Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
2~ 2$~
AQUEOUS COMPOSITION
BACKGROUND OF THE INVENTION
1. Field of the Invention
The present invention relates to an aqueous composition,
and more particularly to an aqueous composition which is
useful as an aqueous lubricant, or an aqueous coolant, and is
transparent in appearance.
2. Description of the Related Arts
Various aqueous lubricants have heretofore been known.
Japanese Patent Publication No. 6991/1985, for instance,
discloses an aqueous lubricant which is transparent in
appearance, and has a high phase-inversion temperature and a
high phase-reversibility.
However, the aqueous lubricant described in the above
patent publication has a disadvantage in that the viscosity
becomes high when it is in the form of transparent aqueous
solution. Accordingly, in order to obtain a composition
having a comparatively low viscosity, the lubricating base oil
as the base fluid must be so low in viscosity that the
lubricity cannot be sufficiently high. In addition, the
aqueous lubricant described in the above publication is
lacking for mechanical and chemical stabilities.
SUMMARY OF THE INVENTION
An object of the present invention is to provide an
aqueous composition being transparent in appearance, and
having a phase reversibility and a low viscosity, to which a
base fluid having a high viscosity can be applied.
20 ~026~ ~
Another obiect of the present inventlon is to
provide an aqueous composition as above, having mechanical and
chemical stabilities.
The present invention provides, an aqueous
composltlon which is formed by dispersing water into a
lubricating base oil as a base fluid in the presence of a
surfactant, wherein the surfactant comprises:
(A) at least one compound represented by the
general formula
Rl-o-~R2-o) -H ,................... (I~
wherein Rl is an alkyl group having 3 to 10 carbon atoms, R
is an alkylene group having 2 or 3 carbon atoms, and n is an
integer of 1 to 20, and when n is an integer of 2 or more, an
alkylene group having 2 carbon atoms and an alkylene group
having 3 carbon atoms may be mixed in the repeating unit
(R2-o), and
(B) at least one ionic surfactant,
wherein the composition contains 1 to 80% by weight
of water.
The present invention also provides an aqueous
composition comprlslng the component (A), the component (B)
and (C) at least one nonionic surfactant other than the
cornponent (A).
BRIEF DESCRIPTION OF THE DRAWINGS
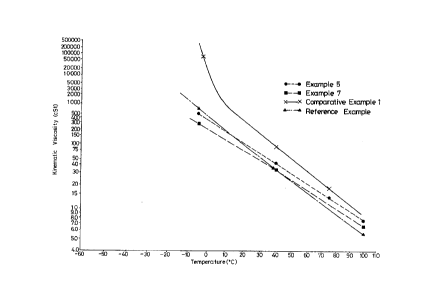
Fig. 1 shows the relations between viscosity and
temperature of the aqueous composition in Examples 5 and 7,
Reference Exarnple, and Cornparative Example 1.
-- 2
73162-26
2 û 2 0 2 6 6
DESCRIPTION OF PREFERRED EM~ODIMENTS
The aqueous compositlon of the present invention is
formed by dispersing water into a base fluid in the presence
of a surfactant. As the base fluid, various lubricating base
oils can be used, as long as they are in llquid or semisolid
form at ordinary temperature and have a melting point of 40~C
or lower. Preferable base fluids are water-insoluble organic
compounds liquid at ordinary temperature, and having a
kinematic viscosity at 40~C of 1 to 1000 cSt, and the most
preferable ones among them are lubricating base oils having a
kinematic viscosity at 40~C of 2 to 500 cSt. The lubricating
base oil may be a mineral oil or a synthetic oil, and may
contain, if necessary, various additives lncluding an oiliness
agent, an extreme-pressure agent, and an antioxidant. The
composition of the present invention contalns the above base
fluid in an amount of usually 90% or less by weight,
preferably 5 to 70% by weight.
The surfactant to be used in the present invention
is described as follows.
