounds. This liquid may
2 ~
be discharged, or ~urther purified as described below.
Waste water rom the packed tower and vacuum distillation is
optionally purified using a combination of filtration, ozone
driven oxidation of volatile oryanic compounds, and activated
polymers ~or removal of charged species. As shown in Figure 3,
waste water is collected in reservoir 210. Introduction of
ozone, produced by ozone generator 280, initiatPs destruction of
volatile organic compounds. Caustic soda (sodium hydroxide
solution) is stored in caustic batch mixing tank 220, and is
added as needed to polymer mixing tank 250, to achieve a pH in
the range 9.1 to g.5. Optionally, base may also be added to raw
product holding tank 210.
Cation and anion polymer suspension, such as those
available from MatrixrM or AquaBenrM are prepared in cation batch
mixing tank 240 and anion batch mixing tank 230 respectively.
The suspensions are prepared to achieve a concentration of about
7 ppm each. The polymer suspension are added to waste water in
polymer mixing tank 250, when the pH of the waste water has been
adjusted with base.
The polymer treated waste water is allowed to stand in
the polymer mixing tank 250 until solids have precipitated. The
sludge is piped away to sludge aging tank 260, and stored.
ThereaEter the sludge is removed and compressed in filter press
270. The polymer treated waste water is piped to ozone reactor
vessel 285, where it is treaited with ozone (5-20%) generated by
spark discharge or ultraviolet radiation in ozone generator 280
~J
`:
Ultraviolet radiation generation of ozone i5 preferred as it is
more energy efficient than spark discharge. The ozone gas
oxidizes and breaks down volatile organic compounds in the
polymer treated waste water.
Water from the ozone reactor vessel is filtered using a
combination of sand (290~ and twin activated carbon absorption
beds (295), before discharge. The sand filter helps prevent
blinding of the activated carbon. While one carbon absorption
bed is in use the other is reyenerated with ozone. The treated
water has contaminant levels reduced to about 1 ppm, which meets
or exceeds present local and federal governmental requirements.
The foregoing description and drawings are for illustrative
purposes only. Other embodiments of the inventions disclosed
will be evident to those of ordinary skill. ''