Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
W09~1446 - 1- P~~ 13
4-BENZOYLISOXAZOLE DERIVATIVES AND THEIR USE AS HERBICIDES
FIELD OF THE INVENTION
This invention relates to novel 4-h~.l vyli~ui~ul~ derivatives,
compositions containing them, processes for their ~ ,p~ ~Liull and
their use as herbicides.
BACKGROUND ART
Herbicidal 4-~l~uyli~u~ul~ are known from the literature,
for example in European Patent Publication Nos. 0418175, 0487195,
0524018, 0527036, 0527037, 0560482 and 0580439. However none
of the above pllhlir~tinnc disclose or suggest the presence of an
alku~..ubull~l~lillùalkylene or ~ul~llull~l~lillvalkylene
~ or Qllhctitllt~d aminoalkylidene ~ on the
benzoyl ring.
DESCRIPTION OF THE INVENTION
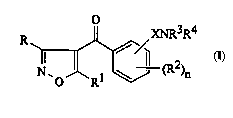
The present invention provides 4-~.,~v~ u~vle derivatives
of forrnula (1):
XNR3R4
R =--(R2)n
(I)
wherein
R represents hydrogen or -CO2R5;
Rl IC~JIC ' -
a straight- or branched- chain alkyl group containing up to six
carbon atomQ which is optionally ~ d by one or more
halogen atoms; or
a cycloalkyl group containing from three to six carbon atoms
which is optionally ~,.I .~I;l.,t. d by one or more halogen atoms which
may be the same or different;
R2 ICIJIU;~
a halogen atom;
.. . . _ _
WO 95131446 2 1 9 0 0 0 1 PCT/EP95101813
a straight- or branched- chain alkyl, alkenyl or alkynyl group
containing up to six carbon atoms which is optionally cl~hs1itlltPd by
one or more halogen atoms;
a straight- or branched- chain alkyl group containing up to six
carbon atoms which is cllhctihltt~d by a group -oR6;
a group selected from nitro, cyano, -CO2R6, -S(O)pR61,
-O(CH2)mOR61,-COR61,-NR6R7~-CoNR8R9, oR6
-CH2S(O)pR61, -NR3R4 and phenyl optionally c-lhCtitl~t~d by from
one to five groups R21 which may be the same or different;
R3 I~,IJlC~
hydrogen;
a straight- or branched- chain alkyl, alkenyl or alkynyl group
containing up to ten carbon atoms which is optionally C~hCtitllt~d by
one or more halogen atoms;
a cycloalkyl group containing from three to six carbon atoms
which is optionally ' - ~ by one or more halogen atoms;
-(CH2)W (phenyl optionally ~. .1.,1 i l l . t- cl by from one to five
groups R21 which may be the same or different);.
or -ORS;
R4 represents -S(O)qR10 or -C(Z)=Y;
or R3 and R4 together with the nitrogen atom to which they
are attached may form a ring selected from the group consisting of:
~E~ ~E~ ~E)
--N~ N~Y --N~l/NR5
Y Y Y
NR5 ~ E
N~NRS and --N~s(o
5
wherein E represents an alkylene or alkylidene group
containing up to three carbon atoms which is optionally c--h'-i-llt~d
.. . : . . . .. .. _ . . . .. . ... .....
WO 95/31446 2 1 9 0 ~ O 1 PCTJEP9rt/0l8l3
-3 -
by one or more groups R5 which may be the same or different;
X represents -(CR11R12)t
n represent3 zero or an integer &om one or four;
R5 represents a straight- or branched- chain alkyl group
containing up to six carbon atoms optionally cllhCtitllt~d by one or
more halogen atoms;
R6 and R7, which may be the same or different, each
hydrogen or
a straight- or branched- chain alkyl, alkenyl or alkynyl group
containing up to six carbon atoms optionally Cl-hctihltl d by one or
more halogen atoms;
R61 represent~s a straight- or branched- chain alkyl, alkenyl or
alkynyl group containing up to six carbon atoms optionally
lS sllhctihlt~d by one or more halogen atoms;
R8 and R9, which may be the same or different, each
1~1~ _
hydrogen;
a straight- or branched- chain alkyl group containing up to six
20 carbon atoms optionally ~.,1,~1;~.. 1.~ by one or more halogen atoms;
or phenyl optionally ' ~ by &om one to five groups
R21 which may be the same or different;
R10 I~ S~....a,
a straight- or branched- chain alkyl, alkenyl or allynyl group
containing up to six carbon atoms optionally ~ . . t~ by one or
more halogen atoms;
a cycloalkyl group containing &rom three to six carbon atoms
optionally ~ d by one or more halogen atoms;
or -(CH2)W (phenyl optionally 1.~ d by from one to five
groups R21 which may be the same or different);
R11 and R12, which may be the same or different, each
3~11t~.-
hydrogen;
a straight- or branched- chain alkyl group containing up to six
carbon atoms optionally ~ by one or more halogen atoms;
a cycloalkyl group containing &om three to six carbon atoms
WO 95131446 2 1 q O O (~ 1 PCT/EP9~i/01813
-4 -
optionally s~lhctitlltpd by one or more halogen atoms;
or -(CH2)w-(phenyl optionally substituted by from one to five
groups R21 which may be the same or different);
Y represents oxygen or sulphur;
Z is selected from the group consisting of R62, -NR63R64,
-N(R63)NR64R65, -SR62 and oR62;
R62 ~
a straight- or branched- chain alkyl, alkenyl or alkynyl group
containing up to six carbon atoms which is optionally ~ by
one or more halogen atoms; or
-(CH2)w-(phenyl optionally sllbctit~lt~d by from one to five
groups R21 which may be the same or different);
R63, R64 and R65, which may be the same or different, each
I ~,IJI,
hydrogen;
a straight- or branched- chain alkyl, alkenyl or alkynyl group
containing up to six carbon atoms which is optionally s~lbctihlt~d by
one or more halogen atoms; or
-lCH2)W (phenyl optionally CllhCtih~t~d by from one to five
groups R21 which may be the same or different);
R21 represents
a halogen atorn;
a straight- or branched- chain alkyl group containing up to
three carbon atoms optionally Cl~hctihlt~d by one or more halogen
atorns;
or a group selected from -oR5, nitro, cyano and -S(o)pR5;
m represents one, two or three;
p represents zero, one or two;
q represents zero, one or two, (preferably zero or hwo);
t represents one or two;
w represents zero or one;
and ~ri~llh-r~lly acceptable salts thereof, which possess
valuable properties.
In certain cases the groups R1, R2, R3, R4, R5, R6, R61, R62
W095~31446 ' ` ;~ 1 9G~O 1 Pcr/Ep9sJol8l3
_ S _
R63, R64, R65, R7, R~, R9, R10, R 11, R12 and R21 may give rise to
Stcl~O;~ a and geometric isomers. All such forrns are embraced
by the present inYention.
These ~ ,u" I~ in certain aspeCb of their properties,for
example their control post ~l.l.l~.,ll.~ of the weed species Ipomoea
~Lr~21~a and Am:lr~nthll~ retrAIfl~ possess un~YI7ert~rlly high
levels of activity.
By the term "~griA~lltllr~lly acceptable salb" is meant salts the
cations or anions of which are known and accepted in the art for the
formation of salb for agricultural or horticultural use. Preferably
the salb are water-soluble. Suitable salts with bases include alkali
metal (eg. sodium and potassium), alkaline earth metal (eg. calcium
and, ~ "),<~,",.-l,;." andamine(eg.,l;. IllAI~r~l_l~l;ll.,,
, o-L,~l l il-C" ~ r and liv~
salts. Suitable acid addition salts, formed by ~ o~ of formula
(I) containing an amino group, include salts with inorganic acids, for
example ll~L. ' ' ' sulphates, L~ s and nitrates and
salb with organic acids, for example acetic acid.
DETAILED DESCRIPI ION OF THE INVENTION
Cnmro--n-lc of formula (I) in which R4 represents -S(O)qR10
are preferred, as are ~Arll ~ 'l"J "' I~ of formula (I) in which X is
(-CH2-)t and t is one.
Another preferred class of l . ' of formula (I) are those
wherein:
Rl l~ b.-
a straight- or branched- chain alkyl group containing up to
three carbon atoms which is optionally ",l ,~l ;l ., l . d by one or more
halogen atoms; or ~lululu,uyl or 1 ..,_~Lyl.r~lu,u.
R2 ~
a halogen atom;
a straight- or branched- chain alkyl, alkenyl or alkYnyl grûup
containing up to four carbon atoms optionally ,l .l ,~ "t~ ~ by one or
more halogen atoms;
WO 95/31446 2 1 9 0 0 0 1 PCTIEP95/01813
- 6 -
a group selected from nitro, cyano, -S(O)pR61, -COR61,
-NR6R7, -CONR8R9 and -oR6;
R3 ~ c~ La.-
hydrogen;
a straight- or branched- chain alkyl, alkenyl or alkynyl group
containing up to six carbon atoms which is optionally ~ i l " ~ d by
one or more halogen atoms;
a cycloalkyl group containing from three to six carbon atoms
which is optionally s~lhstih~t~pd by one or more halogen atoms;
-(CH2h, (phenyl optionally ~llhctitlltpd by from one to five
groups R21 which may be the same or different);
or -oR5;
R4 represents -S(O)qR10 or -C(Z) =Y;
or R3 and R4 together with the nitrogen atom to which they
are attached may form a ring selected from the group consisting of:
~E~ ~E~
N~ and --N`S(O)/
y
wherein E represents an alkylene or alkylidene group
containing up to three carbon atoms which is optionally sl~hCtitllt~d
by one or more groups R5 which may be the same or different;
R6 r.,~ ,S~.Ita.-
a straight- or branched- chain alkyl group containing up to six
carbon atoms optionally sl-hctitl-tprl by one or rnore halogen atoms;
R7 r~ lc~,.. ;a.-
hydrogen or
a straight- or branched- chain alkyl group containing up to six
carbon atorns optionally cl-hstih-ted by one or more halogen atoms;
R8 and R9, which may be the same or different, each
represeMs a straight- or branched- chain alkyl group containing up
to six carbon atoms optionally s~lhctitlltPd by one or more halogen
atoms;
Rl I~
WO g5/31446 2 ~ 9 0 ~ 0 3 PCT/EP95/01813
-7 -
a straight- or branched- chain alkyl group containing up to six
carbon atoms optionally sl-hctitllt~d by one or more halogen atoms;
a cycloalkyl group containing from three to six carbon atoms
optionally cllhCtihlt~d by one or more halogen atoms;
S or -(CH2)w-(phenyl optionally cllh'titl-t~d by from one to five
groups R21 which may be the same or different);
R11 and R12, which may be the same or different, each
hydrogen;
a shraight- or branched- chain alkyl group containing up to
three carbon atoms optionally s~h~tih~t~d by one or more halogen
atoms;
Y represents oxygen;
Z is selected from the group consisting of R62, -NR63R64,
-SR62 and oR62;
R62 represents a straight- or branched- chain alkyl group
containing up to four carbon atoms which is optionally ~ d
by one or more halogen atoms;
R63, R64 and R65, which may be the same or different, each
represents a straight- or branched- chain alkyl group containing up
to six carbon atoms which is optionally ~ d by one or more
halogen atoms;
m represents hwo; and
t represents one.