In the present invention, a compound represented by
the general formula (I):
R1 o (R2 o) H ~--(I)
is used as component (A). Therein, R1 is an alkyl group
having 3 to 10 carbon atorns, including a propyl group, a butyl
group, a pentyl group, a hexyl group, a heptyl group, an octyl
group, a nonyl group, and a decyl group.
When R is an alkyl group having 2 or less carbon
-- 3
73162-26
2 6 ~
atoms, the compound of the general formula ( I ) shows no
ernulsifying ability, and when R is an alkyl group having 11
or more
- 3a -
73162- 26
A
20~2/~2G~
carbon atoms, the viscosity rises largely. Accordingly, both
the cases are not suitable to the object of the present
invention. n is an integer of 1 to 20, preferably 1 to 10,
more preferably 1 to 5. R2 is an alkylene group having 2 or 3
carbon atoms, and more specifically, an ethylene group or a
propylene group. However, when n is an integer of 2 or more,
both R having 2 carbon atoms (ethylene group) and R2 having 3
carbon atoms (propylene group) may exist in the repeating unit
(R -O). Among the compounds represented by the general
formula (I), the most preferred one is a compound wherein R
is an alkyl group having 4 to 9 carbon atoms, R2 is an
alkylene group having 2 carbon atoms, and n is 1 to 5.
The compounds represented by the general formula (I) are
preferably those having a hydrophilic-lipophilic balance (HLB)
value of 3 to 15, more preferably 5 to 12. Specific examples
of these compounds of general formula (I) are monobutylether,
monohexylether, mono-n-octylether and mono-2-ethyl-hexylether
of monoethylene glycol; monobutylether, monohexylether, mono-
n-octylether and mono-2-ethyl-hexylether of diethylene glycol;
monobutylether, monooctylether and mono-2-ethyl-hexylether of
monopropylene glycol; monobutylether, monooctylether and mono-
2-ethyl-hexylether of dipropylene glycol; monobutyl ether,
monooctyl ether and mono-2-ethyl-hexylether of tripropylene
glycol; monobutyl ether, monooctyl ether and mono-2-ethyl-
hexylether of heptapropylene glycol; further, oxyethylene
mono-2-ethyl-hexylethers such as tetraoxyethylene mono-2-
ethyl-hexylether (n=4), and octaoxyethylene mono-2-ethyl-
hexylether (n=8).
2Q2Q26~
73162-26
In the aqueous composition of the present invention,
one or two kinds of the above compounds are used as component (A),
but when two kinds or more are blended, it is preferred to blend
the compounds whose HLB values are two or more apart from each
other (for example, to blend a compound having a HLB value of 6 or
less with a compound having a HLB value of 8 or more, or to blend
a compound having a HLB value of 9 or less with a compound having
a HLB value of 11 or more), or to use a compound having (C2H4O)
unit and a compound having (C3H6O) unit in combination as the
repeating unit (R -O).
The compounds represented by the general formula (I) are
blended in the aqueous composition usually in the amount of 2 to
50% by weight, preferably 5 to 40% by weight.
The aqueous composition of the present invention con-
tains further (B) ionic surfactant. Said ionic surfactant may be
any of anionic, cationic, and amphoteric surfactants. Specific
examples of them are carboxylic acids having 7 to 40 carbon atoms,
such as fatty acids having 7 to 30 carbon atoms, dimer acids of
said fatty acids, dicarboxylic acids having 2 to 36 carbon atoms,
metal (preferaly alkali metal) salts of said carboxylic acids or
alkanol amine salts of said carboxylic acids, metal (preferably
alkali metal), amine or ammonium salts of naphthenic acid, alkyl
(preferably having 10 to 22 carbon atoms) sulfates, alkyl (prefer-
ably having 10 to 22 carbon atoms) sulfonates, alkylaryl (prefer-
ably alkylphenyl having 6 to 14 carbon atoms in the alkyl moiety)
sulfonates, petroleum sulfonates, amino acids and the like. One
20~
- 73162-26
or more of them can be blended in the aqueous composition, and
anionic surfactants are particularly preferable.
