Note : Les descriptions sont présentées dans la langue officielle dans laquelle elles ont été soumises.
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
METHOD OF PERFORMING A PLURALITY OF SYNTHESIS PROCESSES OF
PREPARING A RADIOPHARMACEUTICAL IN SERIES, A DEVICE AND CASSETTE FOR
PERFORMING THIS METHOD
The invention relates to a method of performing a plurality of synthesis
processes of
preparing a radiopharmaceutical in series, a device for performing this method
and a cassette
for use in the method.
Radioisotopes for PET (Positron Emission Tomography) are generally produced
using a
cyclotron. In the cyclotron charged particles are accelerated thereby gaining
energy. Upon
exiting the cyclotron the accelerated particles hit a target thereby producing
positron emitters.
Fluorine-18 (hereinafter 18F) is produced by proton bombardment of oxygen-18
water
(H2180). The proton interacts with the 180 and produces a neutron and 18F. The
thus
produced 18F is allowed to react with a suitable starting material, thereby
producing an
appropriate tracer (radiopharmaceutical) for diagnosis purposes such as cancer
and brain
disorders.
Many synthetic routes to prepare PET radiopharmaceuticals have been developed
during
recent decades. The great majority of PET tracers are labelled with the
positron-emitting
radioisotopes 11C and 18F (radioactivity decay: half-lives of 20 and 110 min,
respectively).
For 18F based radiopharmaceuticals production two preparation methods have
been
developed and used throughout the world, electrophilic and nucleophilic 18F
fluorination.
These reactions are usually performed in a so called synthesizer. Today
commercially
available synthesizers are highly automated devices for the production of the
tracer, wherein
the direct involvement of operating staff and exposure to radiation is reduced
in order to
protect them against radiation.
After irradiation the produced 18F in a 180 enriched water solution is usually
passed to an
anion exchange material, where the 18F is trapped. The 180 water is collected.
Subsequently the 18F is eluted using typically an eluent like K2CO3. K18F is
not soluble in
organic solvents that are suitable for performing the subsequent nucleophilic
reaction steps.
Therefore a so called phase transfer agent is also added. Typical examples
thereof include
tetra alkyl ammonium salts or aminopolyethers, like Kryptofix . As fluoride is
reactive in water-
free media only, any remaining water is usually removed in one of more
evaporation steps,
typically using dry acetonitrile under a flow of inert gas like helium or
nitrogen. The 18F once
dried and solubilised in the presence of the phase transfer agent is ready for
the main
nucleophilic substitution steps. In the production of 18F-FDG (F-18 fluoro-2-
deoxyglucose)
typically a precursor is added like mannose triflate. This compound has a
triflate group as a
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
- 2 -
suitable leaving group, while the acetyl groups ensure that fluorination only
occurs at the
position of the triflate group. This reaction step is usually carried out at
elevated temperature
like 80-90 C. In the next step the protective acetyl groups are removed by
hydrolysis. Both
basic hydrolysis typically using NaOH and acid hydrolysis using HCI can be
employed. Basic
hydrolysis has the advantage that it can be carried out at room temperature in
a short time
interval, whereas acid hydrolysis frequently requires a substantial higher
temperature and
lasts longer. Finally the thus produced 18F-FDG is purified, which is commonly
performed
using several purification steps using different chromatographic materials.
The synthesizers used can be classified into two categories. A first category
comprises
stationary systems without any removable components. All connections, tubing,
valves,
vessels are permanently installed. After completion of a production run, the
components are
rinsed in a CIP (Clean-In-Place) operation. Although this kind of synthesizers
is said to have
the advantage of cost savings due to reusing its components, complete cleaning
and
sterilization may be difficult to achieve. Moreover, a full CIP operation may
be lengthy,
resulting in a serious downtime of the synthesizer. Additionally waste volumes
resulting from
the CIP operation may be relatively high. Also a cleaning operation may lead
to a drop of
labelling yield. Typically such a stationary system is dedicated to the
production of a single
radiopharmaceutical, because its configuration cannot be easily adapted to
allow production
of another tracer.
