Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
1
PACKAGE FOR APPLYING A PERSONAL CARE PRODUCT
FIELD OF THE INVENTION
The present invention is directed to an improved package for applying a liquid
personal
care product to the skin of a user.
BACKGROUND OF THE INVENTION
Liquid personal care products are available in a wide variety of packages,
including
bottles, jars, tubes, and cans. Few liquid personal care products on the
market have built-in
applicators wherein the product flows through the applicator. Some products on
the market, such
as certain deodorants, employ a roller-ball applicator technique to deliver
product. However,
other personal care products such as cosmetics foundations and lotions are
traditionally dispensed
out of a container into the hand or onto a sponge, and then applied to the
skin. This application
method however can be unsanitary, wasteful, and messy, and may lead to
undesirable post-
application hand washing.
Conventional applicators are typically not suitable for desirably applying
cosmetic
foundations or lotions to the entire face. For example, at least some
conventional applicators are
ergonomically designed to apply product onto larger areas of the face such as
the cheeks and
forehead, but not onto smaller skin areas such as the area between the lips
and nose. But
ergonomic packages may not provide intuitive applicator positioning commonly
associated with
cylindrical containers and/or applicators. On the other hand, cylinder-shaped
applicators may not
provide a suitable means to apply product to the smaller areas of the face.
Accordingly, it would be desirable to provide an intuitive package with skin-
friendly
dispensing that can apply a suitable amount of product directly onto the skin.
It would also be
desirable to configure such a package to better fit into a user's hand. It
would further be desirable
to provide an applicator that delivers a suitable amount of product to the
skin with each
movement of the applicator.
SUMMARY OF THE INVENTION
In order to provide a solution to the aforementioned problems, a package for
applying a
personal care product to the skin of a user is disclosed herein. The package
comprises a body for
storing and dispensing the personal care product. The body has a first cross-
sectional shape and
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
2
is graspable by the user's hand. The package also comprises an applicator
joined to the body. The
applicator includes a cylinder-shaped skin-contacting portion. The skin-
contacting portion has a
second cross-sectional shape and a surface pattern. Each of the first cross-
sectional shape and the
second cross-sectional shape has a major dimension oriented along a first axis
and a minor
dimension oriented along a second axis. The major dimensions are greater than
the minor
dimensions.
BRIEF DESCRIPTION OF THE DRAWINGS
While the specification concludes with claims particularly pointing out and
distinctly
claiming the present invention, it is believed the same will be better
understood from the
following description taken in conjunction with the accompanying drawings in
which:
FIG. 1 is a perspective view of an embodiment of a package.
FIG. 2 is a perspective view of the bottom portion of the package shown in
FIG. 1.
FIG. 3a is a front elevation view of the embodiment shown in FIG. 1.
FIG. 3b is a side elevation view of the embodiment shown in FIG. 1.
FIG. 3c is a front elevation view of an embodiment of a package.
FIGS. 4a-g are examples of cross-sectional shapes..
FIG. 5 is a front elevation view of the bottom portion of the package shown in
FIG. 1.
FIG. 6 is a front elevation view of an embodiment of a package.
FIGS. 7A-R are examples of patterns suitable for the skin-contacting portion.
FIGS. 8a-d are examples of patterns and zones on the skin-contacting portion
FIGS. 9a-d are further exemplary embodiments of a package for use herein.
DETAILED DESCRIPTION OF THE INVENTION
The improved package disclosed herein may be understood more readily by
reference to
the following detailed description of illustrative and preferred embodiments.
It is to be
understood that the scope of the claims is not limited to the specific
ingredients, methods,
conditions, devices, or parameters described herein, and that the terminology
used herein is not
intended to be limiting of the claimed invention. Also, as used in the
specification, including the
appended claims, the singular forms "a," "an," and "the" include the plural,
and reference to a
particular numerical value includes at least that particular value, unless the
context clearly
dictates otherwise.
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
3
All percentages and ratios used herein are by weight of the total composition,
and all
measurements made are at 25 C, unless otherwise designated.
Packages suitable for use herein include a body portion with personal care
product
disposed therein. In certain embodiments, the personal care product is in the
form a liquid or
liquid-like substance. Flexible, resilient packages are known in the art and
are typically
deformable by applying pressure to the body of the package (e.g., by
squeezing), which urges a
volume of personal care product to an applicator or skin-contacting portion.