Ionic surfactants are blended in the aqueous composi-
tion of the present invention in an amount of 2 to 30% by weight
- 5a -
:2~2Q~iG
usually, and preferably 3 to 20% by weight.
In the aqueous composition of the present invention,
moreover, (C) nonionic surfactants other than the component
(A) can be blended. Examples of the nonionic surfactants are
(i) fatty acid esters of polyhydric alcohols, (ii) oxyalkylene
ethers or polyoxyalkylene ethers and (iii) aliphatic alcohls.
Oxyalkylene ethers or polyoxyalkylene ethers include the
compounds wherein any of R and R is beyond the
abovedescribed range, among the compounds represented by the
general formula (I).
Specific examples of fatty acid esters of polyhydric
alcohols are ethyleneglycol monolaurate, diethyleneglycol
monolaurate, propyleneglycol monolaurate, propyleneglycol
monostearate, stearic acid monoglyceride, oleic acid
monoglyceride, sorbitan monostearate, sorbitan distearate,
sorbitan tristearate, sorbitan monooleate, sorbitan
sesquioleate, sorbitan dioleate, sorbitan trioleate, sorbitan
monopalmitate and the like.
As oxyalkylene ether or polyoxyalkylene ether, used are
the compounds represented by the general formula (II):
R3 O (R4 O) H ...(II)
wherein R3 is an alkyl group having 12 to 30 carbon atoms, a
phenyl group, an alkyl-substituted phenyl group having 7 to 30
carbon atoms, or a cycloalkyl group having 6 to 30 carbon
atoms, R is an alkylene group having 2 or 3 carbon atoms, and
m is an integer of 1 to 20. However, when m is 2 or more,
plural kinds of R s may exist in the repeating unit (R -O).
Specific examples of the compounds represented by the
2 6 B ~i
;eneral formula (II) are polyoxyethylene laurylether,
polyoxyethylene rnyristylether, polyoxyethylene cetylether,
polyoxyethylene stearylether, polyoxyethylene
nonylphenylether, and the like. Specific examples of the
compourlds other than the compounds of general formula (II) are
polyoxyethylene butylether, hexylether, 2-ethylhexylether,
nonylether, ethyleneglycol nonylether, decylether, and the
like.
Further, the aliphatic alcohols therein are preferably
those having 12 to 30 carbon atoms, such as lauryl alcohol,
myristyl alcohol, cetyl alcohol and stearyl alcohol.
Nonionic surfactants as component (C) as above are not
necessarily added, but if necessary, it is added usually in an
amount of 2 to 30~ by weight in the aqueous composition.
The aqueous composition of the present invention is
formed by dispersing water into base fluid in the presence of
a surfactant as mentioned above, and can contain generally 1
to 80% by weight, preferably 3 to 50~ by weight of water.
In the a~ueous composition of the present invention,
additives such as water-soluble rust-preventives, antifleezing
fluid, and the like can be added, if necessary, to the
abovementioned components.
As water-soluble rust-preventives therein, conventional
ones such as water-soluble rust preventives of nltrogen-
containing organic compound, and water-soluble rust
preventives of inorganic compound can be used optionally.
73162-26
2D~2~6
Examples of them are alkyl amines such as tri-n-butyiamine,
and cyclohexyl amine; alkanol amines such as mono-(di- or
tri-) ethanol amine, mono-(di- or tri-) propanol amine, n-
butyldiethanol amine, diethyldiethanol amine, N-
methyldiethanol amine, N-dibutyldiethanol amine and the like;
amine compounds such as pyperazine, hydroxyethylpyperazine,
morpholine and the like; above alkyl amine salts of carboxylic
acids such as fatty acid having 6 to 20 carbon atoms, aromatic
carboxylic acids, dibasic acids having 2 to 20 carbon atoms;
above alkanol amine salts or ammonium salts; condensates of
various carboxylic acids as above and amines; and inorganic
salts such as sodium nitrite, cobalt nitrite, sodium carbonate
and the like.