A second category comprises synthesizers which are based on the use of
removable kits or
cassettes. In some cassettes the reagents need to be activated prior to use.
Other cassettes
are ready-to-use and need only to be inserted. All process steps including
prior testing
sequences and related process parameters and other data are predetermined and
part of the
software, which is installed in a suitable programmable logical controller
(PLC), server or PC.
Each synthesizer has its own PC, router and PLC, or customized electronic
board. Principally
cassette based synthesizers are useful for performing subsequent syntheses,
which
depending on the selected cassette, reagents kits and software may produce
different
radiopharmaceuticals.
In view of radiation protection (radio-safety) synthesizers are installed in a
so called hot cell, a
protective shielding typically made from lead. The size and amount of
shielding is mainly
dependent on the dimensions and configuration of the synthesizer. Thus
compactness of the
synthesizer is highly desirable in view of costs and weight of the shielding.
After a production
run, the device contains still radioactive residues, so that manually handling
the synthesizer is
dangerous. Decay periods of more than 12 hours are likely to be observed,
before the
residual activity on the spent cassette has dropped below a certain limit and
the synthesizer
can be accessed safely. This is a serious drawback if multiple batches are to
be produced
during a single day.
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
- 3 -
Several approaches to solve these issues are known from the prior art.
E.g. WO 2012/083094 Al discloses that performing two back-to-back synthesis
runs of
fluciclatide in quick succession on two different cassettes is technically
difficult due to the
residual activity, to which the operator would be exposed during spent-
cassette dismounting
procedures. In order to shield the operating staff from this residual activity
on the cartridge
during the short time required for this dismounting procedure it is proposed
in this document
to provide a shielding collar specific for a separation cartridge used on the
synthesis cassette.
WO 2006/119226 A2 has disclosed an apparatus and method for making
radiopharmaceuticals, which synthesizer comprises a stationary processor
having a
disposable kit interface planar structure, a plurality of rotary actuators and
push-on fluidic
connectors protruding from this interface, structure for releasably
interfacing a disposable kit
to the actuators and connectors, and associated disposable kit. Linear
actuators translate the
kit toward and after processing from supports on the processor, so that the
kit can fall in a
suitable container.
One way of preparing multiple batches of radiopharmaceuticals, which may be
the same or
different, is providing a number, e.g. four, of synthesizers in one or more
hot cells, each
synthesizer being controlled with its own dedicated computer, PLC and so
forth, including
waste containers. Such a setup is spacious and expensive in view of shielding
and
equipment.
The invention aims at providing a method and device for performing a plurality
of synthesizing
processes of preparing batches of one or more radiopharmaceuticals in series,
of which the
expenses in equipment are reduced, while ensuring minimal involvement of
operating staff
and relatively short downtime.
Another object of the invention is to provide such a method and synthesizer
allowing an
economical waste management.
According to the invention a method of performing a plurality of synthesis
processes of
preparing a radiopharmaceutical in series comprises carrying out a first
synthesis run
comprising the steps of:
a) providing water containing 18F;
b) trapping the 18F from the water provided in step a) on an anion exchange
material;
c) eluting the trapped 18F from the anion exchange material to a reaction
vessel of a first
radiopharmaceutical synthesis cassette;
d) preparing a radiopharmaceutical incorporating the eluted 18F using the
first
radiopharmaceutical synthesis cassette;
wherein steps a)-d) are repeated in at least one subsequent run using another
radiopharmaceutical synthesis cassette; and
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
- 4 -
wherein the method comprises a reconditioning step of said anion exchange
material
between two consecutive runs.
In the method according to the invention a series of consecutive batchwise
synthesis
processes of preparing a radiopharmaceutical is performed using different
radiopharmaceutical synthesis cassettes, except for the trapping of 18F
supplied from the
target, on an anion-exchanger and subsequent elution thereof. These trapping
and elution
steps are carried out using the same anion-exchanger material and associated
equipment for
each synthesis of the series. Between subsequent runs the anion-exchanger is
reconditioned.