A ~ preferred class of ~ .u~ of formula (I) are
those wherein:
R1 represents .~.Iuulu,u;l,
R2I~ t~.-
a halogen atûm;
a straight- or branched- chain alkyl group containing up to
three carbon atoms optionally ~ t- d by one or more halogen
atoms;
or -S(O)pR61;
R3 represents a straight- or branched- chain alkyl group
containing up to three carbon atoms which is optionally 'llh'titllt~d
by one or more halogen atoms;
21 90001
WO 95131446 PCTII~P95101813
- 8 -
R4 represents -S(O)qR10;
R5 represents a straight- or branched- chain alkyl group
containing up to three carbon atoms optionally cl~hstit~lted by one or
more halogen atoms;
R61 represents methyl or ethyl;
R10 represents a straight- or branched- chain alkyl group
containing up to three carbon atoms optionally Cllhctitllt~d by one or
more halogen ator~s;
t represents one;
R11 and R12 each represent hydrogen; and
n is zero, one or two
Pcu Li~uldlly important ~;ulll~uullda of formula (I) include the
following:
1. 4-[4-bromo-2-(N-methyl-N-I-I~,Ll,ylaull,hu.l,~l~u,,i,,o-
methyl)benzoyl]-s-~y~lulJlu~qliauAclLùlel
2. 4-[4-chloro-3-(N-methyl-N-methylsul,ullu.. ~l~u.lI.lo-
methyl)-2-lll~ hl~ ~uyl]-5-~lu~lu~J~l;auAc~ule;
3. 4-[4-chloro-3-(N-methyl-N-lll~Lh~:~ul~lu..~lcuuil.o-
methyl)-2-1u~,.h~ llull,l~,~uyl]-S ~-~ JlUlJ,~lioUA~VlC:l
4. ethyl5-~luulu,uyl 1 [2-(N-methyl-N-,ll~LllJl~ul~hul.~l-
I)benzoyl]isoxazole-3-c~l,uAyldle,
5. 5 ~lu,ulu~l q [2-(N-methyl-N-
.elL~ l.o~' ' ',~ 4-L~iLh.ulu~ e.~uyl]isoxazole;
6. 4-[3,4-difluoro-2-(N-methyl-N-
methylclllj' ,l- ., .~1)benzoyl]-5 ~y~,lu,ulu~liauAc~Ol~;
7. 4-[4-chloro-3-fluoro-2-(N-methyl-N-
u.,lLy'~ '2~ yl)benzoyl]-5 ~lulJIu,uyliaùA~ul~,
8. 4-[4-chloro-2-(N-methyl-N-
uu.,LIl~laul~uLully~ yl)benzoyl]-5 ~Iulu~u~;~uAc~vle;
9. 4-[2-(N-methyl-N-
methylsull)llu..~l- ; ,u, Ih~l)benzoyl]-5 ~IUylU~qliauAc~ùle; and
10. 4-[4-fluoro-2-(N-methyl-N-
methylsul~lul.~ ---,-, .I-yl)beyl]-5-~lù,ul`uluylisOAc~ûle~
The numbers 1 to 10 are assigned to these ~U~ J~" .l~ for
reference and irl-~ntifi~-~ti-)n h~ ,;ucLrt-,-.
, . . _ _ _ _
WO 95/31446 2 1 ~ ~ 0 ~ 1 P~ 6l3
_ 9 _
Compounds of formula (I) may be prepared by the application
or adaptation of known methods (i.e. methods heretofore used or
described in the literature), for example as hereinafter described.
In the following ri~crrir~ir)n whe}e symbols appearing in
formulae are not specifically defined, it is to be lln~i~rcrc c!d that they
are "as h~ b~vl~ defined" in ac~uldd l~c with the first definition
of each symbol in the crer ifi~rinn It is to be uud~ ood that in
the dc~ ulio.~ of the following processes ~he sequences may be
performed in different orders, and that suitable protecting groups
may be required to achieve the ~ v . -l~ sought.
According to a feature of the present invention ~:U~ UUil~b of
formula (I) in which R represents hydrogen may be prepared by the
reaction of a compound of formula (II):
O O
R~ ~R4
(Il)
wherein L is a leaving group and R 1, R2, R3, R4, n and X are
as hcl ~;llb~. .; defined, with ~.J.L UA~' ~ or a salt of
h~dluAyld~uc. II~ A~lalldulc h~d.. ' '- '- is generally
preferred. Generally L is alkoxy, for example ethoxy, or N,N-
20 dialkylamino, for example diu-.. ~ 1.. . uiuo. The reaction is generally
carried out in an organic solvent such as ethanol or æ~ or a
mixture of a water-miscible organic solvent and water, preferably in
a ratio of orgarïic solvent: water of from 1:99 to 99:1, optionally in
the presence of a base or acid acceptor such as tr;~,l.yl~.~ or
sodium acetate at a Itlll~.ldLul~: from 0 to 100C.
According to a further feature of the present invention
c.. l-u~ .. i~ of formula (I) in which R represents hydrogen may be
prepared by the reaction of a compound of formula (III):
N~
(III)
WO95/31446 ~1 9:~Q~ PCT/EP95101813
- 10-
wherein Rl is as h.,~ b~ru~: defined and yl represents a
carboxyl group or a reactive derivative thereof (such as a carboxylic
acid chloride or carboxylic ester), or a cyano group, with an
~IplV~ C Ul~ it` reagent such as a Grignard reagent or
an vl~;,.. ~vl,ll.;l"., reagent. The reaction is generally carried out in
an inert solvent such as ether or tetrahydrofuran at a tGlll~J~ldlUlC
from 0C to the reflux ~C~ Ul~ of the mixture.
According to a further feature of the present invention
v~ of formula (1) in which R is hydrogen may be prepared
by reaction of a compound of formula (III), as hereinbefore defined,
with an ~ Iu,vlid~e organotin reagent, such as a trialkyltin
derivative, optionally using transition metal catalysis such as
bis)~ yl~ k;~)palladium (II) dichloride, carried out in an
inert solvent such as ~ d-urul~ul or dioxan at a hl~ u
from 0 to the reflux ~ G of the mixture.
According to a further feature of the present invention
of formula (1) in which R is hydrogen may also be
prepared by the oxidation of a compound of formula (Illa):
OH XNR3R4
U ~--(R2)n
(ma)
wherein Rl, R2, R3, R4, X and n are as ~ lb~,fu~ c defined.
The oxidation is performed using for example chromic acid
(prepared from chromium trioxide and aqueous sulphuric acid) in
acetone.
According to a further feature of the present invention
~r., ~ ". .l~ of formula (I) wherein R represents a group -CO2RS, q
represents 0 or 2 and R2 represents a group Ræ which is as
h~l~h~h~,fulG defined for R2 provided that p is 0 or 2, may be
prepared by the reaction of a compound of formula (IV):
WO 95/31446 PCTIEP95/01813
~ ~ 90~ 1
- 11
p
XNR3R4
c R \~/~1 (R22
(IV)
wherein Rl, R22, R3, R4, X and n are as llc.~;lll,~fu~
defined, q is 0 or 2 and P is a leaving group such as
N,N-dialkylamino, with R502CC(Z1) = NOH wherein R5 is as
h~ b~fulc defined and z1 is a halogen atom. Generally zl is
chlorine o} bromine. The reaction is generally performed in an inert
solvent such as toluene or dichlolùu-~,~li~lc either in the presence of
a base such as ~ L.~ lc or a catalyst such as a 4 Angstrom
molecular sieve or fluoride ion.
According to a further feature of the present invention
c~ ,uL~ll 7~ of formula I in which R represents a group -Co2R5, q
represents 0 or 2 and R2 represents a group R22 as h.,l ~i-lb~fvl ~
defined, may be prepared by the reaction of a compound of formula
(V):
. o
l l XNR3R4
Rl ~ (R22)n
(V)
wherein Rl, Ræ, R3, R4, X and n are as h~,~h~h~fu~i defined
and q represents 0 ûr 2, with a compound of formula
R502CC(Zl)=NOH, wherein zl and R5 are as h~ b.,ful~
defined. The reaction is generally performed in an inert solvent
such as toluene or i: ' ~Jll.~lh~ c optionally in the presence of a
base such as ~ ku. il.c or a catalyst such as a 4 Angstrom
molecular sieve or fluoride ion. The reaction can be carried out at a
t~ between room t~ la~ul~ and the reflux
of the mixture.
According to a further feature of the present invention
Cul~ of formula (I) wherein R represents -Co2R5, q
represents 0 or 2 and R2 represents a group RZ as h~ lb~rulc
. , , . . . .. . _ . _ . . _ _ . . . . _ _ . . _ _ .
WO 95/31446 2 ~ 9 ~ PCTIEP95101813
-12-
defined, may be prepared by the reaction of a salt of a compound of
formula (Vl):
O. O
R~
b,~ (R22)
(~)
wherein Rl, Ræ, R3, R4, X and n are as h~,~cillb~,~v~c defined
and q represents 0 or 2, with a compound of formula
R502CC(Z1) = NOH, wherein R5 and z1 are as h~ cfvl ~
defined. Preferred salts include sodium or " ,. ~ ". 1 I l l salts. The
reaction may be performed in an inert solvent such as
I;~IIlVlUll~ dulc or ~c~t~-nitril~ at a ~ between room
L~lu,u~.a~ul~ and the reflux ~IlIu~,ldLUI~ of the mixture.
According to a further feature of the present invendon
compounds of formula I may be prepared by the reaction of a
compound of formula (Vll):
n
(Vll)
wherein R, Rl, R2, X and n are as h.,.~,; L.,fvl~ defined and
L1 is a leaving ~roup, with an amine of formula H-NR3R4, wherein
R3 and R4 are as ll~ ,;lL~,fv~c defined. Ll is preferably selected
from bromine, chlorine or methylsulphonyloxy (most preferably
bromine). The reaction is typically carried out in the presence of a
strong base such as potassium tert-butoxide in a polar solvent such
as N,N- li-~ ylr-l "~"" :Ar at a l.,lllp~,la~ul~ between -20C and
10QC.
lu~ d;à~ in the ~ ,t,d aLiull of f~ l"J~ of formula (I)
may be prepared by the -I.lJI;. ~1;.... or adaptation of known
methods.
Compounds of formula (II) in which L represents alkoxy or
N,N-dialkylamino may be prepared by the reaction of a compound
of formula (Vl) with either a trialkyl ul~ rl~l Ill"lr such as triethyl
.. . _ _ _ , . .. . . . _ ... .
WO9Ç/31446 f'J 1 9 0 ~ ~ j PCrrEP9Ç/01813
-13-
ulll hf~ oraN,N-di~ ylrù ~ ll;flf dialkyl acetalsuchas
N,N-~ lly!r~ l l l l ;flf dimethyl acetal.
The reaction with triethyl u- ~l~oru~ dlC can be carried out in
the presence of acetic anhydride at the reflux tC.lllJ~.ld~UiC of the
mixture and the reaction with di~ lylro~ .. lidf dialkyl acetal is
carried out optionally in the presence of an inert solvent at a
a~ulc from room ~ c to the reflux t~ d~UlC of
the mixture.