As the antifleezing fluid, ethylene glycol, propylene
glycol and the like can be added, and thus the solidifying
point (that is, the lowest temperature for application) of the
aqueous composition can be lowered.
The aqueous composition of the present invention is
obtained by blending and stirring the components as above, and
formulated for lubricant, hydraulic fluid, coolant and so
forth to be used in metal working such as cutting, and
plasticizing, and thermal processing.
The working mechanism of the aqueous composition of the
present invention in use for various purposes is not clear,
but it is presumed that the molecule of surfactant forms
micelles on the interface with the molecule of the base fluid,
to be a protective colloid film.
In the present invention, a specific nonionic surfactant
20202~6
and an ionic surfactant and/or other nonionic surfactants are
used in combination, and thus it is attained to obtain a
transparent dispersing liquid containing a comparatively large
amount of water in the base fluid.
The aqueous composition of the present invention is
transparent in appearance, and has a low viscosity, and can
employ a fluid having a high viscosity in order to obtain a
composition having a desired viscosity. The aqueous
composition of the present invention is excellent in
mechanical and chemical stabilities, stability in storage, and
stability in circulation, and hardly changes with temperature.
Further, phase inversion by temperature or alteration of water
content is reversible in said composition, so it can easily
return to its original state.
Consequently, the aqueous composition of the present
invention is suited to be aqueous lubricant such as metal
working fluid, and oily agent for equipments, aqueous thermal
medium, coolant and the like.
The present invention will be described in more detail
with reference to the following Examples and Comparative
Examples.
EXAMPLE 1 TO 9, COMPARATIVE EXAMPLES 1 TO 3, AND REFERENCE
EXAMPLE
An aqueous composition was prepared by blending the
components shown in Table 1 below in the proportions (by
weight) shown in Table 1.
As the rust preventive, morpholine was used.
on the compositions of Examples 5 and 7, Reference
;2û~D2~
Example and Comparative Example l, the relation between
viscosity and temperature were shown in Fig. 1.
Further, various properties of the resulting aqueous
composition were determined according to the following methods
and the results are shown in Tables 2 and 3.
(a) Kinematic Viscosity and Viscosity Index
Determined according to JIS K 2283.
(b) Clouding Point
Determined according to JIS K 2269.
(c) Shearing by Ultrasonic Wave
Determined according to MIL L H5606A, under the condition
of 28 ~m and 60 minutes.
(d) Heat Test
Heated at 200~C for 36 hours sealed in an autoclave.
(e) Appearance
The sample was placed immediately into a glass vessel,
and observed for its state at 20~C at ordinary pressure.
If the sample is dissolved, the solution becomes transparent
and homogeneous.
(f) Temperature of Phase Inversion to Emulsion
A hundred milliliters of sample was placed in 200 ml-
beaker and heated with stirring from ordinary temperature to
100~C for about 5 minutes. State of the sample therein was
observed. The temperature at which the sample becomes cloudy
emulsion was taken as the phase inversion temperature. The
phase inversion temperature is more favorable, the higher it
is .
(g) State of the Process of Moisture Evaporation
-- 10 -
2 ~ 6
A hundred milliliters of sample was placed into 200 ml-
beaker, heated with stirring to 90 to 100~C, and its state
after 30 minutes were observed. Stirring was performed at 200
rpm. The composition was evaluated by judging whether it was
"liquid" or "solidified".
(h) Reversibility of Phase Inversion
It was judged whether the sample phase-inverted by the
operation (f) or (g) could return to the state before phase
inversion.
(1) After the test (g), water in the same amount as lost
in the evaporation was added to the sample, and stirred at 200
rpm at 20~C at ordinary pressure for 10 minutes, and tested if
it could return to the state just after it was produced.
The result was indicated as "reversible" when the
composition returned to the state, and "irreversible" when it
did not.