It has been discovered that reconditioning of the anion-exchanger can be
performed rather
easily, while maintaining its trapping capacity and without cross-
contamination occurring. The
method according to the invention allows to install the equipment and
chemicals for trapping,
elution and reconditioning, preferably as a cassette, as well as the various
cassettes for the
synthesis runs, which may be the same or different, in one time in a
synthesizer, and to
perform the various subsequent synthesis runs without the need of accessing
the hot cell,
thereby avoiding the operator being exposed to residual activity. Thus the
subsequent runs
are independent and do not need a full CIP operation or cleaning of a spent
cassette. A main
advantage is that the series of processes can be controlled using a single
server, router and
PLC. Another important advantage is related to waste management. A single
recovery bottle
for 180 water suffices, as well as a single waste bottle for the
reconditioning solutions used
for reconditioning the anion-exchanger. The waste liquids resulting from the
subsequent
synthesis runs can be collected in a single waste bottle as well. Compared to
the amount of
waste produced upon a full CIP operation, the volume of the spent
reconditioning liquids is
small in the method according to the invention. This is beneficial in view of
waste
management. Furthermore as the actual 18F labelling synthesis itself is
carried out each time
on a fresh cassette, labelling yield does not suffer from deterioration due to
repeatedly
cleaning. In addition reuse of the anion exchanging material reduces the costs
of the series of
synthesis reactions.
In the context of this application a cassette comprises the
radiopharmaceutical synthesis
process specific hardware components and chemicals required for performing the
respective
synthesis. For example, such a radiopharmaceutical synthesis cassette
comprises one or
more manifolds provided with suitable valves that can be operated by a
synthesizer, having a
plurality of connections such as luer connectors, tubing, one or more reaction
vessel(s) and
vials containing the necessary reagents and other liquids, optionally
separation and/or
purification cartridges. Suitably the vials containing the necessary reagents
and other liquids
may be obtained as a separate kit of chemicals, while the other hardware
components of the
cassette are obtained as a pre-mounted assembly.
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
- 5 -
The design of the method also allows to use a compact synthesizer. E.g. a
synthesizer for
performing the method according to the invention producing three subsequent
batches of
18F-FDG can be installed in a small space having dimensions (width X height X
depth) of 560
mm X 420 mm X 360 mm.
A preferred anion exchange material comprises a quaternary ammonium anion
exchange
material, in particular quaternary methyl ammonium (QMA), such as the silica-
based ion
exchanger cartridges loaded with QMA, e.g. Sep-Pak Accell Plus QMA Plus Light
Cartridge
available from Waters Corporation, or Chromafix PS-HCO3 available from MACH
EREY-
NAGEL GmbH & Co. KG. These QMA cartridges can be easily reconditioned using a
carbonate solution. The carbonate concentration has appeared not very
critical. A suitable
concentration is in the range of 0,01-5 M. For example, both a 1M and a 0.05 M
K2CO3
solution have proven to allow successful reconditioning. In a preferred
embodiment the
carbonate solution is prepared in situ by diluting a concentrated carbonate
solution with water
allowing to reduce the dimensions of the container (bottle) for the carbonate
solution. E.g. a
1M K2CO3 solution can be easily diluted with water for injection in the
trapping, elution and
reconditioning cassette by suitable operation.
A rinsing operation comprising one or more rinsing steps with only pure water
can be used for
reconditioning QMA as anion exchange material. However, pure water will not
remove the
metallic impurities derived from the target and also trapped on the QMA, and
these impurities
maybe released by the eluent in a next elution step. Thus, using pure water
may cause cross-
contamination between subsequent runs.
It is also possible to regenerate the anion exchange material with the
preferred eluent mixture
itself (discussed hereinbelow), but due to the low carbonate concentration
thereof, the volume
required for reconditioning will be higher than with a carbonate solution
having a higher
concentration as exemplified above. The use of the preferred eluent mixture
also as a
reconditioning agent for reconditioning the anion exchange material will
additionally result in
waste of the expensive crown ether (= phase transfer agent).
It is possible to elute the trapped 18F from the anion-exchanger using only an
aqueous
carbonate solution. Ammonia in water works as well. Suitably, in these cases a
phase
transfer agent is added to the reaction vessel in the synthesis cassette.