Cnmro~nrlc of formula (Illa) may be prepared by the
treatment of a compound of formula (m) above in which yl
represents a carboxyl group such as an aldehyde, treatment witb an
d,uplu~ c Ul~ ` reagent such as a Grignard reagent or
or~n~lithi~-m reagent. The reaction is generally carried out in an
inert solvent such as ether of tetral.~d.uru.dn at a t~ ulc rrom
0C to reflux t~ c of the mixture.
C-)mro~r~lc of formula (IV) may be prepared by the reaction
of a compound of formula (Vlll) with a benzoyl chloride of formula
(IX)
Ri~c --XN(RR22R)4,
(Vlll) (IX)
wberein Rl, Ræ, R3, R4, X n and P are as h~,h~b~u~c
defined provided that the group -XNR3R4 is not ortho to the
carboxyl group, and q represents 0 or 2. The reaction is generally
ca ried out in tbe presence of an organic base such as tri~lLyl~. u.~,
in an inert solvent such as toluene or di~hLI-u~ f at a
e between -20C and room ~ I c.
('omrollnAc of formula (V) may be prepared by the
mf t lllqti~m of an acetylene of formula (X):
Rl~CH (X)
- 30 wherein Rl is as ~ b.,fu~c defined, followed by reaction of
the metal salt thus obtained with a benzoyl chloride of formula (IX).
The mf t~llqtinn is generally performed using n-butyl lithium in an
, . . _ _ _ .. . . ... . .. . ..... .. ..
WO 95131446 2 ~ S~ O O O I PCT/EP95101813
-14-
inert solvent such as ether or tetrahydrofuran at a L~ llJCldtulc from
-78C to 0C. The ,~ ~l ,c~ reaction with the benzoyl chloride is
carried out in the same solvent at a tclll~,cl~lul c bet veen -78C and
room ~ ule.
Compounds o formula (Vl) may be prepa}ed by the reaction
of an acid chloride of formula (IX) with the metal salt of a
compound of formula (XI):
O O
R~ OtBu
~XI)
wherein Rl is as h~.uu~ rulc defined, to give a compound of
formula (XII):
O O
J \~2R4
CO2tBu (R )n
(~)
wherein Rl, R22~ R3, R4, X and n are as l~ rul~ defined
and q is 0 or 2, which is ~ y dc~rl,ù~l~tcd to give a
compound of formula (VI). The reaction to produce the metal salt
of a compound of formula (Xl) is generally performed in a solvent
such as a lower alcohol, preferably methanol. Preferably the metal
is ~g ` The metal salt of the compound of formula (Xl) is
~ reacted with an acid chloride of formula (IX) in an
inert solvent such as toluene or ~ ~ t .. ::, ;lr The dc~all,u~l~Liull is
generally performed by refluxing the compound of formula (XII) in
the presence of a catalyst, such as para-t~ nl c~llrhtmir acid, in an
inert solvent e.g. toluene.
Alt~lllaliv~ly if the group -XNR3R4 is in the position ortho to
the carboxyl group then the benzoyl chloride of formula (IX) may
be replaced by a compound of formula (XIII):
woss/3~446 2~ ~05D~ r~ 3
- 15 -
O O
XNR3R4
R150 \~--(R22)n
(xm)
in which R22, R3, R4, X and n are as ~ ln,rul e defined
and R15 is a lower alkyl group, preferably an ethyl group. The
compound of formula (Xm) may be reacted with the metal salt of a
compound of formula (X) in the manner described for the
formation of i"t~ of formula (Xll).
mrolln~ic of formula (Vl) may also be prepared by the
reaction of a benzoic acid ester of formula (XIV):
CO2R1 5
I~ XNR3R4
~;--(R22)n
(xr~ .
wherein R15, R21, R3, R4, X and n are as ~ ,h~.,ru/e
defined, q is 0 or 2, with a compound of formula (XV):
Rl-C(O)-CH3 (XV)
wherein Rl is as }~ b~fulc defined. The reaction is
generally performed in a solvent such as diethyl ether,
urul~l or N,N- lil.l.,lll~ ' Cl ' ~, in the presence of a
base, preferably an aL~cali metal base such as sodium hydride or a
lithiurn amide base such as lithium diiau~lu~ ide~ at a
t~lu,u~ Lul~ from 0C to reflux t~,.lll)~,l_ ti.
mrolln-iC of formula (Vll) may be prepared from the
~,ulli auuuLu~, compound of formula (VII) in which Ll is replaced
by hydrogen. Where Ll is bromine the reaction is preferably
performed using N-l,., ~ in an inert solvent such as
carbon t. 1.~ , optionally in the presence of UV light or a
free radical initiator such as azo-bis-ia~ul~lu~ lile at a
l,u~,~alul~ between room t~ lul~ and the reflux
of the mixture.
Acid chlorides of formula (IX) in which the group -XNR3R4
wo ss/31446 2 ~ 1 F.~ ol~
- 16-
is not ortho to the carboxyl group may be prepared by the reaction
of a benzoic acid of formula (XVI):
C02H
XNR3R4
22
(XVI)
wherein R22, R3, R4, X and n are as ~.. ,.ci~ rulc defined and
q is 0 or 2, and the group -XNR3R4 is not ortho to the carboxyl
group, with a l ' ' 3 agent, for example thionyl chloride at the
reflux lelll~laLulc of the mixture or with oxalyl chloride in an inert
solvent such as di~ -u-l~ u-c or 1,2-d;~lllv-u~ u~ optionally in
the presence of a catalyst such as N,N-dilll~ ' ~ at a
from -20C to the reflux ~f " '1'- "1 ll ' C of the mixture.
A number of the benzoic acids of formula (XVI) are novel and as
such constitute a further feature of the present invention.
T..~ ....P.l;-t~ C of formula (XIII) may be prepared by the
reaction of the benzoic acids of formula (XVI) wi~h an alkyl
~ ' ' urullll.~t~, CIC02R15 such as ethyl ~llo~uru~ dt~. The
reaction is carried out in the presence of a base such as an organic
base (e.g. Ll;_~l,' ' - or pyridine) in an inert solvent such as
Ji~lllvluu~,LL~.~, N,N~ or t~ .uru.~ll at a
~ . . . c between -40C and room ~ c. Compounds of
formula (XVI) may be prepared from ~ .l-u~ in which the
group XNR3R4 is replaced by a group XLI in which L1 is a leaving
group (preferably chlorine or bromine) by reaction with the amino
compound HNR3R4. The reaction is carried out in the presence of
a strong base such as potassium tert-butoxide in a polar solvent such
as N,N-di~ ,.L~"l ' at a t~ UlC between -20C and
room ~ Lulc.
T-- - " of formulae (III), (vm), (X), (XIV) and (XV)
are known or may be prepared by the ~ liu.. or s~ rtzltinn of
known methods.
Those skilled in the art will appreciate that some ~OIIIIJUUII~
of formula (I) may be prepared by the illt~.~Ull._l~iUll of other
W~9~i/31446 2~ qO~l
~UIIl~JUUlld:~ of formula (I) and such illt.,.~:ull.~ iul~ constitute yet
more features of the present invention. Examples of such
illL~.~ull~ iol~ are hereafter described.
According to a further feature of the present invention
~UIll~OUlld:~ in which p is one or two and/or q is one or two may be
prepared by the oxidation of the sulphur atom of the ~;UII~ IJUlldillg
.u~ in wbich p and/or q is zero or one. The oxidation of the
sulphur atom is generally carried out using for example
3-~ lu~u~.uAyb~.lLuic acid in an inert solvent such as
10 dichlululll~.lld-lc at a t~,.. ~.. ~lllllt: from -40C to room
1` ~ .1~ ., 1 . ., ~, or hydrogen peroxide in acetic acid in the presence of
acetic anhydride or Cull~ l dt~ sulphuric acid, or using potassium
p~,-UAylllllllll~llll~h-~t~ as the oxidising agent.
The following EAamples illustrate the ~ dlatiOl~ of
(~ ,.. l-., .. l~ of formula (I) and the Reference Examples illustrate
the ,Ul~tJdldliùu of i--t- . ",. .I: .t. S in their synthesis. In the present
m-p- means melting point. Where the letters NMR
appear, the ~ h~r~ rictirc of the proton nudear magnetic
resonance spectrum follow. Unless otherwise specified the
~.,-~IIL~ are by weight.
F.~IVIPLF. 1
Crude 1-[4-bromo-2-(N-methyl-N-lll~ ulul~ullyl~llillo-
methyl)phenyl]-3-~lu,ulu,u~. 2-(N,N-
dilu.,~ L.~)propan-1,3-dione (0.86g) was dissolved in
a mixture of ethanol and methanol and the mixture was cooled to
-10C II~dluA~ldluil.., hydrochloride (0.21g) was added and the
mixture was allowed to warm to room ~ and stirred for 1
hour. The resultant ~lcr~n~ n was poured onto cold water and
extracted with ethyl acetate. The eAtracts were washed with brine,
dried (arlhydrous Na2SO4) and filtered. The filtrate was
uld~,d to dryness and the residue was purified twice by
~Illullld~utyd,ull~ to give 4-[4-bromo-2-(N-methyl-N-
(Ill~ lsul~u~lu..~ yl)benzoyl]-5-c~lululu~
(0.36g) (compound 1) as a beige gum, NMR (CDC13) 1.2(m,2H),
135(m,2H), 2.6(m,1H), 2.8(s3H), 2.85(s,3H), 4.5(s,2H), 735(d,1H),
.. . .. . .. . . . .. .
WO9~/31446 2;~ q~ PCT/EP95101813
- 18 -
7.55(d,1H), 7.85(s,1H), 8.2(s,1H).
The following ~u~ Juullda were similarly prepared:
4-[3,4-difluoro-2-(N-methyl-N-
methylsul~ ull,~ l)benzoyl]-s-~ lu~ul u~ su~aLule
(compound 6) as a yellow gum, NMR (CDC13) 1.2(m,2H),
1.35(m,2H), 2.59(m,1H), 2.8(s,6H), 2.53(s,2H), 7.25(m,2H),
8.2(s,1H);
4-[4-chloro-3-fluoro-2-(N-methyl-N-
~ L.~lauliJllull,yl~l~ lyl)benzoyl]-5~ luiJluiJ~liau~a~lc
(compound 7) as an orange gum, NMR (CDC13) 1.15(m,2H),
1.28(m,2H), 2.54(m,1H), 2.73(s,6H), 4.45(s,2H), 7.12(m,1H),
7.42(m,1H), 8.14(s,1H);
4-[4-chloro-2-(N-methyl-N-
~ Ll~ ul~i~lu..~ )bellzoyl]-5-~luiJluv~yliau~aLvlc
(compound 8), NMR (CDC13) 1.2(mt2H), 1.28(mt2H), 2.53(m, lH),
2.74(s,3H), 2.8(s,3H), 4.4(s,2H), 7.32(m,1H), 7.63(s,1H), 8.16(s,1H);
4-[2-(N-methyl-N :' ~la~liJl-u-.,' ' ~:)benzûyl]-5-
~.,IU~lUiJ~IiaV.aLVlc (compound 9) as an orange gum, NMR
(CDC13) 1.15(m,2H), 1.29(~,2H), 255(m,1H), 2.72(s,3H),
2.8(s,3H), 4.45(s,2H), 7.38(m,1H), 7.5(m,2H), 7.65(m,1H),
8.18(s,1H);
4-[4-fluoro-2-(N-methyl-N-
methy'isuliul-u--,~ h,~l)benzOyl]-5-~1uiJlu~l;
(compound 10) as a yeiiow gum, NMR (CDC13) 1.19(m,2H),
1.3(m,2H), 2.55(m,1H), 2.78(s,3H), 2.85(s,3H), 4.48(s,2H),
7.05(m,1H), 7.45(m,2H), 8.19(s,1H).