(2) After the test (f), the sample was heated to 100~C,
and then left to cool with stirring at 200 rpm. In the case
where the phase inversion temperature had been determined by
the operation (f), if it returned to the state before phase
inversion around the temperature (+10~C) to be similar in
appearance at 20~C to that of (e), it was indicated as
"reversible", while if not, it was indicated as
"irreversible". When the phase inversion temperature had not
been determined by the operation (f), the sample in cooling
was observed in the same manner. When no phase inversion was
observed in cooling, and the appearance on returning to 20~C
was similar to that of (e), it was judged as "reversible", and
202Q266
if not, as "irreversible".
(i) Stability in Storage: Appearance of Test Piece
Into 200 ml-glass vessel (with stopper) having inner
diameter of 25 mm and height of 150 mm, 150 ml of test sample
was placed. In the said sample, a test piece made of SPCC
(cold-stretched steel plate) with a thickness of 1 mm, width
of 20 mm, and length of 120 mm was placed. The vessel was
closed with the stopper, and left for 6 months at a constant
temperature of 20~C, and after that, the rust on the surface
of test piece was judged. The result was indicated as
"unchanged" when no rust was formed, and "rust" when rust was
formed.
(j) Stability in Storage: Appearance of the fluid
After the test (i), the sample was observed for its
state. The sample composition was observed whether it was
transparent and homogeneous.
(k) Stability in Circulation
A circulation test by gear pump for one month was
performed, and then the tests (e) to (h) as above were carried
out. If the sample was in the same state as that just after
production, the result was indicated as "good", and if not,
indicated as "no good". The test was carried out at tank
temperature of 50~C, with a capacity of 20 liters, at a flow
rate of pump of 4 liters/min., and the water content was
supplemented by an automatic water supplying apparatus to keep
a fixed value.
(1) Anti-wear Property Test
A pump test was carried out at 140 kg/cm2 at 50~C for 250
2~2~
hours according to ASTM D 2882.
-- 13 --
CA 02020266 1998-04-22
~ d' '
Ul I ~ I I I ~ I 1 0
a) .
E ~ ~
~ .
Ln .. ~
I CO I I o~ I I I ~ I
o
~ .
cnu~
V U
o
.. ~ .. U~
V
o o o
o o . o ~ ~
k
u u
3 ~t a ~ c ~
O -- ~ ~ ~ ~ ~ ~ ~ cn O
Q - -~ U ~ U ~ U
E 1 O-rl o ,l o-,l ~ ~ ~ ~ ~ ~ u
-- ~ Ul U
Q ~ ~ ~ O Ul ~
~ 2 2 2 2 - ~ ~ v
-~ o o
U V
14
73162-26
CA 02020266 1998-04-22
~ - o
o tL
r oo
*t~
~ ~ Ln *
o o~ I I ~ ~t~
t~
tL'
O p~r~ ~ I I I
~ In co I ~t~ ~
tL) ~:1
Q tL~ o
o
Q ~
t'l - O ~ ~ I ~ I I I
r ~ u~ I U
,
~ o
t~ ~ I ~t~
O
V V tLl o
~ ~ o
o
t~
~: tLI ~
u~ t~2
V
o .
o~ t~ ~
U U V
o o o
. o o o
Il 11 11 .
tL ~L tL ~' ~~' '~'''
O tL~tL~ tLI O O O O
-rl ~ ~ ~ -rl V V V
L~ (L
L 1 L
O ~ r r O ~
Q _ _ _ tL~ Q ~ -~ V -~ V -~ ~ tL' tL
~ ~ r~ 1 O ~ O ~ O-~
O ~ ~ ~ rl O _ ~ ~ ._~
~_) t~ ~ V ., ~1 (1~ - J ) X
O O O ~ Q ~ k - ~ tL~
~ tLI ~ tL' ~ tL'
_ I I I > O tV ~ tL~ ~ tL) ~
0 0 0 ~ ~ 1 0 0
_ ~ tL
~ tn ~ ~ ~ .