Preferably the phase
transfer agent is a crown ether like Kryptofix 2.2.2., or a tetra alkyl
ammonium salt. More
preferably, the eluent is a mixture comprising carbonate, phase transfer
agent, water and
acetonitrile.
A suitable example comprises a mixture of 0,7 - 7 mg of K2CO3, 0,3 - 1 mL of
CH3CN, 5 ¨ 30
mg of Kryptofix 2.2.2. in 0,1 - 0,5 mL of H20. Here the amount of potassium
carbonate can
be replaced by MexHyCO3 wherein Me represents an alkali metal and x is 1-2 and
x + y = 2,
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
- 6 -
such as Li2003, Cs2003, NaHCO3, KHCO3. Another suitable mixture is composed of
75mM
nBU4NHCO3, 750pL H20, Et0H (stabilizer).
In this way the phase transfer agent and organic solvent like acetonitrile for
drying do not
need to be part of the synthesis cassettes used for the actual production of
the
radiopharmaceutical in question.
The method according to the invention can be used for a variety of
radiopharmaceuticals
based on 18F.
Examples include
18F-FDG ([189- fluoro-2-deoxyglucose),
FMISO (1-(2-nitro-imidazolyI)-3-[18F]-fluoro-2-propanol; 1H-1-(3-[18F]-
fluoro-2-
hydroxypropy1)-2-nitroimidazole),
NaF (sodium [189-fluoride),
18F-FLT (3'-deoxy-3'-[18F] fluoro thymidine):
18F-FET (0-(2418Fpluoro ethyl)-L-tyrosine)
18F-FES (16 a 418F]fluoro-17 13 -estradiol),
FCHOL ([18F] fluorocholine)
FACETATE ([18F] fluoroacetate)
FDGal ([18F] fluorodeoxygalactose)
F DOPA (L-6-[18F] fluoro-3, 4-dihydroxyphenylalanine)
18SFB (N-succinimidyl 4-[18F]fluorobenzoate).
Production of 18F-FDG is a preferred synthesis process.
As described hereinbefore, the subsequent synthesis runs can be directed to
the preparation
of radiopharmaceuticals, which may be the same or different. In a preferred
embodiment all
subsequent synthesis runs produce the same radiopharmaceutical.
According to a second aspect the invention provides a device for performing a
plurality of
synthesis processes of preparing a radiopharmaceutical in series, in
particular as explained
above, comprising
a frame or housing
an inlet for introducing water containing 18F;
an anion-exchanger comprising an anionic exchange material connected to said
inlet;
an eluent container comprising an eluent connected to said anion-exchanger;
a recondition container comprising a reconditioning agent connected to said
anion-exchanger,
distribution means for selectively supplying eluted 18F to a
radiopharmaceutical synthesis
cassette ;
at least two radiopharmaceutical synthesis cassettes, each cassette being
connected to said
distribution means.
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
- 7 -
The frame or housing has the function of providing a structure for mounting
the other
components, in particular the necessary tubes and other conduits including
valves and
actuators thereof, the reagents and waste containers and cassettes. The
radiopharmaceutical
synthesis cassettes are as described above.
In a preferred embodiment thereof in the device according to the invention the
components,
reagents necessary for trapping 18F, elution thereof and reconditioning of the
anionic
exchange material are contained in a ready-to-use cassette.
Advantageously the device according to the invention comprises a single server
loaded with
suitable software, router and PLC for inputting and selecting processes and
controlling the
device.
According to a third aspect the invention also provides a cassette for use in
the device
according to the invention, which cassette comprises
at least one manifold provided with a plurality of valves connectable to and
operable by the
device according to the invention
an anion-exchanger comprising an anionic exchange material;
an eluent container comprising an eluent
a recondition container comprising a reconditioning agent.
The anion-exchanger, eluent container and recondition container are
connectable to the at
least one manifold.
In a preferred embodiment thereof the recondition container contains a
concentrated
carbonate solution, and the cassette also is provided with a container
comprising water.
The advantages as explained above with respect to the method according to the
invention are
applicable in a similar way to the device and cassette according to the
invention.