F~MPLF. 2
LuA.yla~ c ll.~dlu~lllo~idc (0.38g) was added to a stirred
solution of 1-[4-chloro-3-(N-methyl-N~ a~ oll~lal-iil.û-
methyl)-2-lll~L~ aul~ ~l]-3-cyclopropyl-2-
cLLu~lil.,.ll~l~,.l~iJlu~aui-1,3-dione(2.0g)inethanol. Sodium
acetate (0.43g) was then added. The resultant sl-~r~nr;o ~ was
stirred at room lellliJ~-aLul~ for 2 hours and left to stand overnight.
It was evaporated to dryness and the residue was suspended in
ether, washed with water, dried (anhydrous MgSO4) and filtered.
wo 95/3144C 2 1 9 0 0 0 1 PCTIEP95/01813
- 19-
The filtrate was ~ u-d~,d to dryness and the residue was
triturated with a mixture of ethyl acetate and hexane and filtered to
give 4-[4-chloro-3-(N-méthyl-N-~ l-yl~ul~llu-~ yl)-2-
methylsulul~ vyl]-5-~.lu~,.u~li~u,.a~ule (0.9g) (compound
2) as a cream solid, m.p. 128-13ûC
By IJI u~ ; in a similar manner 5-~lvl~l u,uyl~-[2-(N-
methyl-N-IIl~ ul~llull,~ 1 q
l~iLIuoluu~.llJll,~ vyl]isoxazole (compound 5) was prepared from
the au~ulu,uliàlcl~ sl~hctitlltPd starting material, NMR (CDC13)
1.2(m,2H), 1.3(m,2H), 2.6(m,1H), 2.7(s,3H), 2.8(s,3H), 4.45(s,2H),
7.55(d,1H), 7.6(d,1H), 7.9(s,1H), 8.15(s,1H).
F~AMPL~ 3
3 ~'1 ' ùp~,.u.~yl .,-lLU;c acid (60%, 0.84g) was added to a
15 solution of 4-[4-chloro-3-(N-methyl-~ .. l_llyl~ull~'.vl~,
,1111;.1~1.1. ~JI)-2-II.~ V~I~V.YI]-5 (~IU!JIU~JYIi~U~UIC
(0.5g) in 1' ' ' ul~ he. The mixture was stirred at room
l~llu~,~alul~; overnight then coûled to -25C and filtered. The
filtrate was e a~ulat~d to dryness and the residue was purified by
~1ll ulllalu~ . ', on silica eluted with a mixture of ethyl acetate and
' ' ' ul.l~.Lau.~ to give 4-[4-chloro-3-(N-methyl-N-
u~,lLJl..~l~}.v~l- .;...~ .Jl)-2-methylsul~,llvll~ ..Luyl]-5-
~luylu~ ,. ùle (cûmpound 3, 0.14g) as a pale gum, NMR
(CDC13) 1.15(m,2H), 1.3(m,2H), 2.45(m,1H), 2.9(s,6H), 3.4(s,3H),
5.0(bs,2H), 7.25(d,1H), 7.7(d,1H), g.l(s,lH).
E-XAMPLF. 4
Potassium t-butoxide (1.79g) was added to a stirred, cooled
solution of N-~_lll~ 'i ' ' ( 1.74g) in N,N-dimethyl
form~m;~lP at -5C The mixture was stirred at 10C for 1.5 hours
then re-cooled to -8C. A solution of crude ethyl 4-(2-
blulllulllc~ lv~..Lu.~1)-5-~.lvlJlu~L;,v.~a~ulc-3-.a l,u~làLe (6.0g) in
N,N- li~ rul Illalllid~ was added while, ~ the
t~ .alul~ below 0C. The mixture was stirred at 0C for 1 hour
and at room . . .I.. .- ~, . . ~; for 3 hours. The mixture was poured onto
cold dilute hydrochloric acid and extracted with ethyl acetate. The
WO 95/31446 ~ 2 1 9 0 (~ PCT/EP95/01813
-20-
organic phase was dried (anhydrous MgSO4) and filtered. The
filtrate was evdlJo~ ; d to dryness and the residue was purified twice
by ~Ill U~ U~ y on silica eluted with a mixture of ethyl acetate
and ~luh~A~lc to give ethyl 5-cyclopropyl4-[2-(N-methyl-N-
methylsul~,l-ullyl-, .;, -- " ll~yl)benzoyl]i5Oxazole-3 ~bu~l~le
(Q88g) as a clear gum, NMR (CDC13) 1.05(t,3H), 1.15(m,2H),
13(m,2H), 23(m,1H), 2.8(s,3H), 2.85(s,3H), 4.0(q,2H), 4.6(s,2H),
7.25(t,1H), 7.35(d,1H), 7.55(t,1H), 7.75(d,1H).
R~FE;~F~CE EX~I~JIPI.F. 1
A mixture of crude 1-[4-bromo-2-(N-methyl-N-
Ill~.lI,l~UlLJIIUll~l-",;~-J~.r~ l)phenyl]-3-~ "u.u~l~.u~.~..-1,3-
dione (0.8g) and N,N-~ lr..~ dimethyl acetal in toluene
was stirred at room t- . "I,r . ,.1.1l ~ for 0.5 hours and at 80C for 15
minutes. It was cooled to room ~ ; and ~ u~L~,d to
dryness. Toluene was added and it was re c~a~ulah,d to gfve crude
1-[4-bromo-2-(N-methyl-N~ bulpl.ullJl-, ,. :l.yl)phenyl]-3-
~lu~J-u~l-2-(N,N-dilll.,LIl~ )propan-13-dione
(0.86g) as a red oil.
The following ~ u~ were similarly prepared:
3-~.lU~JlU~ [3,4-difluûro-2-(N-methyl-N-
LIl~Lul,ull~ l)phenyl]-2-(N,N-
dilll~,LII~' - 1 ...~.. :h~ )propan-l~3-diûne as an orange gum,
NMR (CDC13) 0.66(m,2H), 0.9(m,2H), 1.17(m,1H), 2.09(m,1H),
2.76(s,6H), 2.8(s,3H), 3.1(s,brm), 4.5(s,2H), 7.2(m,3H);
1-[4-chloro-3-fluoro-2-(N-methyl-N-
.,Lh~l~ul,uh~ l)phenyl]-3 ~ 1ulJlu~J~1-2-(N,N-
liul~ )propan-1,3-dione, NMR 0.75(m,2H),
0.97(m,2H), 2.19(m,1H), 2.8(s,6H), 2.87(s,6H), 3.2(brm,1H),
4.55(s,2H), 7.25(m,2H);
1-[4-chloro-2-(N ~ ,Lllyl N
methylsul,ul.u.l~l~,..: .,... I~.~I)phenyl]-3 ~,1ul~lu~1-2-(N,N-
dilll~Lll.~l - I I I; I I JI I . : l ,~ c)propan~1~3-dione as a yellow gum;
3-~.lol,lul,~ 1-[2-(N-methyl-N-
35 methylsul~hu.lyl-,,.;.,.. , . ll ~I)phenyl]-2-(N,N-
dill-~ ylene)propan-l~3-dione as an orange gum;
wo 95~31446 2 ~ 9 0 D O 1 PCTIEP95/01813
-21 -
3-~.lolulu~1-1-[4-fluûro-2-(N-methyl-N-
IlI~.~II,yl~UI~IIUll,y~ y~)phenyl]-2-(NlN
~UU~.Il~l-.. " :l~ylene)propan-1,3-dione.
~F,Fli',RF,~CE EXAMPI F 2
A miAture of 1-[4-chloro-3-(N-methyl-N-ll.~Lllyl~ull.hul.~l-
~"~ Ih~'l)_2_1l~ UI~ ..JI]-3 (y~ lu~yl,ulu,udll-1,3-
dione (l.Bg) and ~ llylul ~ .r. ,. . I-A~ ( 1.38g) in acetic anhydride
was stirred and heated at refluA for 4 hûurs. It was cooled and
evaporated to dryness. The residue was dissolved in toluene and re-
~a~Jol~t~.d to 6ive 1-[4-chloro-3-(N-methyl-N-
methylsullul.u..,~l~-.-' ,...- :l~l)-2-ll~ llyl~ul~ 'yl~ l]-3~
~lv,u~u,uyl-2 ell-uAyl.-~lllyl~ -uy~ul-l~3-dione (2.0g) as a brown
oil.
lS By u-u~c~.6 in a similar manner 3-~luylu,u~: 2-
~ uA,~u~ -1-[2-(N-methyl-N-l-l~ 's~ ul-J~
4-~lill~.v.u...~ J'i ' jl]propan-1,3-dione was prepared from the
a~lulu,uliat~ d starting material.
12F,FFUF.~CE EX~MPLF` 3
AmiAtnreofethyl 5-~,lùl~ul~yl ~ (2-~ ,.h~ cl. vyl)-
isoAaA,ole-3~1.uAylale (6.71g) and N-l,.l ' ' ''- (4.15g) in
carbon l ~ . l was stirred and irradiated using a UV
r~ .r AI reactor for 1 hour. The mixture was cooled to 0C
for 2 hours and filtered. The filtrate was c~a~ulat~,d to dryness to
give crude eth~yl 4-(2-blu~v~ lb.,l~vyl)-5-~loulu,uyli~uAa~ule-
3 ~lluAylht~, (6.0g).
RF.l;'F~F.1~1~F F,~AMP!,F, 4
A solution of methyl 4-bromo-2-(N-methyl-N-
methylsul,uLu ,' ' :' ,:)benzoate (6.25g) and ~lu~-u,uyl
methyl ketone (3.13g) in tetrah~l-urul~ul was added to a stirred
of sodium hydride (80% oil tli~r~rC;~m; 134g) in
~.lal~ urul UI containing a little methanol while heating at reflux.
The resultant mixture was cooled and methanol was added followed
by cold aqueous sodium b;~alLullale solution. It was extracted with
WO95/31446 . ,`~ 2l9aoal PCT/EP95101813
-22-
ethyl acetate, washed with brine, dried (anhydrous Na2S04) and
filtered. The filtrate was evaporated to dryness. Attempted
purification of the residue by ~ ulllalu~l~LAully on silica gave crude
1-[4-bromo-2-(N-methyl-N-methylsulul~ul.ylA..,;".-",~ il,yl)phenyl]-3-
S ~lvlu~ul~lulup~l-1,3-dione (0.8g) as a white solid which could not
be further purified.