o ~ r -~ O
73162 -26
CA 02020266 1998-04-22
o ~ o
. :~
X O o
0 Lr) I U
o o 0 o
'
' ~ _l
a,) o o
Ln I ~ ~ In 0
a
O
o U~ 0 ~
U X
,~
O O ~'
u o a) . a) 'l
~)~, . ... ..... ,
,Q 4~ X , _,
F 11 11 11
~L ~V C ~ V ~ IV
~V ~ ~L ~V (L
r ~ r ~ V
a, - ~ I ~ I ,v ~ (V
O r~ r~ LI (V (L r~ ~ ~
- ~1 ~ ~1 0 J~ ~V r-l r-l r-l
~1 lV ,xv r-l a) ~ _ _ _ IXv r-l 4 1
O r-l r~ U~ O ~ n~v
~ J~ ~ ~ ~- rl
v ~v (V a~ ~v r V
~ .r~l U~ O ~0
~ ~ ~ ~ V ~ ~ V
O O L ~V O ~ (L O O O ~ O O
U C P~ ~
V
O O
U U ~ ~ * -rl
15a
73162 -26
CA 02020266 1998-04-22
o ~ d~
o\~~ o\~ O *
r ~~ o ~ ~ ~ r~ o~ o ~ ,~ ~
A
~ ~ O
O ~~ ~Ln ~O o\~ O o\~ O ~ ~C
~r ~ I ~-- ~ A p~
~ ~ o ~ ~r
~ ~o ~ ~ or O\o r oP , o ~ ~
In d1 ~ ~ ~~ ~ d~ O ~ O
U~ ~ ~ I --' ~ ~ A p~
~1
o r~
~- ~Du~ r o co ~ O\o o *
X ~~ _ A
~ ~ .
~1: 0
In ~ 0 ~ Ln ~ ~ o * - 1C
Ln r ~ I A ~
O
Ln O
A
O
0 ~0 0 ~ o
~ ~ I
V ~ ~ I O
O u~
O O O t~ ~ L) o ~ n
o Ln o -- ~ a
~I' I ~ ~ U ~ I n ~ 1
V -- -I ~ >
o
J-) ~ ~ O a) v a) I > H
-- ~ O ~1 I H 0
O ~ ~ un
un ~~
un un .5::
un ,~ ~ U ~
o un: un un un 3 u ~ U!~ P~ o ~) W
O O O ~ O C 3 C v -- -I o
k o o v a
o un u~ un ~ ~ un o u. ~ u o o
~ n c ~ 5~ un ~ C
u ~ r ~ u o
O
U O
X
15b
73162 -26
CA 02020266 1998-04-22
' d~ OO ~C) 1~1 d1 ~r o\~ ~ o\~
O (V ~ ~ ~~ ~ I ~~ O~D O
r- O ~1 ~c~
tJ ~
X ' ~ ~
~ ~ ~n ~~ o\o~ o\o
O CO d~ O ~r o
I I O ~
t~ ~
0\o ~
lV o co o\o
O ~ ~ 1-- 0 ~ ~ ~ ~CO ~ OD
Ul O ~ ~a~
X
~V I I ~
o ~
V O O~ t' ~~ o\o O
O ~ ~ LO o ~ t~ o ~ u
( ~ ~ tl O
L L
L r
o o\o .o\~
L L ~ ~ O O ~ ~ O ~ ~ r
O ~ ~ ~ o ~~-- o ~ a~
O o oo ~ ~ ~-
V
O
L' ~ I I O
O - t~ 'V
V I I C) (V
t~ I I V ~ ~D O ~o o Ln
Fl: O V ~ [' ul I .,
~ - O ~ ~
~V
P~
L~
V J ~
V V o U~
(V o o o U~ ~
U OLn O ~r CQ
V d~ I ~1 ~ U: U
~ V ~
Ql ~ ~ ~ o ~ ~ ~
L' ~ ~ ~ o (V V (V
~ -rl O Cq U~ Cq d' ~ ~
L1 U~ L~ U U U '- ~ O
O /V ~1 ~ O '~
~ ~ ~ ~ I~ /V ~ U
U~ ~ O o ~ Ul -~
O O S~ O Ul U~ U~ UUl ~ U~
Q ~ ~l (V -~ Q O O O ~ O O 3 O V
Ul ~_ U U E~ U ~ ) o\~ U 'U O
O L L, O Ul ~ U ~ L, U. o Ul U Ul o
V ~ ~ ~ L, ~ -~ ~ -~ L -~ -- ~ -~-~ -~ ~
O ~ ~ L, ~ '--
O
V ~ U U U ~U L U Ul U ~
~ Q~ Q~ -~ Ul
-~ Q~ Q~ L~ ~ L
~~ -- 1~ k ~ c ~ E~
Q ~ ~ (V o ~l _ (V i ~V ~ ~V
L' L' ~ u O L1 L L' L, t~
V ~ ~l ~ Q
15c
73162 -26
CA 02020266 1998-04-22
o ~ ~ ~
o * * O
a~ o
A 1~ ~1
O ~ ~ I I ~
O * * ' O
~1 ~ a) I I o
A ~ ~)
~ ~ .