In certain
embodiments, the package may include a one-way valve to enable a user to
control the flow of
the personal care product. It is to be appreciated that the packages herein
may include additional
or alternative mechanisms for dispensing the personal care product.
Nonlimiting examples of
known dispensing mechanisms include electronic pumps; manual pumps; screw-
driven pistons or
rods; pressurized gasses such aerosols and the like; and trigger pumps.
In certain embodiments, the package may include an applicator for receiving
personal care
product dispensed from the body of the package and applying it to the skin of
a user. All or a
portion of the applicator may be fixed or movable (e.g., pivotable, rotatable,
detachable, and/or
reattachable). The applicator includes a skin-contacting portion for desirably
transferring the
personal care product to the skin of a user. The skin-contacting portion may
be fixed or movable
depending on the desired use. The skin-contacting portion is configured to
apply the personal
care product onto the skin in a desirable manner (e.g., evenly, smoothly,
and/or natural looking).
In an exemplary embodiment, the skin-contacting portion of the applicator may
include a roller,
which applies the personal care product while rotating about an axis. In this
example, the skin-
contacting portion receives the personal care product on its surface (e.g.,
passively via a discrete
reservoir which is positioned in close proximity to the roller such that the
personal care
composition comes into contact with the surface of the roller as the roller
rotates; through
apertures in the surface of the roller fed by a channel in liquid
communication with the body;
and/or by actively urging the personal care composition directly onto the
surface of the roller
from the body) and, by rotation, deposits the personal care product onto the
skin. In another
exemplary embodiment, the applicator may include a dosing system wherein the
personal care
product is dosed through the center of a fixed skin-contacting portion and
exits apertures
(whether through a porous foam or sponge skin-contacting portion, or via a
solid skin-contacting
portion with holes fed through an axle).
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
4
The skin-contacting portion may includes a surface with a pattern thereon. The
pattern
may be formed by one or more of the same or different surface features. The
surface features
may be the same or different size, type (e.g., protrusion versus recess versus
aperture), or shape
(e.g., elliptical, circular, diamond, square). The surface features may be
selected to configured to
facilitate the transfer and/or application of the personal care product onto
the skin of a user.
Particularly suitable examples of surface features for use herein are
described in more detail
below.
The package may be configured for dual-ended or single-ended dispensing. Dual-
ended
dispensing applicators may be suitable for circumstances in which a small skin-
contacting portion
on one end and a larger skin-contacting portion on the other end is desired.
Dual-ended dispens,
it may be desirable to provide packages that include two or more personal-care
products (e.g., a
cosmetic primer and a foundation or a skin cleaner and a skin moisturizer)
which can be
dispensed through dual-ended dispensing applicators. In certain embodiments,
the body of the
package may include a first end and a second end opposed thereto. The first
end may include an
applicator and associated skin-contacting portion, while the opposing second
end includes a
blending tool joined thereto. The blending tool may be used to blend,
distribute, or spread the
personal care product over the skin.
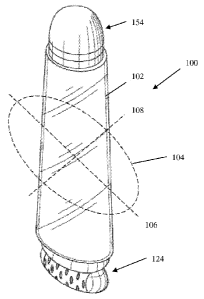
FIGS. 1-4 show an exemplary embodiment of a package 100 suitable for use
herein. As
shown in FIG.1, the package 100 includes a body 102 for receiving, storing
and/or dispensing a
personal care product 180 (e.g., skin care composition, cosmetic composition
such as primer or
foundation, and/or a combination of these). The body 102 of the package 100
includes a
reservoir or similar storage space storing the personal care product. The body
102 of the package
100 is configured to facilitate grasping with a user's hand, for example, by
having an ergonomic
shape. The body 102 of the package 100 has a cross-sectional shape 104 defined
at least in part
by a major dimension 106 oriented along a first axis and a co-planar minor
dimension 108
oriented along a second axis. While the shape of the cross-section of the body
102 shown in FIG.
