Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
CA 0224076~ 1998-06-10
-2-
Background of the Invention
Field of the Invention
The invention relates to a process for removing sulfur dioxide from flue
gasses by scrubbing with a scrubbing solution.
5 Discussion of the Prior Art
It is known that, in flue gas purification plants or flue gas desulfurization
plants, the sulfur dioxide present in the flue gasses can be reacted either
directly with calcium hydroxide or indirectly by means of a scrubbing solution
with addition of sodium hydroxide and subsequent reaction with calcium
10 hydroxide to form gypsum.
A disadvantage of these known processes is that the gypsum obtained,
for example in a waste incineration plant, contains impurities and is therefore
a useless product. Since the product cannot be utilized, it additionally incurs
costs for disposal. The gypsum has to be taken to a landfill which, apart from
15 the high disposal costs, additionally leads to environmental pollution.
Summary of the Invention
It is an object of the present invention to provide a process for flue gas
purification or for flue gas desulfurization which does not require landfilling and
does not cause environmental pollution.
The process of the invention for flue gas desulfurization uses a solution
comprising potassium hydroxide for scrubbing the flue gas to remove the sulfur
dioxide .
An advantage of the process of the invention is that the sulfur dioxide
present in the flue gas reacts with the potassium hydroxide present in the
CA 0224076~ l998-06-lO
scrubbing solution to form potassium sulfate which is a marketable, saleable
product which can be utilized, in particular as fertilizer.
The flue gas comprising sulfur dioxide is scrubbed with a scrubbing
solution which is maintained at a pH of from 4 to 7, preferably a pH of from
5 5 to 6. 5, by addition of potassium hydroxide. The following reactions
presumably occur during scrubbing:
a) KOH + SO2 KHSO3
b) 2 KOH + SO2 K2SO3 + H20
C) K2SO3 + 1/2 ~2 K2SO4
The potassium sulfate can be sold as a marketable product as solution
or in crystalline form, for example as a fertilizer.
The process of the invention is particularly suitable for purifying flue
gasses from waste, sludge or sewage sludge incineration plants.
If the flue gas has a low ~2 content, the reaction c) proceeds slowly. In
15 an advantageous embodiment of the invention, oxygen, for example in the
form of compressed air, is introduced into the scrubbing solution, in particularwith formation of turbulence, which significantly accelerates the reaction c).
In another embodiment of the process, trace elements, such as trace
metals which may be present in the scrubbing solution in addition to the
20 potassium sulfate, are precipitated completely or specifically in such a way that
the fertilizer also contains trace elements in a sufficient concentration. The
concentration can be selected so that the fertilizer contains trace elements in
a concentration preferred for fertilization or for the fertilization purpose.
The various features of novelty which characterize the invention are
25 pointed out with particularity in the claims annexed to and forming a part ofthe disclosure. For a better understanding of the invention, its operating
CA 0224076~ 1998-06-10
advantages, and specific objects attained by its use, reference should be had
to the drawing and descriptive matter in which there are illustrated and
described preferred embodiments of the invention.
Brief Description of the Drawings
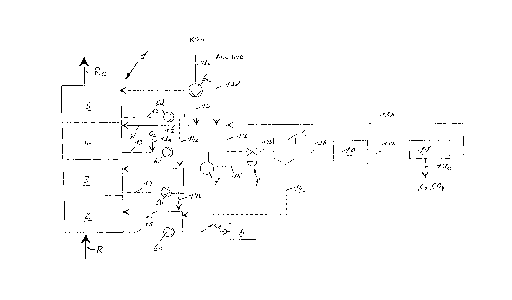
5 Fig. 1 schematically shows an apparatus for removing sulfur dioxide
from flue gasses of a waste incineration plant pursuant to the
inventive process.
Detailed Description of the Preferred Embodiments
A flue gas R from a combustion chamber (not shown) in which waste
10 is incinerated is fed to the flue gas purification apparatus 1 also referred to as
a flue gas scrubber. After flowing through the flow gas purification apparatus
1 the gas is discharged to the environment as purified gas Re. In order to
absorb the hydrogen halides (HCI, HF, HBr) present in the flue gasses R
separately from sulfur dioxide S02 and to saturate the flue gas R with water
15 vapour, the flue gas R is first fed to a first scrubbing stage, viz. a quench stage
2, which removes from about 80% to 90% of the hydrogen halides present in
the flue gas R. The quench stage 2 has a water circuit 15 and a circulation
pump 6a. A substream of the quenching water is conveyed via a line 1 5a and
valves not shown to a wastewater treatment plant A. A second scrubbing
20 stage arranged downstream is configured as a packed stage 3 and serves to
improve the saturation of the flue gas R and to remove hydrogen halides
remaining in the flue gas R after passing through the quench stage 2 so that
the purified gas Re and the fertilizer produced have a low halide content. The
packed stage 3 has a liquid circuit 14 and a circulation pump 6b. The liquid
25 circuit 14 is connected to the liquid circuit 15 by a connection 14b.