Ul; ~RFI~CE EXAMPLE 5
A solution of 4-chlorû-3-(N-methyl-N-l~ yl~ul~hullyl-
:i, ,;".. ". IllJl)-2-lll.,.llJl~ullJ}l~ lb~l-Lu~l chloride (2.4g) in toluene
was added to a solution of t-butyl 3~ lulu~u~1-3-u~uulu~;ul~dle
enolate (1.9g) in toluene and the mixture was stirred at
room t~.~l,u.,.~lul~i overnight. Il~J,. ' ' - acid (2M) was added
and the mixture was stirred for 1 hour. The layers were separated
and the organic layer was washed with I~JdIU~IIIUIiC acid (2M) and
water, dried (anhydrous MgS04) and filtered. 4-Toluene sulphonic
acid (Q076g) was added to the resultant solution and the mixture
was stirred and heated at reflux for 6 hours. After cooling the
solution was washed with brine, dried (anhydrous MgS04) and
filtered. The filtrate was e~a~ul~LI,d to dryness and the residue was
purified by .,11l~ on silica eluted with a mixture of ethyl
acetate and hexane to give 1-[4-chloro-3-(N-methyl-N-
methylsul,ul,uu~ 1)-2 ..,~ J s~ lJ~ Ull~l]-3-
~lu,ul uy~ ulu< u-1,3-dione (1.Og) as a yellow oil, NMR (CDC13)
l.O5(rn,2H), 1.25(m,2H), 1.75(m,1H), 2.4(s,3H), 2.7(s,3H),
3.0(s,3H), 4.9(s,2H), 6.1(s,1H), 7.4(d,1H), 7.45(d,1H), 15.9-
16.1(bs,1H).
By ~ diu,~ in a similar manner 3 ~,luu~ullyl-l-
(2-1"~,Ll,~l,ul,~,l":)propan-1,3-dione was prepared, NMR(CDC13)
1.0(m,2H), 1.25(m,2H), 1.7(m,1H), 2.55(s,3H), 6.0(s,1H), 7.2-
7.4(m,3H), 7.5(d,1H), 15.9-16.4(bs,1H).
l2F.F~I~Fl~CE EX~MP! F. 6
T~ lyl~ul ill~ (1.34ml) was added to a stirred cooled solution
of 2-(N-methyl-N-methylsul~uhu,~ ".;.,.. ". illJI)-4-
trifluu, UUI~.ll~ll,.,llLUiC acid (3.0g) in N,N-dil~ ylrul l~ullidc while
... . . ... .. .. . ~ _ _ _ _ ,
WO 9~1446 2 ~ 9 ~ G O 1 PCT/EP95/~1813
-a -
" ~ i"; . ,~ the i~ ,u ~ below 0C The mixture was stirred at
-10C for 0.5 hours then ethyl chlu-urullllalc (0.92ml) was added.
The miAture was again stirred at -10C for 0.75 hours then a
solution of t-butyl 3-~lu~lulJyl-3-u~uylu~J;ulld~c ~
enolate (1.95g) in N,N-dil.l~.ill~lr.,""~ was added while
A;l~ the~ ult;below-5C. Themixturewasstirred
at 0C for Q5 hours and at room L~ l,u~,la~u~ overnight. It was
treated with l.,J.~ acid (2M) and stirred for 0.5 hours then
extracted with ethyl acetate, dried (anhydrous MgSO4) and filtered.
The filtrate was ~v~ul~ to dryness and the residue was dissolved
in toluene and treated with 4-to~ ,l,./";r acid (O.lg). The
mixture was heated at reflux for 2 hours, cooled and diluted with
ethyl acetate. The mixture was washed with brine, dried (anhydrûus
MgSO4) and filtered. The filtrate was cv~.l,u.~t~,~ to dryness and
15 the residue was purified by "I.,.. ~t~ on silica eluted with a
mixture of ethyl acetate and ~.loll~,A~.Le to give 3 ~1u~J~u~yl-1-[2-
(N-methyl-N-l..~ l.u..~ 4-
uu~u~ h_l.yl]propan-1,3-dione (0.53g) as a clear ûil.
The following ~- '-''l'~J'''''I` were similarly prepared:
3-~lulJ~u,ujl-1-[3,4-difluoro-2-(N-methyl-N-
...~,ih,~ h,l)phenyl]propan-1,3-dione as a beige
solid, m p. 9~91C;
1 -[4-chloro-3-fluoro-2-(N-methyl-N-
methylsul,ul.v..~' hyl)phenyl]-3-~.lu~..u~lp.u~.all-1,3-
dione, m p. 8~82C;
1-[4-chlûro-2-(N-methyl-N-
I-u--~l--~ '------ lll~ )phenyl]-3-~lu~Jlu~ u~ rll3
dione, as a beige gum NMR (CDC13) 0.95(m,2H), 1.15(m,2H),
1.69(m,1H), 2.74(s,3H), 2.83(s,3H), 4.51(s,2H), 5.9(s,1H),
7.25(m,1H), 7.38(m,1H), 7.59(m,1H);
3-~.luu. u~l-1-[2-(N-methyl-N-
ul~l~ull~ l)phenyl]propan-1,3-dione as an orange
gum, NMR (CDC13) 1.02(m,2H), 1.24(m,2H), 1.78(m,1H),
2.78(s,3H), 2.9(s,3H), 4.6(s,2H), 5.99(s,1H), 7.36(m,1H), 7.5(m,2H),
7.68(m, lH);
3-.~.lu~-u~yl-1-[4-fluoro-2-(N-methyl-N-
WO 95/31446 ~ 1 q O ~ 0 7 PCT/l~P95101813
-24.
methylsul~ o~ . il.yl)phenyl]propan-1,3-dione as a beige
gum, NMR (CDCl3) 0.95(m,2H), I.l5(m,2H), 1.69(m,1H),
2.75(s,3H), 2.85(s,3H), i.55(s,2H), 5~9(s,1H), 6.95(m,1H),
7.35(m,1H), 7.,45(m,1H).
RFI FUF.NCE EX~MPr.F 7
A mixture of ~ L''' - ""' (3.7g) in methanol containing carbon
L~ o (0.5ml) was warmed gently to initiate the reaction. It
was heated at 50C for 1 hour then cooled t-butyl 3-~ U~lU~1-3-
~v~lulJ;ollalc (27.6g) was added and the mixture was stirred and
heated at reflux for 2 hours. It was cooled and ~va,uul~Lcd to
dryness. Toluene was added and it was re c.a~ulaL~,d to give the
; "., enolate of t-butyl 3-c~lu~lu~yl-3-o~.v~lu~;~llà~e (38.6g)
as a white solid.
RFFEI'FNCE F.X~Mp! F. 8
Oxalyl chloride (l.Oml) was added to a solution of 4-chloro-3-
(N-methyl-N-~ Lll~ yhu~~ l)-2- ' ,~bull ll~"lyl-
benzoic acid (2.3g) in~' ' ' uLll~,.hal~c containing
dul.~,Lll~ul '- (1 drop). The mixture was stirred and heated at
reflux for Q75 hours then cooled and e.a,uulat~,~ to dryness to give
4-chloro-3-(N-methyl-N-LIl.,.h~ 1,yl)-2-
lU",h~ LLV~I chloride (2.4g) as a brown oil.
R l ~ r.l .CE EXAMpl F. 9
Amixture of 1-(2-Lu~.~ Jh~ 1)-3 ~1u~lluL~ Jlu,uàll-1,3-
dione (10.23g) and ~ (1.21g) in methanol was heated with
carbon l~ .l, l .l. .. ;~ f (Q3ml). The mixture was stirred until
.,llce ceased and the mixture was c~a~ulat~,~ to dryness.
Toluene was added and it was e.alJUlat~ to dryness. The residue
was dissohed in ~t .--`~ and heated at 70C. A solution of
ethyl chloro-~ t~t.' (7.67g) in ~ ~: ...., l . il~ was added and
the mixture was stirred and heated at 70C for 1 hour. The mixture
was cûoled and h~llu.,~l.,lic acid (2M) was added. It was stirred for
0.25 hours then extracted with ethyl acetate, washed with brine,
dried (anhydrous MgSO4) and filtered. The filtrate was eva~Jul~l~,d
wo 95/31446 2 1 9 0 ~ O ~ PCTÆP9510]8~3
- 25 -
to dryness and the residue was purified by ~ ulllaLu~la~lly on silica
eluted with cyclohexane. The main product was dissolved in
di~ lull~Lllallc and washed with aqueous sodium hydroxide (2M),
brine, dried (anhydrous MgSO4) and filtered to give ethyl 5-
S y~lu~i~l q (2-1.. ~"l.yll,~ )isoxazole-3-~all.uAyla~e (8.41g) as a
brown gum, NMR (CDCl3) 1.15(t,3H), 1.2(m,2H), 1.3(m,2H),
2.35(r41H), 2.55(s,3H), 4.05(q,2H), 7.2-7.4(r44H).
Rli.F~RFl~'F F~Mpl,F 10
Potassium t-butoAide (5.89g) was added to a solution of
N-methyl~ ly~ (6.0g) in N,N-dil--~l-y1 r. " .,.~.,. rl~
The mixture was stirred for 0.75 hours and methyl 4-bromo-2-
blu~ul~lc~lylbenzoate (15.4g) was added. It was stirred for 0.25
hours then poured ontû a mixture of ice and brine. It was treated
with aqueous r ' chloride solution and eAtracted with ethyl
acetate, washed with water, dried (anhydrous MgSO4) and filtered.
The filtrate was cva~ula~,d and the residue was re-crystallised from
a mixture of ethyl acetate and n-hexane tû give methyl 4-bromo-2-
(N-methyl-N-methylsul~uLvu~' ~I)benzoate (15.33g) as a
white solid, m p. 105.6-107.2UC
By 1~ u~.,cliu~l!, in a similar manner the fûllowing ~ v~
were prepared from the al/~lu~ t~ d starting materials.
methyl 4-chloro-2-fluoro-3-(N-methyl-N-
.u~,L~ h~,..,' ', :)benzoate, NMR (CDC13) 2.75(s,3H),
2.95(s,3H), 3.95(s,3H), 4.55(s,2H), 7.85(d,1H), 7.9(t,1H);
methyl 2-(N-methyl-N-l..~,Lh~' Il.l.~...,' ~ .,1)-4
Lli[luulu,..~LJ ~ ~va~e~ mp. 92.2-9A6C;
methyl 3,4-difluoro-2-(N-methyl-N-
methylsul~ u.. yl ~ .... Ih,:)berlzoate, as a brown gum, NMR(D6-
DMSO) 2.62(s,3H), 2.95(s,3H), 3.86(s,3H), 4.62(s,2H), 7.6(m,2H);
methyl 4-chloro-3-rduoro-2-(N-methyl-N-
methylsuL~Jllo.l.~l-".;.... ll~yl)benzoate, m.p. 80-81.8C;
methyl 4-chloro-2-(N-methyl-N-
ul~ h.. ll~JllvllJ~. l;l,.. :I,~I)benzoate m.p. 92-95C;
methyl 2-(N-methyl-N-uu~ Jl ,;" "". ll,yl)benzoate
as an orange oil, NMR (CDC13) 2.82(s,3H), 2.92(s,3H), 3.88(s,3H),
WO95131446 ` 2t sooal PCrlEP95/01813
-26-
7.35(m,1H), 7.55(m,1H), 7.68(m,1H), 7~95(m,1H);
methyl 4-fluoro-2-(N-methyl-N-
methylsul~ u-lyl~l":~ lyl)benzoate, NMR (CDC13) 2~81(s,3H),
2~,88(s,3H), 3~85(s,3H), 4~71(s,2H), 6~96(m,1H), 7~38(m,1H),
5 7.95(m,1H)-
12F.FFUl;.NCE FX~VIPI F. 11
Sodium ll~`u~ 2g) was addcd to a stirred solution
of methyl 4-chloro-2-fluoro-3-(N-methyl-N-
u~,Lllyl~ul~llu.l~ I)benzoate (5~0g) in tetrahydrofuran
and the mixture was stirred at room t~ Lulc overnight~ It was
washed with water, dried (anhydrous MgSO4) and filtered~ The
filtrate was evaporated to dryness to give methyl 4-chloro-3-(N-
methyl-N-ul~,.l.,~.~u.,ul.u.. ,~l-,,.;.. ~ ::.JI)-2-methylsulphenyl-
benzoate (4.0g) as a yellow gum, NMR (CDC13) 2~4(s,3H),
2.7(s,3H), 3.0(s,3H), 3.95(s,3H), 4.9(s,2H), 7.45(s,2H).