o ~ ~ I)I) ~
O * * ~ ~ O
a~ ~ - o
A . p~ tJl
, ~ r
o ~ .
o * ~ O
a) I ~ o
A
O ~ ~ ~
¢ O * * O
- ~ ~ a) o
G A
a
a
'
~ .
.
¢
E~ ~
o
-
~
O ~ n
a) ,
¢
3H
r~ O
H Oa~ G) ~
¢l -r~, J.J
a.) u~ 1)
¢n ¢!,~
V ~ ~ rn ~ rJ
o Cl ~ ~ a) ~
S~ O ~ ~ ~-~1
rn r~
O
c a) ~ ~ o o
~) ~ ¢~ c ~ a) a~
n
r 1-- S- S- ~ t~ >
a) a
a) ~ oJ~ aa,
k ~ ~ ~ >
a) ~ ~ > lJ
rn 1) ~ r~ rl~ * *
15d
73162 -26
20~2~G
- Table 3
Wear Example Example Comparative Commercial Reference
amount 4 5 Example 1 ProductExample
Water-Glycol
Cam ring213 28 570 976 198
(mg)
Vane 11 5 13 28 8
(mg)
Total 223 33 583 1004 206
(mg)
The results given in the above tables show the following
facts.
(i) The aqueous composition of the present invention has a
lower kinematic viscosity than those of the conventional ones
(kinematic viscosity at -5~C, 40~C of Comparative Example 1),
even if a base fluid having the same kinematic viscosity.
Particularly, as shown in Examples 6 and 7, when plural
compounds of general formula (I) are added, the viscosity of
the aqueous composition is desirably lowered. In the
conventional composition, on the other hand, even if the
kinematic viscosity of the base fluid is lowered, the
resulting kinematic viscosity becomes not so low (see
Comparative Examples 2 and 3).
(ii) The aqueous composition of the present invention has a
high viscosity index. As seen from Fig. 1, the lines
representing the kinematic viscosities of the compositions in
Examples 5 and 7 show small inclinations on the graph, and are
on the side indicating lower viscosity. That means, the
composition of the present invention has a low viscosity, and
is small in change with temperature. On the other hand, in
the compositions of Comparative Examples 1 to 3 and Reference
- 16 -
2 o~
Example, the kinematic viscosities vary with temperature
largely, and the viscosities rise remarkably in the range of
lower temperature.
(iii) The composition of the present invention shows little
change in viscosity after being sheared and after heat test
(see Examples 4 to 7, and Comparative Examples 1 to 3).
(iv) In the composition of the present invention, wear amount
resulting in anti-wear properties test by pump is small.
Particularly, the composition of Example 5 is far superior to
the mineral base oil (see Reference Example) in anti-wear
properties.
(v) In the composition of the present invention, it is
possible to obtain an aqueous solution being low in viscosity
and stable, if the amount of water b~ended is 30% by weight or
40~ by weight (see Examples 8 and 9).