The packed stage 3 is followed by a further packed stage 4 to remove
sulfur dioxide. Downstream of the packed stage 4 there is arranged a further
scrubbing stage, configured as a fine dust removal stage 5, for example
CA 0224076~ 1998-06-10
comprising a ring jet stage. This fine dust removal stage 5 is provided for
removal of very fine dust and aerosols and likewise has a liquid circuit 12 witha circulation pump 6d. The liquid circuit 12 is connected to the liquid circuit
14 via the line 1 4c. If removal of very fine dust is achieved by an upstream or5 downstream cloth filter, the ring jet stage can be omitted.
Potassium hydroxide (KOH) is metered into the scrubbing solution W
which is used in the packed stage 4 to remove sulfur dioxide and is conveyed
around a liquid circuit 13 by means of a circulation pump 6c. The potassium
hydroxide is stored in a stock container (not shown) and is conveyed by a
10 pump 6e through the feed line 1 3c to the liquid circuit 13. In an advantageous
embodiment, the pH of the scrubbing solution W is measured using a sensor
and this signal is used to control the pump 6e so as to keep the pH of the
scrubbing solution W at a pH of from 4 to 7 by means of the addition of
potassium hydroxide.
The flue gas R from a waste incineration plant has, for example, an
oxygen excess of from 10% to 1 1%. If the waste incineration plant has flue
gas recirculation, the oxygen excess in the flue gas R is reduced, for example,
to from 6% to 7%, which leads to a high proportion of potassium sulfite
(K2SO3) in the scrubbing solution W. In order to reduce the content of
20 potassium sulfite and to increase the content of potassium sulfate, oxygen, for
example in the form of compressed air, is advantageously introduced via a line
1 3a into the scrubbing solution W after leaving the packed stage 4, for
example by generating turbulence in the scrubbing solution W immediately
upstream of the circulation pump 6c so as to significantly accelerate the
25 above-mentioned reaction c).
During scrubbing, the scrubbing solution W usually also takes up traces
of heavy metals such as Hg, Cd, Zn or Pb. To obtain a high-quality potassium
sulfate, it is found to be advantageous to remove these trace metals and also
further trace metals, either partially or completely. This is achieved by addition
30 of a sulfur-containing precipitant which is fed via a feed line 1 3d into the liquid
CA 0224076~ 1998-06-10
circuit 13 in order to precipitate sparingly soluble compounds. Suitable
precipitants are, for example, trimercaptotriazine or Nalmet~. As a further
additive, activated carbon or open-hearth furnace coke can be added in
aqueous suspension to achieve the following effects:
5 - to remove organic trace impurities originating from the flue gas, for
example chlorinated phenols, benzenes, dioxins or furans,
- to remove metallic mercury which can be formed by reduction of
mercury compounds, e.g. HgCI2, by residual sulfite concentrations,
- as a filtration aid in the downstream filtration located in feed line 1 3b.
The concentration of potassium sulfate in the scrubbing solution W is
preferably kept at a constant high value by measuring the concentration in the
scrubbing solution W by means of a conductivity measuring apparatus 7 and
using this value, via an electrodiode connection 15, as a setting parameter for
the regulating valve 8, so that a regulated amount of scrubbing solution W is
15 taken from the circuit 13. The concentration is advantageously regulated suchthat the scrubbing solution W has a potassium sulfate concentration of at most
15%. A filtration apparatus 9 is located downstream of the regulating valve 8
and serves to remove solid constituents such as impurities and additives from
the scrubbing solution W. The filtration apparatus 9 is preferably configured as20 a candle filter with membrane-coated filter cloths. The solids collect in theconical bottom of the filtration apparatus 9 and are periodically taken off a
sludge via the line 1 4a. The sludge can be recirculated to the liquid circuit 15
of the quench stage 2.
The purified scrubbing solution W is passed to an evaporation or
25 crystallization unit 10 arranged downstream of the filtration apparatus 9. Inthis unit 10, the potassium sulfate concentration is increased to above 20%.
The potassium sulfate which crystallizes is separated off in a downstream
decanter 1 1 and discharged as end product via the outlet 1 3e. The remaining
solution is recirculated via the line 1 3b to the liquid circuit 13. If the potassium
30 sulfate is required only in the form of a solution, crystallization of the
CA 0224076~ 1998-06-10
-7-
potassium sulfate can be omitted by concentrating the scrubbing solution W
to, for example, 15% by evaporation and then selling it as solution.
The invention is not limited by the embodiments described above which
are presented as examples only but can be modified in various ways within the
5 scope of protection defined by the appended patent claims.