RFFE~FNCE FX~MPI.F 17.
A mixture of methyl 4-chloro-2-fluoro-3-ul~,.i.~lL..,..~u~.Lc (63g)
and N-l,.. ~ (63g) in carbon tetrachloride was stirred
and heated at reflux for 4 hours. The cooled solution was washed
with water, dried (arlhydrous MgSO4) arld filtered. The filtrate was
Lv~uldt~ to dryûess to give methyl 3-LIu~u~ -4-chloro-2-
[luul~L (87g) as a red oil NMR (CDC13) 3.95 (s,3H),
4.65(s,2H), 7.25(d,1H), 7.9(t,1H).
By plù e~ iri a similar marmer methyl 2-blulllulll~uyl-4-
~liLL.~lulll~,.h~lL..,.~Luale, NMR (CDC13) 3~92(s,3H), 4~9(s,2H),
7~65(d,1H), 7~75(s,1H), 9.1(d,1H) was prepared from the
a,u~lU~l;dt~ h~tit--t~d starting material. In this case the reaction
was il I ~.di~t~d with UV light irlstead of heating to reflux~
Similarly prepared were the following c~.. ~l..~..,..l~
methyl 2 ~1UIl.~ ,LIIJ' 3,4--lia~JIubc~uaLe as an orange oil,
NMR (CDC13) 3.95(s,3H), 5.03(s,2H), 7.2(m,1H), 7.73(m,1H);
methyl 2-bluluul..~,Ll.yl 1 chloro-3-llL~..ùl; ~.~u~L~, m~p~ 91-
93C;
methyl 2-L I ulllulll~ 4-~l~lul u~ Lu~Le as a brown oil, NMR
-
WO~S/31446 2 I q (~ F~.l/r.l~ . 16l3
- 27 -
(CDC13) 3.95(s,3H), 4.9(s,2H), 7.35(m,1H), 7.6(m,1H), 7.95(m,1H);
methyl 2-b~u~l~u~ yl4-Auvlul/~,l~udlc as a pale brown oil,
NMR (CDC13) 3.9(s,311), 4.9(s,2H), 7.02(m,2H), 7.95(m,1H).
S RF.FF.RF.I~ . F.XAMPr.F. 1'~
A mixture of methyl 2-(N-methyl-N-..~ ulyllu~
lyl)-4-~linU~lUlll~ lh~llLUdl~ (4.27g) and aqueous
sûdium hydroxide (2M) in methanol was stirred and heated at reflux
for 20 minutes. The cooled solution was poured into ice and
acidified with Lrd-u~ lolic acid. The solid was filtered off to give 2-
(N-methyl-N-methylsul,ul.u.,~'~,.,;.. ,. Ihrl)-4
I~iAuu~ulu~ u;c acid (4.03g) as a beige solid, m.p. 178.6-
180C
The following ~ were similarly prepared:
3,4-difluoro-2-(N-methyl-N-
'i Jl -~ JI)benzoic acid as a brown solid,
NMR(D6-DMSO)2.67(s,3H), 2.95(s,3H), 4.19(s,2H), 7.5(m,1H),
7.65(m,1H);
4-chloro-3-fluoro-2-(N-methyl-N-
methylsul,uhc,..... ~' ' JI)berlzoic acid as a white solid, m.p.
150-160.5C;
4-chloro-2-(N-methyl-N :' ,' 'j ' ~I-~,;,,I~,;I)benzoic
acid as a cream solid, m p. 205-205.5C;
2-(N-methyl-N ~ I ",;,Ihyl)benzoic acid,
m p. 158-159C;
4-fluoro-2-(N-methyl-N-methylsullul.u..~'-..;...~.., :I.yl)benzoic
acid, m.p. 186-1925C (dec.).
RF.li'F~F.l~('F. FX~MP~ F. 14
~`- .. ,. _ . I . .. I ~ d sulphuric acid (8 ml) was added to a solution of3,4-difluoro-2-u.~ll.ylb~l.,uic acid (43 g) in methanol, and stirred for
16 hours under refluL The cooled mixture was . ~ ,. ,- - ,1. ,. l . d to half
volume in vacuo, water added, and the remaining methanol
e~d~uu.dt~d. The mixture was extracted (ethyl acetate) and the
extract washed (sodium carbonate solution then with brine), dried
c ' ~ sulphate) and c ~ a~ul dt~,d to give methyl 3,4-difluoro-2-
wo 95/31446 ~ 1 9 o ~ o ~ PCT/EP95/01813
-28-
ylb~l~udlc (34 g) as an ûrange solid, m.p. 44~6C.
The following C,UIII~JUUlld:~ were similarly prepared:
methyl 4-chloro-2-~ .,,u~le, NMR (CDC13) 2.5(s,3H),
3.8(s,3H), 7.1~(m,2H), 7.88(d,1H);
methyl 4-fluoro-2--1l.,.l.~ll,.,.lLu~lc, NMR (CDC13) 2.6(s,3H),
3.88(s,3H), 6.9(m,2H), 7.92(m,1H).
E2F~l'FNCE F.XAMP~ F. l.C
A stirred solution of 3,4-d;Lluûlul/~.~uic acid (Slg) in dry
tetrall, ~1 urul~ was treated at -75C under an inert ~lllos~ c
with a solution of n-buL~" ' in hexanes (139 ml of 2.5M
solution) during 1 hour. After 1 hour, an additional 139 ml of the n-
butyl lithium solution was added. After 20 hour, methyl iodide (50
ml) was added during 30 minutes and the mixture allowed to slowly
warm to room ~ c overnight, then poured onto ice and
saturated sodium bi~.l,u..~t~, solution and stirred for 30 minutes.
The mixture was washed with ether, saturated with sodium chloride
and acidified with }.~JI. "( ~ acid. This was extracted (ethyl
acetate) and the extract washed ~brine), dried ( ~,
sulphate) and c~.,u, ' to give 3,4-difluoro-2--ll~.. ;.. ~lb~.~ uic acid
(25.1g), m.p. 152.5-153.5C
By ,UI U~ ,dillg in a similar manner 4-chloro-3-fluoro-2-
Ill~,lll~lb~.~u;c acid, m.p. 198-199C was prepared.
~F.~ F.N('F F.XAMPI.F. lC
Potassium carbonate (82.9g) was added to a stirred solution of
4-chloro-3-fluoro-2-u.~,Lll.~ ~u;c acid (112g) in dry acetone. After
1 hour methyl iodide (92.1g) was added and the mixture stirred
overnight under reflux. The cooled mixture was filtered, washed
with acetone and the solvent ~. ~,u, ' The residual oil was
dissolved in ether, washed (water), dried ( ~ ~ sulphate) and
ulaLc~ to give methyl 4-chloro-3-fluoro-2-lll.,Lh.~L~.,Lu
(116.8g) as a pale yellow oil, NMR (CDC13) 2.53(d,3H), 3.90(s,3H),
7.25(m,1H), 7.63(m,1H).
WO 95/31446 2 1 ~ O O O ~ PCT/EP95/01813
-29 -
UTILIlY
According to a feature of the present invention, there is
provided a method for controlling the growth of weeds (i.e.
undesired vegetation) at a locus which comprises applying to the
locus a herbicidally effective amount of at least one isoxazole
derivative of formula (I) or an agrir lltllr~lly acceptable salt thereof.
For this purpose, the isoxazole derivatives are normally used in the
form of herbicidal ~ e~ in A~o~ l with ~
diluents or carriers and/or surface active agents suitable for use in
herbicidal .. ,.~ ), for example as h~ drt~r described.
The ~ of formula (I) show herbicidal activity against
dicotyledonous (i.e. broad-leafed) and I-ul.~cul~.cdul.uu~ (i.e.
grass) weeds by pre- and/or post ~ ...u~,C Al~L~ ilJ~
By the term "pre~ .~n~,c ~ Alillll" is meant application
to the soil in which the weed seeds or seedlings are present before
e.~ ..., e of the weeds above the surface of the soil. By the term
"post ~ is meant -;-,u' ~ lll to the aerial or
exposed portions of the weeds which have emerged above the
surface of the soil.
For example, the ~ u .l~u -1~ of formula (1) may be used to
control the growth of:
broad-leafed weeds, for example, ~h-ltil~n ll.f .
~mor~nth-lc retrofl~lnlc Ri~1~nc pilnco (~ lyl~ hllm
(~olillm ~orin~ rynmfl~A s~ e.g. [pon~ ~ pll~Fllrea~i~
~a~a sinq~ic ~rV~ncic ~rllAmlm n~nlm and~a~hil~m
~lIUI~ and
grass weeds, for example Aln~ mlc ~v ~ rif c Avena
~a. D~~tArio ~A l ~ A I ;~ Echin~l hlrlA crl lc--e..Ili ~ ~m
bicolor, Fl~llcin~ in-lica and r~tAriq c~ e g r~tArio fo~l-rii or
~i~tArio viri~lic and
sedges, for example, CS per~-c f crlll~nt~lc
The amounts of ~. . .I.u~ of formula (I) applied vary with
the nature of the weeds, the ~ used, the time of
Allllli. Al ;~11 the climatic and edaphic conditiorls and (when used to
control the growth of weeds in crop-growing areas) the nature of the
crops. When applied to a crop-growing area, the rate of arplir:lti~n
WO 95/31446 2 1 9 ~ PCT/EP95/01813
-30-
should be sufficient to control the growth of weeds without causing
sllh~t~nti~l p~,.lll~lllCl.l damage to the crop. In general. taking these
factors into account, application rates between O.Olkg and Skg of
active material per hectare give good results. However, it is to be
S ~lntl~rctnod that higher or lower "~ rates may be used,
depending upon the particular problem of weed control
U ulllel cd.
The colllluuulul~ of formula (I) may be used to control
select*ely the growth of weeds, for example to control the growth of
those species h~ b~ful~ m~nti--nP~ by pre- or post e.-le~ ce
~ iUll in a directional or non-directional fashion, e.g. by
directional or non-directional spraying, to a locus of weed
infestation which is an area used, or to be used, for growing crops,
for example cereals, e.g. wheat, barley, oats, maize and rice, soya
beans, field and dwarf beans, peas, lucerne, cotton, peanuts, flax,
onions, carrots, cabbage, oilseed rape, sunflower, sugar beet, and
p. . . "~ . ,. .l or sown grassland before or after sowing of the crop or
before or after e..l~ ,..cc of the crop. For the selective control of
weeds at a locus of weed infestation which is an area used, or to be
used, for growing of crops, e.g. tbe crops L ~-hlb~fol~i m~ntinn~
~rplir--~ n rates between O.Olkg and 4.0kg, and preferably between
O.Olkg and 2.0kg, of active material per hectare are particularly
suitable.