The invention will be illustrated by reference to the attached drawing,
wherein:
Fig. 1 shows a diagrammatic view of a part of a synthesizer, in particular a
cassette suitable
for trapping 18F, eluting and reconditioning;
Fig. 2 shows an example of a process scheme for performing a number of
synthesis
processes according to the invention; and
Fig. 3 shows a diagrammatic view of a synthesizer for performing a number of
synthesis
processes according to the invention.
In Fig. 1 a part of a synthesizer being an embodiment of a disposable
reconditioning cassette
according to the invention, for trapping 18F on an anionic exchange material,
subsequent
elution and reconditioning of the anion exchange material, is shown
diagrammatically. In the
embodiment shown the cassette indicated in its entirety by reference numeral
10, comprises
a first manifold 12 and a second manifold 14. Each manifold 12, 14 comprises
five 3way
valves 16, individually indicated 16a through 16e, and 16f-16j respectively.
These reference
numerals are also used to indicate the respective positions. The valves 16 are
preferably
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
- 8 -
operated by compressed air as disclosed in WO 2013/127439 Al, which is
incorporated by
reference. Typically connections with tubing occurs by luerlock connectors 17,
while (reagent)
vials and other bottles are usually closed by suitable septums 19 that are
spiked.
Water containing 18F is derived from a cyclotron (not shown) and introduced at
16j and
trapped on anion-exchanger 18, such as a Sep-Pak Accell Plus QMA Carbonate
Plus Light
cartridge available from Waters Corporation, which is connected to the right
hand ends 20, 22
of manifolds 12 and 14 via tubing 24. The water is removed at 16e and
collected into a bottle
or other container (not shown). Once the 18F is trapped by anion-exchanger 18
an eluent,
typically a mixture of (potassium) carbonate, a transfer agent like Kryptofix,
water and
acetonitrile, from vial 26 is metered in syringe 28 via valves16d, 16a and
then passed over
the anion-exchanger 18 thereby extracting the 18F and sending it to a reaction
vessel of one
of the synthesis cassettes (see Fig. 3) at valves 16f-h. The left end 30 of
manifold 12 is
connected to a source of an inert gas such as nitrogen or helium.
For reconditioning the anion-exchanger 18 reconditioning agent, typically an
aqueous
carbonate solution, is used. In the embodiment shown in Fig. 1, this solution
is prepared in
situ in syringe 28 from a high molar K2CO3 solution in vial 32 at position 16c
and water from
bottle 34 at position 16b. It will be recognized that the vials 26, 32, 34 can
be placed in any
order. Then the diluted K2CO3 solution thus prepared is ejected from syringe
28 over anion-
exchanger 18 to a waste bottle (not shown) connected to the left hand end 36
at position 16f
of manifold 14. Subsequently one or more rinsing steps are carried out with
water from bottle
34 using syringe 28. The spent water is collected in the same waste bottle at
36. The anion-
exchanger 18 is dried using the inert gas introduced at 30. The water at 16i
is used for each
synthesis step requiring water, that is to say water is sampled from 16i for
subsequent
synthesis runs. Cross-contamination is not possible because the water arrives
from 16 f g h in
the different runs.
Fig. 2 shows an example of a process scheme for performing multiple reactions
in series.
The series of reactions start with supply of 18F in water from a target and
trapping thereof on
the anion-exchanger. Then the trapped 18F is eluted to radiopharmaceutical
synthesis
process 1. The anion-exchanger is reconditioned. Thereafter the sequence of
steps A)
through D) is repeated using the same anion-exchanger, but the eluted 18F is
now guided to
a second synthesis process 2. Upon finishing this synthesis process, the anion-
exchanger is
reconditioned once more, and the sequence of steps A) through D) is repeated
for the last
time.