1 appears substantially elliptical, it is to be understood that any suitable
cross-section shape may
be used as long as it is readily graspable by a user's hand. The body 102 of
the package 100 may
be formed from a suitable natural or synthetic material such as, for example,
high density
polyethylene ("HDPE"), low density polyethylene ("LDPE"), polyethylene
terephthalate
("PET"), polypropylene ("PP"), polyvinyl chloride, polycarbonate, nylon, and
fluorinated
ethylene propylene. In certain embodiments, it may be desirable for form the
body 102 from a
flexible or pliable material. The body 102 may be made via a number of
processes known in the
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
art, such as blow molding, injection molding, and the like. The body 102 may
be transparent,
translucent, or opaque, as desired. Alternatively or additionally, the body
102 may be formed
from a combination of such materials. For example, the body 102 may include a
transparent
portion to display a particular feature of the personal care product stored
therein. In certain
embodiments, it may be desirable to display a particular color, texture,
and/or pattern of the
personal care product 180 to provide a nonverbal cue to a potential consumer
that the product has
certain desirable features (e.g., white color to indicate sensitivity or swirl
pattern to indicate
multiple benefits).
The package 100 may include an applicator 124 and a blending tool 154. The
applicator
124 may be attached to body 102 by any suitable means known in the art (e.g.,
with screw-
threads, a snap-fit collar, or the like). In certain embodiments, the
applicator 124 may be integral
with body 102 (i.e., the applicator is assembled with the body during
manufacturing and is not
intended to be removed by a consumer during the product lifetime). In certain
embodiments, the
applicator 124 may be removably attached to body 102, (i.e., the applicator
may or may not be
assembled with the body during manufacturing and is intended to be removable
by a consumer
during the product lifetime without causing damage to the package which would
impair its
intended function), e.g., for cleaning or replacement with another applicator
124. In certain
embodiments, the applicator 124 may be removed to enable user to dispense bulk
product directly
from body 102. The applicator 124 may be fixed or pivotable, relative to body
102. If in a fixed
position, the package 100 desirably includes a suitable opening and closing
mechanism (e.g.,
lever, switch, or valve) to start and stop product dispensing. If the position
of the applicator 102
is not fixed, then the position of the applicator 124 may be changed to permit
a user to start and
stop the dispensing of product. For example, the applicator 102 may be
configured to pivot in a
plane parallel to the plane of the cross-section 104 such that when the
applicator 102 is pivoted a
particular amount (e.g., between 1 and 360 degrees or between 1 and 180
degrees), an opening is
provided through which the personal care product 180 may be dispensed. When
the applicator is
pivoted back to the previous position and/or when the applicator is pivoted
further (e.g., another 1
to 180 degrees) the opening is closed. Additionally or alternatively, the
applicator may be
configured to pivot to other positions to change the size and/or shape of the
opening, thereby
controlling the rate at which the personal care product is dispensed.
The package may include a one-way valve made from rubber or similar material,
which is
substantially impermeable to the personal care composition 180. The one-way
valve may be
positioned, for example, between the storage space in the body 102 and the
skin contacting
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
6
portion of the applicator 124. The one-way valve may be configured to be
opened by squeezing
the body 102 of the package 100, thereby peimitting the personal care product
180 to flow
through one-way valve, and closed when the squeezing pressure is removed.
As shown in FIG. 2, the applicator 124 includes a skin-contacting portion 126
having a
cross-sectional shape 128. The cross-sectional shape 128 of the skin-
contacting portion is at least
partially defined by a major dimension 130 oriented along a first axis and a
co-planar minor
dimension 132 oriented along a second axis. The applicator 102 may be
configured to control the
amount and spacing of applied product. For example, at least a portion of the
skin-contacting
portion 126 may be designed to fit between a user's nose and upper lip. As
such, the minor
dimension 132 of the skin-contacting portion 126 may be less than or equal to
1.9 cm or even
1.25 cm. Alternatively or additionally, the major dimension 130 of skin-
contacting portion 126
may be less than or equal to 3.8 cm. The skin-contacting portion 126 may be in
the shape of a
cylinder, sphere, or other suitable shape, as desired. Suitable materials for
the skin-contacting
portion 126 include rubber, sponge, felt, cloth, wicking fibers, flocking, and
combinations
thereof. For example, the skin-contacting portion may be configured as a
cylindrical roller
formed from a soft, pliable material such as a commonly known thermoplastic
elastomer
("TPE").