The ~ u ~ of formula (I) may also be used to control the
growtb of weeds, especially tbose indicated above, by pre- or post-
C.ll~,.L~ rrlir~t in ~ h- d orchards and other tree-
growing areas, for example forests, woods and parks, and
, e.g. sugar cane, oil palrn and rubber ~ , For
this purpose tbey may be applied in a directional or non- directional
fashion (e.g. by directional or non-directional spraying) to the weeds
or to the soil in which they are expected to appear, before or after
planting of the trees or p~-"~ at ~ uli~ ~;.." rates between
0.25kg and 5.0kg, and preferably between 0.5kg and 4.0kg of active
material per hectare.
The .~ .u~ of formula (I) may also be used to control the
growth of weeds, especially those indicated above, at loci which are
.. , .... . _ _ . . . . ... . ... .. ..... . _ . _ . _
WO 95/31446 ;;~ ~ ~ o ~ ~ ~ PCTIEP95~01813
-31 -
not crop-growing areas but in which the control of weeds is
nevertheless desirable.
Examples of such non-crop-growing areas include airfields,
industrial sites, railways, roadside verges, the verges of rivers,
irrigation and other waterways, scrublands and fallow or
uncultivated land, in particular where it is desired to control the
growth of weeds in order to reduce fire risks. When used for such
purposes in which a total herbicidal effect is frequently desired, the
active ~ u.~ are normally applied at dosage rates higher than
those used in crop-growing areas as hcic;~ fulc described. The
precise dosage will depend upon the nature of the vegetation
treated and the effect sought.
Pre- or post ~ ,e ~ and preferably pre-
. '- ~,~ - - dlJ~ iUll~ in a directional or non-directional fashion
(e.g. by directional or non-directional spraying) at ~ ." rates
between l.Okg and 20.0kg, and preferably between 5.0 and lO.Okg,
of active material per hectare are particularly suitable for this
purpose.
When used to control the growth of weeds by prc-e.ll~ c~
~ the ~ of formula (l) may be ill~ull~u.~ into
the soil in which the weeds are expected to emerge. It will be
c-~J,ulc~;dted that when the ~ . ' of formula (I) are used to
control the growth of weeds by post ell.~ lKc ~l~u~ l i.e. by
1 ;.... to the aerial or exposed portions of emerged weeds, the
~ .,,.1.~, ,,,,l~ of formula (I) will also normally come into contact with
the soil and may also then exercise a pre e..l.,. ~ CC control on
later ~. .,,;,.~1;,.~ weeds in the soil.
Where especially prolonged weed control is required, the
application of the ~..,.I.u.,..,l~ of formula (I) may be repeated if
required.
According to a further feature of the present invention~ there
are provided ~ r~ C suitable for herbicidal use ~
one or more of the isoxazole derivatives of formula (1), in
~CCOriotion with, and preferably ~ b~ l,y dispersed in, one or
more ~ c a~ri~ltl-r~ly- acceptable diluents or carriers
and/or surface active agents [i.e. diluents or carriers and/or surface
WO 95/31446 2 1 9 0 0 0 7 PCTIEP95101813
-32- ~
active agents of the type generally accepted in the art as being
suitable for use in herbicidal ~U~ OaiLiu-ls and which are
~lmpqtihko with ~ of formula (I)]. The term
"1"~",~ y dispersed~ is used to include ~ o~ in which
the culll,uuullda of formula (I) are dissolved in other ~;ulll,u~ La.
The term "herbicidal ~ J~;l ;""`" is used in a broad sense to
include not only ~ ~ which are ready for use as herbicides
but also .. ,..... , l .. l . ~ which muât be diluted before use. Preferably,
the ~ulll,uOSiLiulla contain from 0.05 to 90% by weight of one or
more ~..... l,.J.. l~ of formula (I).
The herbicidal ~-.,.l.r~;l ;....~ may contain both a diluent or
carrier and surface-active (e.g. wetting, dispersing, or emulsifying)
agent. Surface-active agents which may be present in herbicidal
~:ulll~u~iLiuua of the present invention may be of the ionic or non-
ionic types, for example ~ l- t- ~, yuat-,.ll~l~ ""~l-~'l-;"'
derivatives, products based on .~ s of ethylene oxide with
alkyl and polyaryl phenols, e.g. nonyl- or octyl-phenols, or carboxylic
acid esters of ~Jlluaulbi~ which have been rendered soluble by
. ;r~ of the free hydroxy groups by .. ",~ ;.... with
ethylene oxide, alkali and alkaline earth metal salts of sulphuric
acid esters and sulphonic acids such as dinonyl- and dioctyl-sodium
51111-1.... ...... ~t.r ~ and alkali and alkaline earth metal salts of high
molecular weight sulphonic acid derivatives such as sodium and
calcium li~nclllrh~qt~c and sodium and calcium alk~ll,.,. ~..c
clllrhnnq-tPs~
Suitably, the herbicidal ~ according to the present
invention may comprise up to 10% by weight, e.g. from 0.05% to
10% by weight, of su.L~e q~tive agent but, if desired, herbicidal
according to the present invention may comprise
higher u- upo. Liulla of aulL~c q~tive agent, for example up to 1~%
by weight in liquid rn~-~lcifiql ~o Cllcren~ . ,-t~ ~ and up to
25% by weight in liquid water soluble ~ t~,
Examples of suitable solid diluents ûr carriers are qlllminillm
silicate, talc, calcined magnesia, kieselguhr, tricalcium phosphate,
powdered cork, absorbent carbon black and clays such as kaolin and
bentonite. The solid ~ n~ I i-J ~ (which may take the form of
-
WO 95/31446 2 ~ ~ O ~ O 1 PCTIEP9~/01813
-33 -
dusts, granules or wettable powders) are preferably prepared by
grinding the ~ of formula (I) with solid diluents or by
iullyl U~ the solid diiuents or carriers with solutions of the
. ~.."l/.,,l~ of formula (I) in volatile solvents, ~aluOl~ the
S solvents and, if necessary, grinding the products so as to obtain
powders. Granular f~rrn~ tilmc may be prepared by absorbing the
lu ~ of formula (I) (dissolved in suitable solventsl which may
if desired, be volatile) onto the solid diluents or carriers in granular
form and, if desired, ~v~ul~ the solvents, or by ~ ulula~ill~,
.ulll~u~iliùlLc in powder form obtained as described above. Solid
herbicidal ~ UI~llly wettable powders and
granules, may contain wetting or dispersing agents (for example of
the types described above), which may also, when solid, serve as
diluents or carriers.
Liquid ~ according to the invention may take the
form of aqueous, organic ûr aqueous-organic solutions, ~ nc
and emulsions which may h.~ul~ulrle a ~uuL~,e - ~, agent.
Suitable liquid diluents for i~.ul~uul~iull in the liquid ~ ;h
include water, glycols, hL-~h, L urU. rU.yl alcohol, ~' ~l r' ~
~l ' '- ~l isu~ullulullc~ toluene, xylenel mineral, animal and
vegetable oils and light aromatic and, ~I.h l....'r fractions of
petroleum (and mixtures of these diluents). Surface-active agents,
which may be present in the liquid ~ ul"~ may be ionic or
non-ionic (for example of the types described above) and may, when
liquid, also serve as diluents or carriers.
Powders, dispersible granules and liquid ~ in the
form of ~ may be diluted with water or other suitable
diluents, for example mineral or Yegetable oils, rua~ ul~u ly in the
case of liquid l l lll~ . ll . ,,t~; in which the diluent or carrier is an oil,
to give ~ ready for use.
When desired, liquid ~ of the compound of
formula (I) may be used in the form of self elU~;ryi lg ~ . . I I l ..t~ .~
containing the active substances dissolved in the ~luulsiryill~ agents
or in solvents containing ~I..ul~iryi~ agents ~ r with the
active cllhst~r~c, the simple addition of water to such conC~ u.,
producing ~ ready for use.
.. . . . . . . .
WO 9S/31446 ~ PCTIEP95101813
-34-
Liquid concentrates in which the diluent or carrier is an oil
may be used without further dilution using the cl~ ù~ tic spray
technique.
Herbicidal .U.~ o~iLiol~ according to the present invention
S may also contain, if desired, cull.. ,.. iùllàl adjuvants such as
adhesives, protective colloids, thickeners, ~cll~.la~ agents,
stabilisers, sP?~P~Prin~ agents, anti-caking agents, colouring agents
and corrosion inhibitors. These adjuvants may also serve as carriers
or diluents.
Unless other~vise specified, the following l,~ lLa~s are by
weight. Preferred herbicidal comrnCiti()nc according to the present
invention are:
aqueous SllcrPncir~ , which comprise from 10 to
70% af one or more ~..,.,I.u.,,..l~ of formula (I), from 2 to 10% of
surface-active agent, from 0.1 to 5% of thickener and from 15 to
87.9% of water;
wettable powders which comprise from 10 to 90% of one or
more ~ ù . .l.., ,- 1~ of formula (I), from 2 to 10% of surface-active
agent and from 8 to 88% of solid di.uent or carrier;
water soluble or water dispersible powders which comprise
from 10 to 90% of one or more .u ~l~u~ of formu.a (I), from 2 to
40% of sodium carbonate and from 0 to 88% of solid diluent;
liquid water soluble .u ''`f 1~ C which comprise from S to
50%, e.g. 10 to 30%, of one or more, ,,u~..d~ of formula (1),
from 5 to 25% of bUlra~ ive agent and from 25 to 90%, e.g. 45 to
85%,ofwatermisciblesolvent,e.g. ~ h~yl~ P ora
mixture of water-miscible solvent and water;
~.iquid ~ r ' cù~...,.4~lat~,j which comprise
from 10 to 70% of one or more l~,,,,,l, ,,-l~ of formul.a (I), from 5 to
15% of ~u.r~ agent, from 0.1 to 5% of thickener ar.d from
10 to 84.9% of organic solvent;
granules which comprise from 1 to 90%, e.g. 2 to 10% of one
or more c~ of formula (I), from 0.5 to 7%, e.g. 0.5 to 2%,
of surface-active agent and from 3 to 98.5%, e.g. 88 to 97.5%, of
granular carrier and
Pnnlllcifiol~lP cùl~C~ lat~ which comprise 0.05 to 90%, and
.
WO 95/31446 ~ PCT/EP95101813
-35 -
-
preferably from 1 to 60% of one or more ~:UIIIIJUUI-d~ of formula (I),
from 0.01 to 10%, and preferably from 1 to 10%, of surface-active
agent and from 9.99 to 99.94%, and prefeMbly from 39 to 98.99%,
of organic solvent.
Herbicidal ~ according to the present invention
may also comprise the ~;CIIII~UUII~ of formula (I) in ~ccr~ri:ltit)n with,
and preferably h-...,..~ dispersed in, one or more other
pesticidally active c~ u ~ and, if desired, one or more
~nmr~tihl~ pesticidally acceptable diluents or carriers, surface-
active agents and ~ull._llLiu~al adjuvants as h~ L.. ,EuiG described.