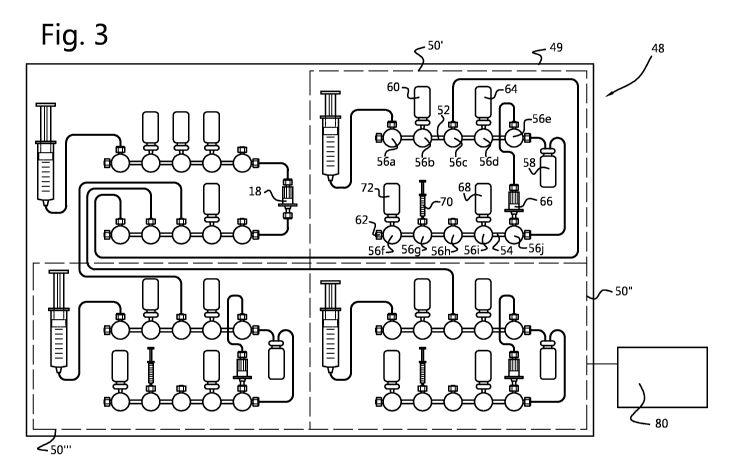
Fig. 3 shows an embodiment of a synthesizer 48 having a frame or housing 49
allowing
insertion of cassettes, wherein 3 consecutive batches of 18F-FDG are prepared
using - in this
embodiment - identical radiopharmaceutical synthesis cassettes 50
(diagrammatically shown
in broken lines). Each cassette comprises two manifold 52 and 54, and each
manifold has
CA 02986186 2017-11-16
WO 2016/184980
PCT/EP2016/061319
- 9 -
five 3way valves 56 (or positions), numbered 56a-e and 56f-j respectively. In
the top left part
of the synthesizer the eluent mixture containing 18F is prepared as explained
above with
reference to Fig. 1. This eluent mixture is used in the first synthesis
process of 18F-FDG. The
eluent mixture is introduced in a cassette 50' at position 56c and collected
in reactor vessel
58, which can be heated by heating means (not shown). Therein the eluent
mixture
comprising 18F is dried using acetonitrile from bottle 60. Waste is collected
at left hand end
62 of manifold 54. Precursor is added from vial 64 at position 56d. After
reaction the thus
produced intermediate product, after dilution with water sampled in 16i is
separated on a
suitable solid phase extraction cartridge 66 at position 56j and eluted back
into the reaction
vessel 58 using a suitable eluent such as Et0H from vial 68 at position 56i.
If the chemistry
allows so, the water container or bag may be incorporated in the
radiopharmaceutical
synthesis cassette 50, 50' or 50". The protective groups of the intermediate
product are
removed by means of basic hydrolysis using NaOH contained in syringe 70 at
position 56g.
The final product after buffering with buffer from vial 72 at position 56f is
removed via valve
56h for subsequent formulation and quality control. After approval it can be
used for diagnosis
purposes. The syringe on position 56a is used for the pressurization of vials
60, 64, 72, 68,
and for sampling of chemicals solutions, for dilution of reactive bulk
mixture, and loading of
the separation cartridges.
After this first production run the reconditioning of the anion-exchanger 18
is performed as
outlined above with respect to Fig. 1. Fresh 18F containing water is again
trapped on the thus
reconditioned anion exchanger 18 and subsequently eluted to the second
synthesis process
cassette 50", where 18F-FDG is produced in the same way as described with
respect to
cassette 50'. Also after this second production run reconditioning of the
anion-exchanger
takes place. Then a third batch of eluent mixture containing 18F is prepared
and used in
synthesis process 3 in cassette 50".
The synthesizer 48 is operated by a single control system 80 comprising PLC,
router and
server (PC).
In an embodiment of the synthesizer 48 the cassette 10 and cassettes 50', 50"
and 50- are
releasibly mounted on a fixed upright front plate of the frame or housing 49,
while common
pumps, drivers and other electronics and the like are mounted on a detachable
upright rear
plate (not visible in Fig. 3). For example the rear plate is connected to the
frame or housing
49 using removable hinges, that are horizontally arranged at the bottom. For
access to the
components on the rear plate it is rotated backwards over a certain angle
allowing inspection,
servicing and maintenance of the components, while hooks or other connecting
elements
maintain the rear plate in this inclined position. Upon detachment of these
hooks, the rear
plate can rotate further down to an essentially horizontal position. In this
position the rear
plate can be removed as a module from the synthesizer.