The package 100 may optionally include a blending tool 154 for manipulating
the
personal care composition after it has been applied to the skin (e.g.,
smoothing, spreading, and/or
blending the personal care composition with the same or different personal
care composition) .
The blending tool 154 may be attached to body 102 opposite the applicator 124
by any suitable
means known in the art, such as with screw-threads, a snap-fit collar, or the
like. The blending
tool 154 may be removably attached to body 102 so that a user can remove the
blending tool 154
to clean it or replace it with a new or different blending tool 154. In
certain embodiments, the
blending tool 154 may be removed to permit bulk dispensing of the personal
care product from
the package 100. The blending tool 154 may include a plurality of bristles, a
paintbrush, a
sponge, a loofah, a shower puff, a massager, or other like utensil suitable
for blending,
distributing, or spreading a liquid personal care product. For example, the
blending tool 154 may
be made from flexible and/or soft materials such as rubber, TPE, sponge,
combinations thereof
and the like. It may be desirable to form the blending tool from a material
that is rinsable with a
suitable solvent such as water, isopropyl alcohol, or acetone.
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
7
FIGS. 3a and 3b show the major dimension 106 and minor dimension 108 of the
body 102
substantially aligned with the respective major dimension 130 and minor
dimension 132 of the
skin-contacting portion 126. But in certain embodiments, such as shown in FIG.
3c, the major
dimension 106 of the body 102 substantially aligns with the minor dimension
132 of the skin-
contacting portion 126. FIGS. 1 and 2 depict the cross-sectional shape 104 of
the body 102 and
the cross-sectional shape 128 of the skin-contacting portion 126 aligned
vertically, but it is to be
appreciated that the cross-sectional shapes 104 and 128 need not necessarily
be in vertical
alignment. For example, the skin-contacting portion 126 may be offset from the
body 102 such
that the skin-contacting portion 126 is not positioned vertically above the
body 102, but rather
extends outwardly from the side of the body 102. The skin-contacting portion
126 may be fixed
at a particular angle relative to the longitudinal dimension of the body or it
may be adjustable.
The cross-sectional shapes 104 and/or 128 of the body 102 and/or the
applicator 124,
respectively, may take a variety of different shapes, as shown in FIGS. 4a-g.
The shapes 104 and
128 may be substantially the same or different. For example, the cross-
sectional shape 104 of the
body 102 may resemble an oval shape while the cross-sectional shape 128 of
skin-contacting
portion may be more of a teardrop shape. In certain embodiments, the package
100 may include
more than two distinct cross-sectional shapes.
FIG. 5 shows an exemplary embodiment of an applicator 124. As shown, the skin-
contacting portion 126 includes a first portion spaced away from the
applicator base 138 by a first
distance 134, and a second portion spaced away from the applicator base 138 by
a second
distance 136. It may be desirable to configure the first distance 134 to be
greater than the second
distance 136 or vice versa. In certain embodiments, the skin-contacting
portion 126 may include
three or more portions in various zones spaced away from the applicator base
138 by three
different distances. The applicator 124 may include two or more arms 140,
which extend
downwardly from the body 102 of the package 100. The arms 140 may be
configured to house
skin-contacting portion 126 therebetween, and join the skin-contacting portion
126 to other
portions of the package 100 (e.g., the body 102). The arms 140 may be rounded
to form a divot
as shown in FIG. 5, or may be flush with body 102, as shown in FIGS. 8a-8c.
The skin-
contacting portion 126 may be joined to one or both of the arms 140 via an
axle, one or more
pins, or any other known means suitable for joining a roller to two or more
arms. The arms 140
and, optionally, the axle or other joining means, may include a channel or the
like to place the
body 102 in liquid communication with the skin-contacting portion 126.
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
8
The skin-contacting portion 126 includes a pattern 144 on its surface. The
pattern 144
may be in the form of discontinuous dots, lines, and/or shapes; a continuous
matrix; and/or a
combination of these. The pattern on the skin-contacting portion 126 produces
a corresponding
pattern on skin when the personal care product 180 is applied thereto. The
pattern 144 may be
configured to deliver a discontinuous film of product to the skin, resulting
in natural looking
coverage. The pattern 144 may be formed by providing apertures that extend
through the surface
of the skin-contacting portion 126. Alternatively or additionally, the pattern
144 may be formed
from recessed and/or raised portions disposed on the surface of the skin-
contacting portion 126.