Examples of other pesticidally active ~(.. "l.u.. l~ which may be
included in, or used in ~.. j .. ; ;.-ll with, the herbicidal .. ,l.~
of the present invention include herbicides, for example to increase
the range of weed species controlled for example alachlor [2-chloro-
2,6'-diethyl-N-(methoxy-methyl) ~ l l ], atrazine [2-chloro-4-
~ih~lalliulo-6-isu~lul)~' -1,3,5-triazine], IJIUIIIU~IIII [3,5-
dibromo 1 ~ -u~L '-], ~' ' i ' u~.[N'-(3-chloro-4-
ujl)-N~N-diul.,;l~l~u~ ]~ cyanazine [2-chloro-4-(1-cyano-
1- .ll.,~I~,lh,' -) 6 1,l1l~1a... ll~-1,3,5-triazine], 2,4-D [2,4-
2û A~ rtir acid], dicamba [3,6-dichloro-2-
u~ - acid], .l;f .~ [1,2- dimethyl-3,5-diphenyl-
Lu~ salts], flall.~Jlu~ l [methyl N-2-(N- benzoyl-3-
chloro-4-lluùlùa~ r ' ' ], r.~ ulull [N'-(3-trifluoro-
-I.,llljll~ll~,l.~l)-N~N-~ Lh~ d]~ isu~ulu~ull [N'-(4-
25 isu,u.u~l~ )-N,N-di~.,;Ljlu.. ~.], ;.. l;- A- ~, e.g. synthetic
uld~, e.g. F ~ and ~,~ .. ,l,. i.., and fungicides, e.g.
~all,~Ul.àt~,S, e.g. methyl N-(1-butyl-carbamoyl- L ' ' 2-
yl)~alballlalG~ and triazoles e.g. 1-(4-chloro-phenoxy)-3,3- dimethyl-
1-(1,2,4-triazol-1-yl)-butan-2-one.
Pesticidally active ~ u~ and other biologically active
materials which may be included in, or used in . , with, the
herbicidal ~ r ~ of the present invention, for example those
h.,l ~,h-L ~f~.- G m~nti- nP~I and which are acids, may, if desired, be
utilized in the form of ~;ull~l.;iol~al derivatives, for example alkali
metal and amine salts and esters.
According to a further feature of the present invention there is
... . . . ... . . ... . . .... . . ..
WO 95/31446 ~ I q (3 ~ ~ ~ PCT/EP95/01813
_
36
provided an article of m ~nllf~rtllre comprising at least one of the
isoxazole derivatives of formula (I) or, as is preferred, a herbicidal
~ o~ as h~, ~;lL~ulc described, and preferably a herbicidal
COllCc~lLlaLc which must be diluted before use, ~,UIII~Jli~lill~ at least
one of the isoxazole derivatives of formula (I) within a container for
the aforesaid derivative or derivatives of forrnula (I), or a said
herbicidal ÇUIII~U~iLiUII, and instructions physically associated with
the aforesaid container setting out the maMer in which the
aforesaid derivative or derivatives of formula (I) or herbicidal
~u~ o~iL;u~ contained therein is to be used to control the growth of
weeds. The containers will normally be of the types conventionally
used for the storage of chemical substances which are solid at
normal ambient ;~ r ~ ,,, and herbicidal çullllJu~ilio~
particularly in the form of ço,~ , for example cans and drums
of metal, which may be internally lacquered, and plastics materials,
bûttles ûr glass and plastics materials and, when the contents of the
container is a solid, for example granular, herbicidal ÇUlll~U~ilio,~,
boxes, for example ûf cardboard, plastics materials and metal, or
sacks. The corltainers will norrnally be of sufficient capacity to
contain amounts of the isoxazole derivative or herbicidal
~ mr~iti-~n~ sufficient to treat at least one acre of ground to
control the growth of weeds therein but will not exceed a size which
is ,UII~ .. for .,OI~ tiullal methods of handling. The
instructions will be physically associated with the container, for
example by being printed directly thereon or on a label or tag
affixed thereto. The directions will normally indicau that the
contents of the container, after dilution if necessary, are to be
applied to control the growth of weeds at rates Of l ,lul i~
between O.Olkg and 20kg of active material per hectare in the
30 manner and for the purposes L~,.. ,;llb~,'ulé described.
The following Examples illustrate herbicidal ~ ;o~
according to the present invention:
E~MPLF. Cl
35 A soluble ~on~ llldLe is formed from:
Active ingredient (compound 1) 20% w/v
, .... _ ...... _ .. ... ......... .... . . . _ . .
WO 9S131446 2 ~ 9 () L~ O 1 PCTIEP9~101813
-37-
Potassium hydroxide solution 33% w/v 10% v/v
Tetral.r.llu~u.~u.~l alcohol (THFA) 10% v/v
Water to 100 vûlumes~
by stirring THFA, active ingredient (compound 1) and 90%
volume of water and slowly adding the potassium hydroxide solution
until a steady pH 7-8 is obtained t~hen making up to volume with
water.
Similar soluble c-,, . l .. l~c may be prepared as described
above by replacing the isoxazole (compourld 1) with other
. ~ l,.J .l~ of formula (I).
~XAMPLI~
A wettable powder is formed from:
Active ingredient (compound 1) 50% w/w
Sodiumdodc~ll,.,.lL~.-c~ ll.l.. ---t~ 3% w/w
15Sodium l;v~ 5% w/w
Sodiumr ''-~icall~l ~ l r~ 2% w/w
Microfine silicon dioxide 3% w/w and
China clay 37~o w/w
by blending the above ingredients together and grinding the
20mixture in an air jet mill.
Similar wettable powders may be prepared as descTibed above
by replacing the isoxazole (compound 1) with other ~.. l u .l~ of
formula (I).
F.~AMPI F C3
25A water soluble powder is formed from:
Active ingredient (compound 1) 50% w/w
Sodium dodc~lh. ~ . lrhrr^~^ 1% w/w
Microfine silicon dioxide 2% w/w
Sodium' ' 47% w/w
by mixing the above ingredients and grinding the above
mixture in a hammer mill.
Similar water soluble powders may be prepared as described
above by replacing the isoxazole (compound 1) with other
l.u~ of formula (I).
The ~ ., 1 ~, .1~ of the invention have been used in herbicidal
~plir^~ti~m~ according to the following ~ cd..-~.
21 so~al
wo 95/31446 PCT/EP95/01813
-38-
MFTHOD OF USE OF HERBICIDAL COMPOUNDS:
a) General
Appropriate quantities of the CUIII~JUUllt~:~ used to treat the
plants were dissolved in acetone to give solutions equivalent to
~rrlir:ltinrl rates of up to 4000g test compound per hectare (g/ha).
These solutions were applied from a standard laboratory herbicide
sprayer delivering the equivalent of 290 litres of spray fluid per
hectare.
b) Weed rnntrol: Prc~ . .,. P
10 The seed3 were sown in 70 mm square, 75 mm deep plastic
pots in non-sterile soil . The quantities of seed per pot were as
follows:-
Weed ~crPriP~ Avvrox n~lmhPr of seeds/vot
1) Broad-lP~fP~wpp~lc
Abutilon ~- ''l'k'''`l; 10
Am~r nth-lc retroflexus 20
Galium aparine 10
Ipomoea purpurea 10 .
Sinapis arvensis . 15
Xanthium~LI~..... l~.liulll 2
2) Gr~cc w~P~ic
Alopecurus Ill~J~ulo;d~;, 15
Avena fatua 10
Frhir- ' '~~ crus-galli 15
Setaria viridis 20
3)~ç5!y~,c
Cyperus esculentus 3
1) Broad-lP:~fP~l
Cotton 3
Soya 3
2) ~a~c
Maize 2
Rice 6
Wheat 6
WO95/31446 ~ ~aO(:1l PC'rJEP95~0~813
-39 -
The ~ v~ of the invention were applied to the soil
surface, containing the seeds, as described in (a). A single pot of
each crop and each weed was allocated to each treatment, with
unsprayed controls and controls sprayed with acetone alone.
After treatment the pots were placed on capillary matting kept
hn a glass house, and watered overhead . Visual ~c.~ccnn~ nt of crop
damage was made 20-24 days after spraying. The results were
expressed as the p~ reduction in growth or damage to the
crop or weeds, in ~ with the plants hn the control pots.
c) Weed control: Post ~ e
The weeds and crops were Sown directly into John Innes
potting compost in 75 mm deep, 70 mm square pots except for
Amaranthus which was pricked out at the seedhing stage and
f.,..~ to the pots one week before spraying. The plants were
then grown in the ~ ' until ready for spraying with the
u .-l~ used to treat the plants. The number of plants per pot
were as follows :-
1) Broad l~f~rl w~ c
Weed 5rr~ c Nllmh~ r of ~t~ntC per ~ot Growth C~
20Abuthon~ l.. ;.. t; 3 1-21eaves
Am~nthllc retroflexus 4 1-2 leaves
Galium aparine 3 15t whorl
Ipomoea ptlrpurea 3 1-2 leaves
Shnapis arvensis 4 2 leaves
Xanthium :~llUlLlaliULU 1 2-3 leaves
2) Gr~cc w~ ~-ic
Weed svecies N~lmh~r of pl~nt~ per ~ot Growth ,ct:~,r~
Alopecurus ~h~J~u-~lidcs 8-12 1-2 leaves
Avena fatua 12-18 1-2 leaves
Frhinrl~hlr~a crus-galli 4 2-3 leaves
Setaria viridis 15-25 1-2 leaves.
3) Sed~es
Weed ~c~ c NllmhPr nf ~nl~ntc ~ner pot Growth ct:~e
Cyperus esculentus 3 3 leaves.
WO 95/31446 2 1 9 ~ ~ PCT/l~P95~01813
-40-
1) Broad l~f~
Crops Nllmh~r of vl~ntc per vot Growth
Cotton 2 1 leaf
Soya 2 2 leaves.
2) Grass
Crovs Nllmh~r of ~Iqnt~ per vot Growth Ct:tSt'
Maize 2 2-3 leaves
Rice 4 2-3 leaves
Wheat S 2-3 leaves.
The .. l,u ~ used to treat the plants were applied to the
plants as described in (a). A single pot of each crop and weed
species was allocated to each treatment, with unsprayed controls
and controls sprayed with acetone alone.
After treatment the pots were placed on capillary matting in a
glass house, and watered overhead once after 24 hours and then by
controlled sub-irrigation. Visual: ~ of crop damage and
weed control was made 20-24 days after spraying. The results were
expressed as the y~ reduction in growth or damage to the
crop or weeds, in ~ . with the plants in the control pots.
The ~"..~ of the invention, used at 1 kg/ha or less, have
shown an excellent level of herbicidal activity together with crop
tolerance on the weeds used in the foregoing ~
When applied pre- or post ~ at lOOOg/ha or less
~l .. ,l.. ,.. - l~ 1 to 10 gave at least 905~ reduction in growth of one or
more of the weed species.
When applied post ~ at 250g/ha or less ~ulllyuuud~ 1
to 10 gave at least 80% reduction in the growth of Ipomoea
~rQa and Am~r~qnthl~c retr-lfl~ ~n.c