In certain embodiments, the personal care product 180 disposed in the package
100 may flow
through apertures of the pattern 144 onto the surface of the skin-contacting
portion 126 during
dispensing and application. In certain embodiments, (e.g., if the pattern 144
includes raised
portions on the surface of the skin-contacting portion 126), the raised areas
may be configured to
pick up the personal care product 180 from, e.g., a trough or tray positioned
in close proximity to
the surface of the skin-contacting portion. In embodiments where the skin-
contacting surface
includes recessed portions disposed on its surface, the personal care product
180 may be
deposited in the recessed portions and subsequently transferred to the skin of
a user.
Exemplary surface patterns 144 for use on the skin-contacting portion 126 are
illustrated
in FIGS. 7A-R and 8a-d. The shapes of the surface features on the skin-
contacting portion 126
that form the pattern 144 may be selected from any suitable shape known in the
art. For example,
the surface features may include donut-shaped dimples (i.e., recessed
portions) which are shallow
in the center and deeper around the outer circumference. Other exemplary
shapes include star-
shaped, square, etc. The skin-contacting portion 126 may configured to include
two or more
zones that include the same or different patterns and/or are formed from the
same or different
materials. Each zone may be further subdivided into two or more different
sections based on the
pattern and/or the material used to form the section. For example, the skin-
contacting portion 126
may include a first zone formed from a relatively soft, pliable material with
a discontinuous dot
pattern on the surface, and a second zone formed from a relatively soft,
absorptive material with
no pattern on the surface. In another example, the entire skin-contacting
portion 126 may be
formed from the same material, but have a first zone with recessed and/or
protruding surface
features and a second zone with apertures. In still another example, the skin-
contacting portion
126 may include a first zone with two differently shaped apertures spaced away
from one another
by a first distance, and a second zone with the same shaped apertures as the
first zone but spaced
apart by a second distance, which is greater than the first distance. In this
example, the distance
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
9
between two apertures is the shortest straight line distance between the
centers of the two
adjacent apertures.
FIG. 6 shows an exemplary embodiment of a package 100 that includes one or
more caps
176 and 178. The caps 176 and/or 178 may be provided to cover the applicator
124 and/or the
blending tool 154. The caps 176, 178 may prevent damage or disfigurement to
the applicator 124
and/or blending tool 154; reduce the likelihood of contacting and/or
contaminating surfaces other
than those intended to receive the personal care product 180; and/or help
prevent moisture loss
from the personal care product 180. In certain embodiments, the caps 176, 178
may be provided
as temporary covers, which are intended to be removed and discarded by the
consumer after
purchase. Alternatively, the caps 176 and/or 178 may be reattachable such that
they can be
reused over the intended life of the personal care product 180 and/or package
100.
The personal care products suitable for use herein may be provided in a
variety of product
forms including brushable gels and creams, non-foaming liquids, mechanically
pumpable liquids,
non-aerosol gels, aerosol gels, aerosol foams, pastes, serums, and sprays.
Example I: a multichromatic, liquid foundation is prepared as follows:
Ingredient Wt%
Colored crosslinked gel 40.00
network"
Dimethicone copolyol 5.00
crosspolymer (KS G21)
Cyclomethicone 19.35
(DC245)
Propylparabens 0.10
Ethylparabens 0.20
Water 15.00
Titanium dioxide 8.25
Iron oxides 1.75
Glycerin 10.00
Benzyl alcohol 0.25
Methylparabens 0.10
Ammonium polyacrylate 0.12
(Darvan 821A*2)
Disodium EDTA 0.10
*1
Colored gel comprising 10% pigments (titanium dioxide and iron oxides) having
a 60 micron average
particle size, approximately 12% polymer, and 78% cyclomethicone fluid.
' Supplied by Vanderbilt.
In a suitable vessel, the water, glycerine, disodium EDTA and benzyl alcohol
are added
and mixed using conventional technology until a clear water phase is achieved.
When the water
CA 02788305 2012-07-26
WO 2011/100664
PCT/US2011/024719
phase is clear add methylparabens and mix again until clear. Then add the
ammonium
polyacrylate, titanium dioxide and iron oxides and mix to disperse. Mix the
resultant phase with
a SiIverson SL2T or similar equipment on high speed (8,000 rpm, standard head)
to fully
deagglomerate the pigments. In a separate vessel, add the KSG21, hydrophobic
titanium dioxide
and iron oxides, DC245 and the parabens. This mixture is milled using a
SiIverson on high speed
until homogeneous. Next, the colored water phase and the colored, silicone
phase are combined
and milled using a SiIverson on high speed until the water is fully
incorporated and an emulsion
is formed. Finally, a colored gel is then chosen so as to be significantly
different in colour to the
blend of titanium dioxide and iron oxides. This is then added and the product
is mixed again
using a SiIverson on high speed. The resulting multichromatic, finished
product is then
incorporated into the appropriate package.
Example II: a skin-care composition is prepared as follows:
Ingredient Wt%
Phase A
Water QS (-20% total water)
Glycerin 10.0000
Allantoin 0.1000
Sodium Ascorbyl Phosphate 0.10000
Vitis Vinifera (Grapeseed) Extract 0.02000
Green Tea Extract 0.1
Propylene Glycol 1.0000
Butylene Glycol 1.03950
Benzyl Alcohol 0.4000
Matrixyl *1 3.0
Glycolic Acid 3.9900
Triethanolamine 1.0000
Sodium Hydroxide 1.1250
Phase B
Water QS (-80% total water)
d-Panthenol 0.5000
Hydroxyethylcellulose 1.00000
Ultrez 21*2
Sepigel 305*3
Simulgel I-NS 100*4
Simulgel EG*
Sepiplus 600*6
AxCel CDG-PX *7 0.10000
After Mixing Phase A into Phase B, add:
Colorona Aborigne Amber *8 0.02000
*1
Anti-aging peptide solution from Sederma, Inc., Edison, NJ.
CA 02788305 2014-03-13
11
= Acrylates/CIO-Ifi alkyl acrylate crosspolymer from Noveon Comma
Specialties, Cleveland, OIL
Polyacrylamide and C 13-14 Isoparaffin and I uureth-7 from SIPPIC, Inc.,
Fairfield, NJ.
= Ilydroxyethyl acrylate/sodium aeryloyldi methyl taurate copolymer and
iFohexadccane and polysorbate 60 from
Inc., Fairfield. NJ.
.5 Sodium acrylatc/sodium acryloyldimethyl taurate copolymer and
is.ohexadecarte and polysorbate 80 from
Ine., Fairfield, NJ.
= Ilydroxyethyl aerylate/sodium acryloyldimethyl taurate copolymer and
isohexadecane anti polysorbate 60 trom
SLPPIC. tnc., lUirfield, NJ.
.7 Mixture of micmlibrous cellulose. xanthan gum. and sodium
carboxymethylcellulose from CP Kelco. San Diego,
CA.
" Mica coated with iron oxide, iron oxide Mack, and titanium dioxide from
EMI) Chemicals, Inc., Gibbstown, NJ.
Phase A materials are blended in a container. Phase B materials are blended in
a separate
container. Phase B is blended into Phase A. The platelet paniculates are added
after combining
Phase A and Phase B.
The dimensions anti values disclosed herein are not to he understood as being
strictly
limited to the exact numerical values recited. instead, unless otherwise
specified, each such
dimensioiì is intended to mean both the recited value and a functionally
equivalent range
surrounding that value. For example, a dimension disclosed as "40 nun" is
intended to mean
"about 40 nun."
The citation of all documents is, in relevant part, not to be construed as an
admission that it is
prior art with respect to the present invention. To the extent that any
meaning or definition of a term
in this written document conflicts with any meaning or definition of the term
in a cited document, the
meaning or definition assigned to the term in this written document shall
govern.
While particular embodiments of the present invention have been illustrated
and described, it
would be obvious to those skilled in the art that various other changes and
modifications can be
tnade. The scope of the claims should not be limited by the preferred
embodiments set forth in the
examples, but should be given the broadest interpretation consistent with the
description as a whole.